Advanced One-Pot Synthesis of Flupirtine Hydrochloride for Commercial Scale-Up
Advanced One-Pot Synthesis of Flupirtine Hydrochloride for Commercial Scale-Up
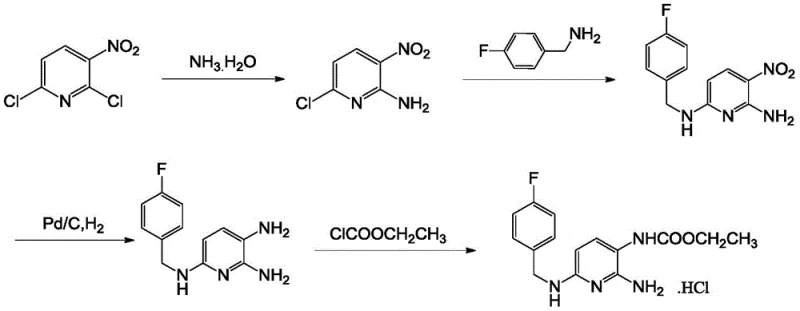
The pharmaceutical industry continuously seeks robust synthetic pathways that balance high purity with operational safety and cost efficiency. A pivotal advancement in this domain is detailed in patent CN109053563B, which discloses a highly efficient method for preparing flupirtine hydrochloride, a critical intermediate for the non-opioid analgesic flupirtine maleate. This technology represents a significant departure from conventional multi-step protections and deprotections, introducing a streamlined "one-pot" strategy that combines catalytic hydrogenation and acylation. For R&D directors and procurement specialists evaluating reliable pharmaceutical intermediate suppliers, this process offers a compelling value proposition by mitigating the risks associated with hazardous reagents while ensuring the production of high-purity active pharmaceutical ingredients (APIs). The innovation lies not just in the chemical transformation but in the holistic optimization of the workflow, addressing long-standing pain points regarding intermediate stability and operator safety.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of flupirtine derivatives has been plagued by complex operational procedures and significant safety hazards. Prior art, such as that described in Chinese patent CN105541705A, relies on the use of expensive starting materials like 2-amino-3-nitro-6-chloropyridine and necessitates the use of di-tert-butyl dicarbonate for amino protection. This approach introduces unnecessary steps, including subsequent deprotection with hydrochloric acid, which complicates the workflow and increases waste generation. Furthermore, traditional methods often employ dangerous catalysts like Raney nickel, which is pyrophoric and poses severe fire risks during handling. Perhaps most critically, the acylation step in older processes typically requires the dropwise addition of ethyl chloroformate by opening the reaction kettle cover. Since ethyl chloroformate is a highly toxic and volatile chemical, this practice exposes operators to significant health risks and creates potential environmental contamination issues, making it unsuitable for modern, green manufacturing standards.
The Novel Approach
In stark contrast, the methodology outlined in CN109053563B revolutionizes the production landscape by adopting 2,6-dichloro-3-nitropyridine as a cost-effective starting material and eliminating the need for amino protection groups entirely. The core innovation is the implementation of a "one-pot" technique where the key intermediate, 2-amino-3-nitro-6-p-fluorobenzylaminopyridine, undergoes simultaneous hydrogenation and acylation within a closed system. By utilizing palladium on carbon (Pd/C) as a safer alternative to Raney nickel and introducing ethyl chloroformate directly into the pressurized hydrogenation vessel, the process completely avoids the need to open the kettle during the addition of toxic reagents. This seamless integration of reduction and acylation not only drastically simplifies the operational procedure but also prevents the deterioration of the sensitive polyaminopyridine derivative, which is prone to oxidation if isolated as a free amine. The result is a cleaner, safer, and more economically viable route to flupirtine hydrochloride.
Mechanistic Insights into Catalytic Hydrogenation and Acylation
The mechanistic elegance of this process centers on the concurrent reduction of the nitro group and the acylation of the resulting amine. In the final step, the purified nitro-intermediate is suspended in a fatty alcohol solvent, such as n-propanol or isopropanol, along with 5% Pd/C catalyst and ethyl chloroformate. Upon the introduction of hydrogen gas at pressures ranging from 1.0 to 2.0 MPa and temperatures between 60-70°C, the nitro group is rapidly reduced to a primary amine. Crucially, the presence of ethyl chloroformate ensures that this newly formed amine is immediately acylated to form the carbamate functionality. This immediate trapping of the reactive amine species is vital because the corresponding 2,3-diamino-6-p-fluorobenzylaminopyridine intermediate is highly susceptible to oxidative degradation and color formation upon exposure to air. By maintaining a closed, hydrogen-rich environment throughout the acylation, the process effectively suppresses side reactions and polymerization, leading to a product with exceptional chromatographic purity.
Furthermore, the purification strategy employed prior to this critical step plays a pivotal role in the overall success of the synthesis. The intermediate 2-amino-3-nitro-6-p-fluorobenzylaminopyridine is refined using fatty alcohol recrystallization before entering the hydrogenation reactor. This pre-purification removes residual starting materials and by-products that could otherwise poison the palladium catalyst or participate in side reactions during the high-pressure hydrogenation phase. The use of excess ammonia water in the initial ammonolysis step also serves a dual function; beyond acting as the nucleophile, it functions as an acid-binding agent during the subsequent substitution with p-fluorobenzylamine. This clever utilization of reagents minimizes the need for additional bases, reducing the salt load in the reaction mixture and simplifying the downstream workup. Such attention to chemical detail ensures that the final flupirtine hydrochloride achieves a purity of greater than 99%, meeting the stringent specifications required for pharmaceutical applications.
