Revolutionizing Organic Electronics: Inside-Out Synthesis of Triphenylene Derivatives for Commercial Scale-Up
The landscape of organic optoelectronic materials is undergoing a significant transformation driven by the need for more efficient and scalable synthetic routes for polycyclic aromatic hydrocarbons (PAHs). Patent CN110105160B introduces a groundbreaking methodology centered on a novel triphenylene alkyne-type precursor, specifically 3,7,10-tris(trimethylsilyl)-2,6,11-tris(trifluoromethanesulfonate)triphenylene. This innovation addresses long-standing challenges in the synthesis of discotic liquid crystals and charge transport materials by shifting the synthetic paradigm from an "outside-in" cyclotrimerization approach to a more controlled "inside-out" growth strategy. By establishing the triphenylene core first and functionalizing it outwardly through Diels-Alder reactions with diene-like derivatives, this technology offers a robust pathway to complex, star-shaped conjugated systems. For R&D directors and procurement specialists in the electronic chemicals sector, this represents a critical advancement in achieving high-purity OLED materials with reduced process complexity and environmental impact.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of triphenylene-centered derivatives has relied heavily on the cyclotrimerization of single-branched aryne precursors. As illustrated in the conventional synthesis of compounds like TM-1, this process typically necessitates the use of palladium catalysts, such as tetrakis(triphenylphosphine)palladium(0), alongside cesium fluoride promoters. 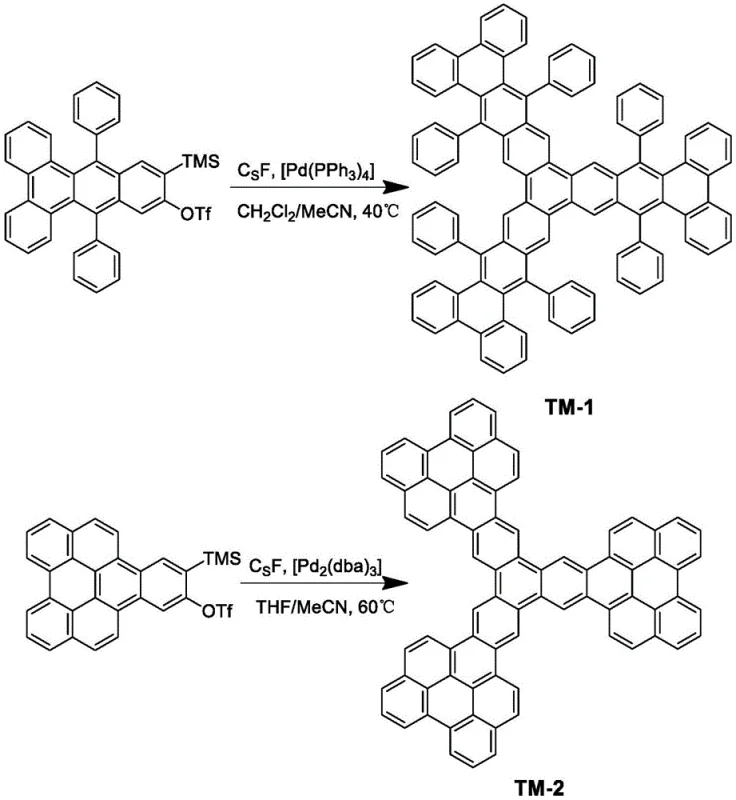 This conventional route is fraught with significant inefficiencies; the reaction conditions are often uncertain and harsh, leading to incomplete conversions and the formation of troublesome dimer by-products that are difficult to separate. Furthermore, the reliance on precious metal catalysts introduces substantial cost burdens and necessitates rigorous purification steps to meet the stringent purity specifications required for electronic applications. The yield limitations are stark, with traditional methods for TM-1 reporting yields as low as 22%, indicating a massive loss of valuable starting materials and a bottleneck for commercial scale-up of complex polymer additives or small molecule emitters.
This conventional route is fraught with significant inefficiencies; the reaction conditions are often uncertain and harsh, leading to incomplete conversions and the formation of troublesome dimer by-products that are difficult to separate. Furthermore, the reliance on precious metal catalysts introduces substantial cost burdens and necessitates rigorous purification steps to meet the stringent purity specifications required for electronic applications. The yield limitations are stark, with traditional methods for TM-1 reporting yields as low as 22%, indicating a massive loss of valuable starting materials and a bottleneck for commercial scale-up of complex polymer additives or small molecule emitters.
