Advanced Synthesis of BN-NP Perylene Derivatives for Next-Generation OLED Applications
Advanced Synthesis of BN-NP Perylene Derivatives for Next-Generation OLED Applications
The rapidly evolving landscape of organic optoelectronics demands materials with precise electronic properties and structural stability. A significant breakthrough in this domain is documented in patent CN114853794A, which discloses a robust preparation method for a novel polycyclic aromatic hydrocarbon known as BN-NP. This compound features pyrrole and boron-nitrogen units strategically embedded in the bay areas on both sides of a perylene core, creating a unique zigzag edge condensed ring structure. For R&D directors and procurement specialists seeking a reliable OLED material supplier, this technology represents a pivotal shift towards more efficient manufacturing of high-performance electronic chemicals. The patent outlines a bottom-up synthetic strategy that not only simplifies the construction of complex heteroatom-doped frameworks but also ensures high purity and yield, addressing critical bottlenecks in the supply chain of advanced display materials.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of boron-nitrogen doped polycyclic aromatic hydrocarbons (PAHs) has been fraught with significant challenges that hinder their widespread commercial adoption. Conventional routes often rely on harsh reaction conditions, requiring extreme temperatures or highly reactive reagents that compromise the integrity of the delicate conjugated systems. Furthermore, introducing heteroatoms like boron and nitrogen into specific positions of a rigid perylene skeleton typically involves multi-step protection and deprotection sequences, leading to cumulative yield losses and increased waste generation. The lack of regioselectivity in older methodologies often results in complex mixtures of isomers, necessitating expensive and time-consuming purification processes such as preparative HPLC. These inefficiencies translate directly into higher production costs and longer lead times, making it difficult for manufacturers to achieve cost reduction in electronic chemical manufacturing while maintaining the stringent quality standards required for optoelectronic applications.
The Novel Approach
In stark contrast, the methodology presented in CN114853794A offers a streamlined, two-step pathway that elegantly overcomes these historical obstacles. The process begins with a transition metal-catalyzed C-N coupling reaction to install the pyrrole unit, followed by a direct electrophilic borylation to embed the boron-nitrogen unit. This approach eliminates the need for excessive protecting groups and allows for the direct functionalization of the perylene core under relatively mild conditions. By utilizing a bottom-up strategy, the synthesis ensures precise control over the molecular architecture, resulting in a product with a large conjugated system and a two-dimensional plane bending structure. The operational simplicity is further enhanced by the use of common organic solvents and standard purification techniques like silica gel column chromatography. This novel route not only accelerates the development timeline for new materials but also provides a scalable framework for the commercial scale-up of complex polymer additives and optoelectronic intermediates.
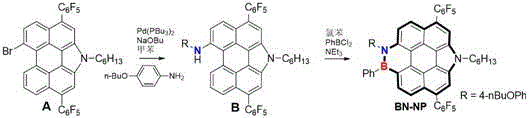
Mechanistic Insights into Pd-Catalyzed C-N Coupling and Electrophilic Borylation
The core of this synthetic innovation lies in the sophisticated interplay between palladium catalysis and electrophilic substitution mechanisms. In the first stage, the reaction utilizes bis(tri-tert-butylphosphine)palladium as a catalyst to facilitate the C-N bond formation between the brominated perylene precursor (Compound A) and tetrabutoxyaniline. The mechanism likely proceeds through a standard catalytic cycle involving oxidative addition of the aryl bromide to the Pd(0) center, followed by ligand exchange with the amine and subsequent reductive elimination to form the C-N bond. The use of sodium tert-butoxide as a base is critical for deprotonating the amine intermediate, thereby driving the equilibrium towards the formation of Compound B. This step is conducted in anhydrous toluene at 130°C for 12 hours, conditions that are optimized to balance reaction kinetics with the thermal stability of the bulky perylene substrate, ensuring high conversion rates without significant decomposition.
Following the isolation of Intermediate B, the second stage involves an electrophilic borylation reaction that introduces the boron atom into the conjugated system. This transformation is achieved by reacting Compound B with phenylboron dichloride in the presence of triethylamine as a base scavenger. The mechanism involves the activation of the aromatic ring towards electrophilic attack, likely facilitated by the electron-rich nature of the newly formed pyrrole-fused system. The reaction is performed in anhydrous chlorobenzene at 140°C for 5 hours, a temperature sufficient to overcome the activation energy barrier for borylation while maintaining the structural integrity of the molecule. The resulting BN-NP molecule exhibits a unique electronic distribution due to the isoelectronic replacement of C=C units with B-N units, which significantly enhances the fluorescence quantum yield and introduces dipole moments essential for its sensing capabilities. This precise mechanistic control allows for the production of high-purity OLED material with consistent batch-to-batch reproducibility.