How to Synthesize Flupirtine Hydrochloride Efficiently
The synthesis of flupirtine hydrochloride via this patented route involves a logical sequence of ammonolysis, nucleophilic substitution, and a final tandem reduction-acylation. The process begins with the reaction of 2,6-dichloro-3-nitropyridine with aqueous ammonia in an alcoholic solvent, followed by the addition of p-fluorobenzylamine to install the benzyl side chain. After isolating and purifying the nitro-intermediate through recrystallization, the material is subjected to the key one-pot transformation. Detailed standardized operating procedures, including specific molar ratios, temperature ramps, and crystallization protocols, are essential for reproducing the high yields and purity reported in the patent literature. For process chemists looking to implement this technology, adhering to the precise conditions for the hydrogenation pressure and catalyst loading is paramount to achieving optimal results.
- Perform ammonolysis of 2,6-dichloro-3-nitropyridine with ammonia water, followed by substitution with p-fluorobenzylamine to form the key nitro-intermediate.
- Purify the crude 2-amino-3-nitro-6-p-fluorobenzylaminopyridine intermediate using fatty alcohol recrystallization to ensure high purity.
- Execute a one-pot reaction adding ethyl chloroformate and Pd/C catalyst to the wet intermediate, conducting simultaneous hydrogenation and acylation at 60-70°C.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis method translates into tangible strategic benefits beyond mere chemical efficiency. The elimination of expensive protected intermediates and the use of commodity chemicals like 2,6-dichloro-3-nitropyridine significantly lower the raw material cost base. Moreover, the consolidation of the reduction and acylation steps into a single unit operation reduces the overall cycle time, allowing for faster throughput and improved responsiveness to market demand. The enhanced safety profile, characterized by the closed-system handling of toxic reagents and the avoidance of pyrophoric catalysts, also lowers the regulatory burden and insurance costs associated with manufacturing, contributing to substantial cost savings in pharmaceutical intermediate manufacturing.
- Cost Reduction in Manufacturing: The economic advantages of this process are driven by the simplification of the synthetic route and the removal of costly protection-deprotection sequences. By avoiding the purchase of expensive precursors like 2-amino-3-nitro-6-chloropyridine and eliminating reagents such as di-tert-butyl dicarbonate, the direct material costs are drastically reduced. Additionally, the ability to use excess ammonia as an acid scavenger negates the need for purchasing separate organic bases, further streamlining the bill of materials. The high yield of approximately 70% over the final steps, combined with the high purity achieved without extensive chromatography, ensures that waste disposal costs are minimized, leading to a more sustainable and cost-effective production model.
- Enhanced Supply Chain Reliability: From a supply chain perspective, the robustness of this method ensures consistent availability of high-purity flupirtine hydrochloride. The use of stable, commercially available starting materials reduces the risk of supply disruptions often associated with specialized or custom-synthesized intermediates. The simplified workflow, which requires fewer isolation and purification stages, decreases the likelihood of batch failures and production delays. This reliability is crucial for maintaining continuous supply to downstream API manufacturers, ensuring that lead times for high-purity pharmaceutical intermediates are kept to a minimum and that inventory levels can be optimized without the fear of quality deviations.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, utilizing standard hydrogenation equipment and common solvents like alcohols that are easily recovered and recycled. The replacement of hazardous Raney nickel with Pd/C, which can be filtered and potentially regenerated, aligns with modern environmental, health, and safety (EHS) standards. Furthermore, the closed-system acylation prevents the release of volatile organic compounds and toxic vapors into the workplace, ensuring compliance with strict occupational exposure limits. These factors collectively facilitate the commercial scale-up of complex pharmaceutical intermediates, allowing manufacturers to increase capacity from pilot scale to multi-ton production with confidence in both safety and environmental performance.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis is vital for stakeholders evaluating its implementation. The following questions address common inquiries regarding the safety, purity, and mechanistic advantages of the patented process. These insights are derived directly from the experimental data and technical disclosures within the patent documentation, providing a clear picture of the method's capabilities and limitations in a commercial setting.
Q: How does the new process improve safety compared to traditional methods?
A: The novel process eliminates the need for open-kettle dropwise addition of toxic ethyl chloroformate by performing acylation in a closed hydrogenation vessel, significantly reducing operator exposure and environmental risk.
Q: What is the expected purity of flupirtine hydrochloride using this method?
A: Experimental data from the patent indicates that the method consistently yields flupirtine hydrochloride with a chromatographic purity exceeding 99%, with maximum single impurities controlled below 0.1%.
Q: Why is the one-pot hydrogenation and acylation step critical?
A: This step prevents the isolation of the unstable 2,3-diamino intermediate, which is prone to oxidation and discoloration, thereby stabilizing the reaction pathway and simplifying the overall workflow.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Flupirtine Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to meet the evolving demands of the global pharmaceutical market. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovations like the one-pot hydrogenation method described in CN109053563B can be seamlessly transferred to industrial manufacturing. We are committed to delivering flupirtine hydrochloride and other complex intermediates with stringent purity specifications, supported by our rigorous QC labs that utilize state-of-the-art analytical instrumentation to verify every batch. Our dedication to quality assurance guarantees that the products we supply meet the highest international standards, providing our partners with the confidence needed to advance their drug development pipelines.
We invite potential partners to engage with our technical procurement team to discuss how our manufacturing capabilities can support your specific project requirements. By leveraging our expertise in process optimization and cost-effective synthesis, we can provide a Customized Cost-Saving Analysis tailored to your volume needs. We encourage you to contact us today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our commitment to technological excellence can drive value and efficiency in your supply chain.
