Optimizing Pemetrexed Diacid Production: A Strategic Shift to Palladium-Free Synthesis for Global Supply Chains
Introduction to Advanced Pemetrexed Diacid Manufacturing
The pharmaceutical landscape for oncology treatments demands rigorous adherence to purity standards and cost-effective manufacturing protocols. Patent CN103254196A introduces a transformative preparation method for Pemetrexed Diacid, a critical active pharmaceutical ingredient (API) used in the treatment of malignant pleural mesothelioma and non-small cell lung cancer. This technology represents a significant departure from legacy synthetic pathways by eliminating the reliance on transition metal catalysts and hazardous nitro-functionalized intermediates. By leveraging a direct condensation strategy followed by controlled halogenation and cyclization, this method addresses the dual challenges of environmental sustainability and economic efficiency. For global procurement leaders, this patent offers a blueprint for securing a more resilient supply chain for high-value antifolate agents, ensuring that production scales from laboratory bench to multi-ton commercial batches without compromising on quality or regulatory compliance.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Pemetrexed has been plagued by complex multi-step sequences that introduce significant operational bottlenecks. Traditional routes, such as those described in earlier patents like CN1821219A, heavily relied on Heck coupling reactions to construct the carbon skeleton. These processes necessitate the use of expensive palladium catalysts, which not only inflate raw material costs but also create severe downstream purification challenges due to strict limits on residual heavy metals in final drug substances. Furthermore, alternative pathways reported in patents like CN1827604A utilized nitro-functionalized intermediates and Nef reactions. The presence of nitro groups raises substantial red flags regarding genotoxicity, requiring extensive and costly purge studies to ensure patient safety. These conventional methods often involve harsh reaction conditions, including strong acids and bases, which can lead to unpredictable side reactions and lower overall yields, thereby destabilizing the supply chain for this critical cancer medication.
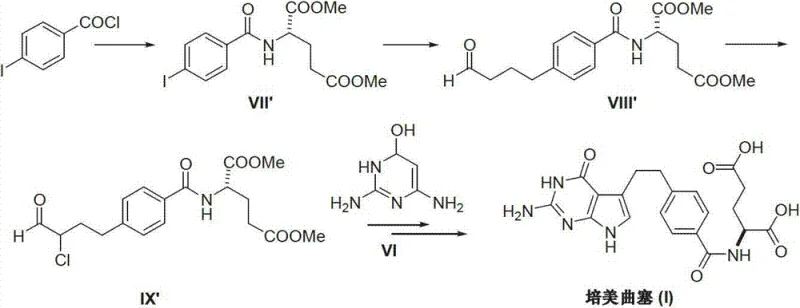
The Novel Approach
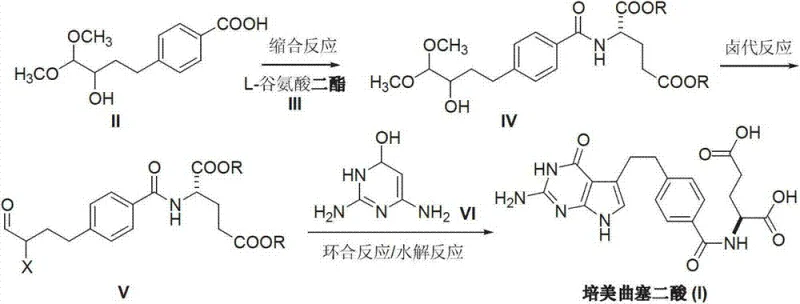
In stark contrast to the cumbersome legacy methods, the novel approach detailed in CN103254196A streamlines the synthesis into a highly efficient three-stage process. The strategy initiates with the condensation of a protected benzoic acid derivative with L-glutamic acid diester, bypassing the need for metal-catalyzed cross-coupling entirely. This is followed by a precise halogenation and acidolysis sequence to activate the side chain, culminating in a cyclization with 2,4-diamino-6-hydroxy pyrimidine. This route is characterized by its use of readily available, commodity-grade raw materials rather than specialized organometallic reagents. By avoiding both palladium and nitro groups, the process inherently reduces the burden on quality control laboratories and waste treatment facilities. The result is a cleaner reaction profile that facilitates easier isolation of the product, directly translating to higher throughput and reduced manufacturing lead times for pharmaceutical producers seeking to optimize their oncology portfolios.
Mechanistic Insights into Condensation and Cyclization Dynamics
The core innovation of this synthesis lies in the strategic selection of coupling agents and the management of reactive intermediates. The initial condensation step utilizes activating agents such as dimethoxy s-triazine in the presence of bases like N-methylmorpholine. This specific combination promotes the formation of the amide bond between the benzoic acid derivative and the glutamic acid ester with high fidelity, minimizing racemization of the chiral center which is critical for the biological activity of the final API. The subsequent halogenation step employs phosphorus tribromide to convert the hydroxyl group into a leaving group, simultaneously facilitating acidolysis to reveal the ketone functionality required for the final ring closure. This tandem transformation is meticulously controlled to prevent over-halogenation or degradation of the sensitive peptide-like backbone. The final cyclization occurs under mild basic conditions in a methanol-water solvent system, where the nucleophilic attack of the diaminopyrimidine on the activated ketone drives the formation of the pyrrolo-pyrimidine core. This mechanistic pathway ensures that impurities are structurally distinct and easily separable, providing a robust impurity profile that satisfies stringent regulatory requirements.