How to Synthesize BN-NP Efficiently
The synthesis of BN-NP is designed to be accessible for laboratory-scale optimization while retaining the robustness necessary for industrial translation. The protocol relies on widely available reagents and standard inert atmosphere techniques, minimizing the need for specialized equipment. The process is divided into two distinct stages: the formation of the nitrogen-doped intermediate and the subsequent boron doping. Each step has been optimized for yield and purity, with detailed parameters provided in the patent examples to guide process engineers. For teams looking to implement this technology, understanding the specific stoichiometry and workup procedures is crucial for maximizing efficiency. The detailed standardized synthesis steps for producing this advanced material are outlined in the guide below.
- Perform Pd-catalyzed C-N coupling of Compound A with tetrabutoxyaniline in anhydrous toluene at 130°C for 12 hours to yield Intermediate B.
- Purify Intermediate B via silica gel column chromatography using a petroleum ether and dichloromethane mixture.
- Conduct electrophilic borylation of Compound B with phenylboron dichloride in anhydrous chlorobenzene at 140°C for 5 hours to obtain final BN-NP.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain and procurement perspective, the adoption of the BN-NP synthesis route described in CN114853794A offers substantial strategic benefits. The primary advantage lies in the drastic simplification of the manufacturing workflow, which directly correlates to reduced operational expenditures. By eliminating complex protection-deprotection sequences and utilizing high-yielding catalytic steps, the overall material throughput is significantly improved. This efficiency gain means that manufacturers can produce larger quantities of the final active material with fewer raw material inputs, effectively lowering the cost of goods sold. Furthermore, the use of common solvents like toluene and chlorobenzene simplifies solvent recovery and recycling processes, contributing to a more sustainable and cost-effective production cycle that aligns with modern green chemistry initiatives.
- Cost Reduction in Manufacturing: The economic viability of this process is underpinned by the exceptional yields reported in the patent examples, particularly in the final borylation step where yields reach up to 97.5%. Such high efficiency minimizes the loss of valuable intermediates, which is a common cost driver in the synthesis of complex polycyclic aromatics. Additionally, the avoidance of expensive noble metal catalysts in the second step and the use of relatively inexpensive phosphine ligands in the first step help keep catalyst costs manageable. The purification process, relying on standard silica gel chromatography rather than preparative HPLC, further reduces downstream processing costs, making the final product more competitive in the global market for electronic chemicals.
- Enhanced Supply Chain Reliability: The reliance on commercially available starting materials, such as Compound A derivatives and phenylboron dichloride, ensures a stable supply chain that is less susceptible to disruptions. Unlike proprietary reagents that may have single-source suppliers, the reagents used in this protocol are commodity chemicals with established global supply networks. This accessibility reduces the risk of raw material shortages and allows for flexible sourcing strategies. Moreover, the robustness of the reaction conditions, which tolerate standard laboratory variations, ensures consistent production output, enabling suppliers to meet tight delivery schedules and maintain inventory levels required by downstream electronics manufacturers.
- Scalability and Environmental Compliance: The synthetic route is inherently scalable, as the reaction conditions (temperature and pressure) do not require extreme engineering controls that are difficult to replicate on a large scale. The use of closed systems for handling volatile solvents and the straightforward filtration workups facilitate safe scale-up from grams to kilograms. From an environmental standpoint, the high atom economy of the coupling reactions and the ability to recover solvents reduce the volume of hazardous waste generated. This compliance with environmental regulations simplifies the permitting process for manufacturing facilities and reduces the long-term liabilities associated with waste disposal, making it an attractive option for companies focused on sustainable operations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of BN-NP. These answers are derived directly from the technical specifications and experimental data provided in the patent documentation. They are intended to clarify the feasibility of the synthesis route and the performance characteristics of the final material for potential partners and stakeholders. Understanding these details is essential for evaluating the integration of this material into existing product lines or research pipelines.
Q: What are the key advantages of the BN-NP synthesis method described in CN114853794A?
A: The method offers a bottom-up synthesis strategy that introduces pyrrole and boron-nitrogen units into the perylene bay areas with high yields (up to 97.5%) and simple operational conditions, avoiding complex multi-step protections.
Q: What are the primary applications of the BN-NP compound?
A: BN-NP is designed for organic optoelectronic materials, exhibiting sensitive fluoride ion and pyridine monitoring capabilities, excimer emission, and global diamagnetism suitable for advanced sensor and display technologies.
Q: Is the synthesis process scalable for industrial production?
A: Yes, the process utilizes standard transition metal catalysis and common solvents like toluene and chlorobenzene, with straightforward purification via column chromatography, indicating strong potential for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable BN-NP Supplier
The development of advanced materials like BN-NP requires a partner with deep technical expertise and a commitment to quality. NINGBO INNO PHARMCHEM stands ready to support your organization in bringing this innovative technology to market. As a leading CDMO, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and reliability. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of BN-NP meets the exacting standards required for high-performance optoelectronic applications. We understand the critical nature of material consistency in the electronics industry and are dedicated to delivering products that perform flawlessly in your devices.
We invite you to collaborate with us to explore the full potential of this patented synthesis route. Our team is prepared to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and production goals. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments. By partnering with NINGBO INNO PHARMCHEM, you gain access to a reliable supply chain and a wealth of chemical expertise, positioning your company at the forefront of the next generation of organic electronic materials.