The Novel Approach
In contrast, the novel approach detailed in the patent utilizes a pre-synthesized triphenylene core equipped with orthogonal reactive groups—trimethylsilyl (TMS) and trifluoromethanesulfonate (OTf). This strategic design allows for the in situ generation of a highly reactive triphenylene aryne intermediate under mild fluoride activation. Instead of struggling to form the core from three separate pieces, the chemistry builds the complex periphery onto a stable central nucleus. 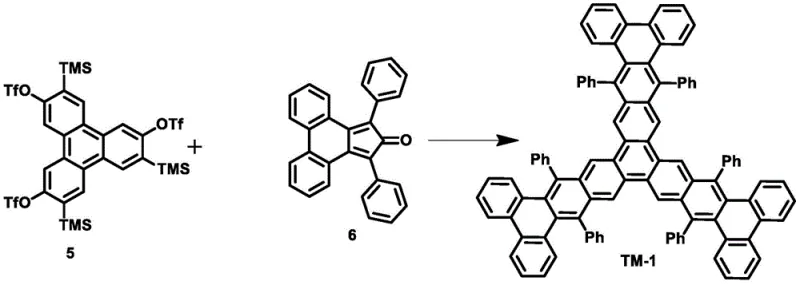 This "inside-out" methodology dramatically improves reaction selectivity and suppresses the formation of dimers, resulting in a single, well-defined tridendritic product. Experimental data within the patent demonstrates that this method can nearly double the yield for TM-1 synthesis to 41% and increase TM-2 yields from 46% to 66%. By eliminating the need for palladium and utilizing simpler fluoride salts, the process not only enhances chemical efficiency but also aligns perfectly with the goals of cost reduction in electronic chemical manufacturing.
This "inside-out" methodology dramatically improves reaction selectivity and suppresses the formation of dimers, resulting in a single, well-defined tridendritic product. Experimental data within the patent demonstrates that this method can nearly double the yield for TM-1 synthesis to 41% and increase TM-2 yields from 46% to 66%. By eliminating the need for palladium and utilizing simpler fluoride salts, the process not only enhances chemical efficiency but also aligns perfectly with the goals of cost reduction in electronic chemical manufacturing.
Mechanistic Insights into Fluoride-Activated Aryne Diels-Alder Cycloaddition
The core mechanistic breakthrough lies in the generation and trapping of the triphenylene aryne intermediate. The precursor molecule contains adjacent TMS and OTf groups on the aromatic rings. Upon exposure to a fluoride source like cesium fluoride (CsF), the silicon-fluorine bond formation drives the elimination of the triflate group, generating a highly strained and reactive benzyne-like species on the triphenylene scaffold. This transient aryne acts as a potent dienophile, rapidly undergoing a [4+2] Diels-Alder cycloaddition with externally added diene-like derivatives, such as tetraphenylcyclopentadienone. This mechanism bypasses the statistical randomness of trimerization, ensuring that every reactive event contributes to the growth of the desired three-armed structure. The reaction proceeds smoothly in mixed solvent systems of anhydrous acetonitrile and dichloromethane at temperatures ranging from 60°C to 150°C, providing a wide operational window for optimization.
From an impurity control perspective, this mechanism is superior because the rate of the intermolecular Diels-Alder reaction with the excess diene outcompetes potential side reactions of the aryne, such as dimerization or nucleophilic attack by solvent. The use of the triphenylene core as the limiting reagent ensures that the stoichiometry favors the formation of the fully substituted tridendritic product. 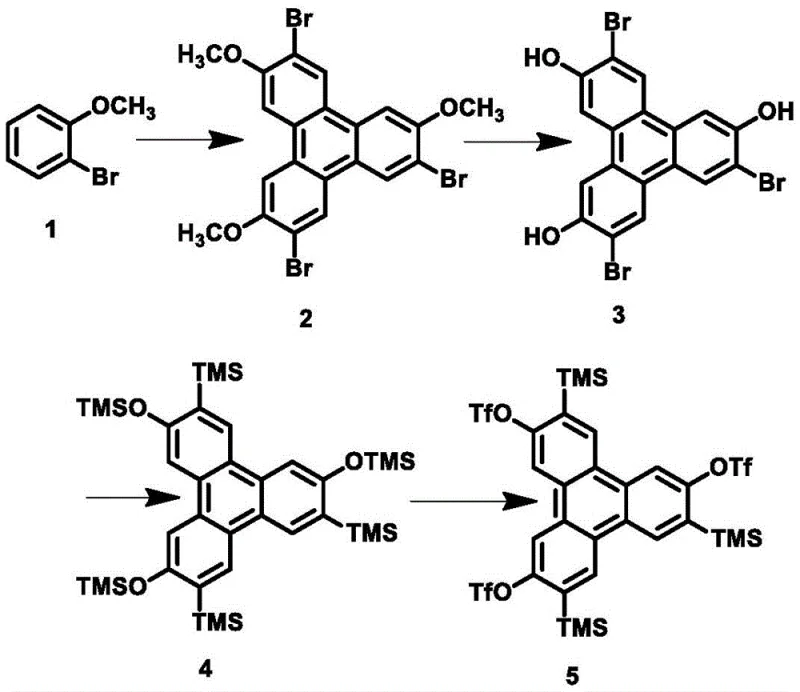 Furthermore, the absence of transition metals means there is no risk of metal-catalyzed homocoupling side reactions, which are common pitfalls in palladium-mediated cross-couplings. The result is a cleaner crude reaction mixture that requires less aggressive chromatography, preserving the integrity of the sensitive conjugated pi-systems essential for charge transport in organic semiconductors.