Furthermore, the avoidance of nitro-group chemistry fundamentally alters the safety profile of the manufacturing process. In traditional Nef reaction pathways, the conversion of nitro groups to carbonyls often generates hazardous byproducts and requires careful monitoring for mutagenic alerts. By circumventing this chemistry, the novel route eliminates a entire class of potential genotoxic impurities at the source. This "safety by design" approach simplifies the toxicological assessment of the drug substance, reducing the time and resources required for regulatory filings. The hydrolysis step at the end of the process is performed using standard alkali metal hydroxides, which are inexpensive and easy to handle on a large scale. The overall mechanism demonstrates a sophisticated understanding of green chemistry principles, prioritizing atom economy and hazard reduction while maintaining the structural integrity necessary for potent antineoplastic activity.
How to Synthesize Pemetrexed Diacid Efficiently
The synthesis of Pemetrexed Diacid via this patented route involves a logical sequence of condensation, functional group modification, and heterocyclic ring formation. The process begins with the activation of the carboxylic acid moiety of the benzoic acid derivative, followed by coupling with the amino group of the glutamic acid ester. Subsequent treatment with a halogenating agent converts the protected alcohol into a reactive haloketone precursor. Finally, condensation with the pyrimidine diamine and hydrolysis yields the target diacid. This methodology is designed for scalability, utilizing solvents and reagents that are compatible with standard stainless steel reactor setups found in most fine chemical manufacturing plants. The detailed standardized synthesis steps are provided below for technical reference.
- Perform a condensation reaction between 4-[(4,4-dimethoxy-3-hydroxyl) butyl] benzoic acid and L-glutamic acid diester using a coupling agent like dimethoxy s-triazine to form the amide intermediate.
- Execute a halogenation and acidolysis sequence on the intermediate using phosphorus tribromide to generate the reactive 4-oxo-3-halo butyl ketone species.
- Conduct a cyclization reaction with 2,4-diamino-6-hydroxy pyrimidine followed by basic hydrolysis to yield the final Pemetrexed Diacid product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthesis route offers compelling strategic advantages that extend beyond simple unit cost calculations. The primary benefit stems from the decoupling of production from the volatile market for precious metals. By eliminating the need for palladium catalysts, manufacturers can insulate their cost structures from the fluctuations inherent in the platinum group metals market. Additionally, the removal of heavy metal catalysts simplifies the purification workflow, reducing the consumption of scavenger resins and specialized filtration media. This leads to a significant reduction in the overall cost of goods sold (COGS) and shortens the batch cycle time, allowing for faster response to market demand spikes. The use of common organic solvents and reagents further enhances supply chain resilience, as these materials are widely sourced and less prone to logistical disruptions compared to specialized organometallic complexes.
- Cost Reduction in Manufacturing: The elimination of expensive palladium catalysts and the associated purification steps results in substantial cost savings. Without the need for trace metal removal technologies, the downstream processing becomes more efficient, reducing solvent usage and waste disposal costs. The use of commodity chemicals like phosphorus tribromide and dimethoxy s-triazine ensures that raw material procurement remains economical and predictable, driving down the overall manufacturing expense for the API intermediate.
- Enhanced Supply Chain Reliability: The reliance on readily available starting materials such as substituted benzoic acids and glutamic acid esters ensures a stable supply base. Unlike complex vinyl-alcohol derivatives required for Heck reactions, these precursors are produced by multiple global suppliers, mitigating the risk of single-source dependency. This diversification of the supply base enhances continuity of supply, ensuring that production schedules can be maintained even during regional logistical challenges or raw material shortages.
- Scalability and Environmental Compliance: The process is inherently scalable due to the absence of hazardous nitro intermediates and the use of mild reaction conditions. This simplifies the environmental health and safety (EHS) permitting process for new manufacturing lines. The reduction in genotoxic impurities lowers the burden on waste treatment facilities and reduces the regulatory complexity associated with handling hazardous waste streams. Consequently, the process supports sustainable manufacturing goals and facilitates easier expansion of production capacity to meet growing global demand for oncology therapies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the patent specifications and are intended to clarify the operational benefits for potential partners. Understanding these nuances is essential for evaluating the feasibility of technology transfer and long-term supply agreements.
Q: Why is the elimination of palladium catalysts critical in Pemetrexed synthesis?
A: Traditional Heck coupling routes rely on expensive palladium catalysts which are difficult to remove completely. Residual heavy metals pose significant toxicity risks and require costly purification steps to meet strict ICH Q3D guidelines for elemental impurities in pharmaceutical products.
Q: How does this novel route improve safety regarding genotoxic impurities?
A: Unlike previous methods that utilized nitro-functionalized intermediates and Nef reactions, this patented process avoids nitro groups entirely. This significantly reduces the risk of forming genotoxic impurities, simplifying the safety assessment and regulatory filing process.
Q: What are the key raw materials required for this synthesis?
A: The process utilizes readily available starting materials including 4-[(4,4-dimethoxy-3-hydroxyl) butyl] benzoic acid, L-glutamic acid diesters, and 2,4-diamino-6-hydroxy pyrimidine, ensuring a stable and scalable supply chain without reliance on complex vinyl-alcohol derivatives.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pemetrexed Diacid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust synthetic routes in the production of life-saving oncology medications. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from pilot plant to full-scale manufacturing is seamless. We are committed to delivering high-purity Pemetrexed Diacid that meets stringent purity specifications, utilizing our rigorous QC labs to monitor every step of the synthesis. Our facility is equipped to handle the specific reagents and conditions required by this patented process, guaranteeing a consistent supply of material that adheres to global regulatory standards.
We invite pharmaceutical partners to collaborate with us to leverage this advanced manufacturing technology. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out for specific COA data and route feasibility assessments to determine how this optimized synthesis can enhance your supply chain efficiency and reduce your overall production costs. Let us be your strategic partner in delivering high-quality antifolate intermediates to the global market.