Furthermore, the absence of transition metals means there is no risk of metal-catalyzed homocoupling side reactions, which are common pitfalls in palladium-mediated cross-couplings. The result is a cleaner crude reaction mixture that requires less aggressive chromatography, preserving the integrity of the sensitive conjugated pi-systems essential for charge transport in organic semiconductors.
How to Synthesize Triphenylene-Centered Core Derivatives Efficiently
The synthesis of these advanced materials begins with the preparation of the key precursor, 3,7,10-tris(trimethylsilyl)-2,6,11-tris(trifluoromethanesulfonate)triphenylene, via a four-step sequence involving trimerization of o-bromoanisole, demethylation, silyl protection, and final triflation. Once this versatile building block is secured, the final assembly involves reacting it with the chosen diene component in the presence of a fluoride activator. The detailed standardized synthesis steps for producing high-purity batches are outlined below.
- Synthesize the core precursor 3,7,10-tris(trimethylsilyl)-2,6,11-tris(trifluoromethanesulfonate)triphenylene through trimerization, demethylation, protection, and triflation.
- Prepare the diene-like derivative reactant, such as substituted cyclopentadienones or other conjugated dienes suitable for aryne trapping.
- React the precursor with the diene derivative and a fluoride catalyst (e.g., CsF) in anhydrous acetonitrile/dichloromethane at elevated temperatures (60-150°C) to form the final tridendritic compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the shift to this palladium-free, inside-out synthesis model offers compelling economic and logistical benefits. The elimination of expensive palladium catalysts directly translates to a reduction in raw material costs, while the simplified workup procedures reduce solvent consumption and waste disposal fees. The higher yields observed in the patent data suggest a more efficient utilization of starting materials, which is critical when scaling up production of high-value electronic chemicals. Additionally, the robustness of the reaction conditions allows for greater flexibility in manufacturing scheduling and reduces the risk of batch failures due to sensitive catalyst deactivation.
- Cost Reduction in Manufacturing: The removal of palladium catalysts is a major driver for cost optimization. Palladium complexes are not only expensive to purchase but also require specialized recovery processes or extensive purification to meet ppm-level metal residue limits in electronic materials. By switching to inexpensive cesium fluoride, the process significantly lowers the bill of materials. Furthermore, the improved selectivity reduces the burden on downstream purification, lowering the overall cost of goods sold (COGS) through reduced chromatography media and solvent usage.
- Enhanced Supply Chain Reliability: Relying on precious metal catalysts often exposes supply chains to geopolitical volatility and price fluctuations. This new method utilizes commodity chemicals like o-bromoanisole and standard silylating agents, which are widely available from multiple global suppliers. This diversification of the supply base mitigates the risk of production stoppages and ensures a steady flow of intermediates. The milder reaction conditions also reduce equipment wear and tear, contributing to longer asset life and consistent production uptime.
- Scalability and Environmental Compliance: The "inside-out" approach is inherently more scalable because it avoids the exothermic risks and mixing issues associated with trimerization reactions that must bring three molecules together simultaneously. The stepwise addition of branches to a core is easier to control in large reactors. Moreover, the reduction in heavy metal waste aligns with increasingly strict environmental regulations regarding hazardous waste discharge, facilitating smoother regulatory approvals for new manufacturing lines and supporting sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this triphenylene synthesis technology. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation to ensure accuracy for potential partners.
Q: What is the primary advantage of the inside-out synthesis method for triphenylene derivatives?
A: The inside-out method utilizes a pre-formed triphenylene core with reactive handles, allowing for higher selectivity, fewer by-products like dimers, and significantly improved yields compared to traditional cyclotrimerization of single-branched precursors.
Q: Does this synthesis method require expensive palladium catalysts?
A: No, the novel method described in patent CN110105160B eliminates the need for palladium catalysts, utilizing cesium fluoride instead, which drastically reduces raw material costs and simplifies downstream purification by removing heavy metal residues.
Q: What types of derivatives can be synthesized using this triphenylene alkyne precursor?
A: This precursor is highly versatile and can react with various diene-like derivatives to produce a wide range of tridendritic compounds, including those with pyrene, phenanthrene, or perylene arms, making it ideal for tuning properties in organic photoelectric applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Triphenylene Derivative Supplier
The technological potential of triphenylene-based architectures for organic light-emitting diodes (OLEDs) and organic photovoltaics (OPVs) is immense, yet realizing this potential requires a manufacturing partner with deep chemical expertise. NINGBO INNO PHARMCHEM stands ready to support your development pipeline with our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facility is equipped with rigorous QC labs capable of verifying stringent purity specifications, ensuring that every batch of triphenylene precursor or derivative meets the exacting standards required for high-performance electronic devices.
We invite you to collaborate with us to evaluate the feasibility of integrating this novel inside-out synthesis into your supply chain. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how we can deliver high-purity OLED materials with superior consistency and value.
