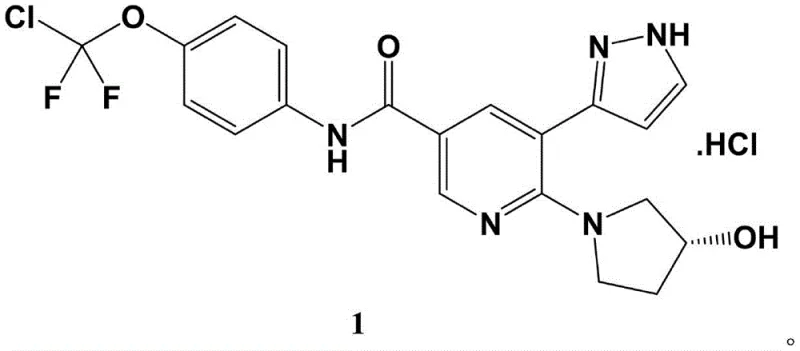
Scalable Preparation of Asciminib Hydrochloride: A Safer Route for Commercial API Production
The pharmaceutical industry is constantly seeking robust, scalable, and safe manufacturing processes for critical oncology therapies. Patent CN114369085B discloses a novel preparation method for Asciminib hydrochloride, a potent allosteric inhibitor used in the treatment of Chronic Myelogenous Leukemia (CML). This technical disclosure represents a significant advancement over prior art by utilizing 2-chloro-3-acetylpyridine as a cost-effective starting material and employing a sequence of conventional yet highly optimized reactions. The route successfully navigates complex chemical transformations, including ketoester condensation, N-alkylation, and palladium-catalyzed carbonylation, to deliver the target API intermediate with high purity. By prioritizing recrystallization over chromatographic purification, this method addresses key bottlenecks in industrial scale-up, offering a compelling value proposition for reliable Asciminib intermediate suppliers aiming to optimize their production pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Asciminib and its precursors has relied on pathways that introduce significant operational hazards and inefficiencies. Prior art, such as the routes filed by Novartis, often necessitates the use of highly corrosive reagents like thionyl chloride or oxalyl chloride during amidation steps. These reagents decompose in moisture to release toxic gases such as sulfur dioxide and hydrogen chloride, posing severe risks to reaction equipment integrity and personnel safety. Furthermore, the de-protection steps in traditional routes frequently employ trifluoroacetic acid, a substance known for its strong corrosivity and potential to cause chemical pneumonia upon inhalation. Beyond safety concerns, these conventional methods heavily depend on column chromatography for purification, a technique that is notoriously time-consuming, solvent-intensive, and difficult to translate from laboratory benchtop to multi-ton commercial manufacturing environments.
The Novel Approach
In stark contrast, the methodology outlined in CN114369085B introduces a streamlined synthetic strategy that mitigates these risks while enhancing process efficiency. By selecting 2-chloro-3-acetylpyridine as the foundational building block, the inventors have established a route that avoids the need for hazardous chlorinating agents and strong acidic de-protection conditions. The entire process is designed around reaction types that are conventional and common, ensuring that no special or exotic reaction conditions are required. Crucially, the purification strategy shifts entirely to recrystallization modes. This transition not only simplifies the post-treatment workflow but also drastically reduces the consumption of organic solvents and the generation of hazardous waste. For procurement managers and supply chain heads, this translates to a more predictable production schedule and reduced dependency on complex waste treatment infrastructure, making it an ideal candidate for cost reduction in API manufacturing.
Mechanistic Insights into the Five-Step Synthetic Cascade
The core of this innovation lies in a meticulously orchestrated five-step reaction sequence that builds the complex molecular architecture of Asciminib hydrochloride. The process initiates with a ketoester condensation where 2-chloro-3-acetylpyridine reacts with ethyl formate under basic conditions to form a beta-keto aldehyde intermediate. This is followed by a stereoselective N-alkylation with (S)-3-hydroxypyrrolidine, facilitated by a phase transfer catalyst and inorganic base, which installs the chiral pyrrolidine ring essential for biological activity. Subsequent chlorination using N-chlorosuccinimide (NCS) prepares the pyridine ring for the pivotal carbonylation step. Here, a palladium-catalyzed reaction under carbon monoxide atmosphere couples the chlorinated intermediate with a protected aniline derivative, forming the critical amide bond. The sequence concludes with a Knorr pyrazole synthesis using hydrazine hydrate, which cyclizes the side chain to form the final pyrazole ring, followed by salt formation with hydrochloric acid.
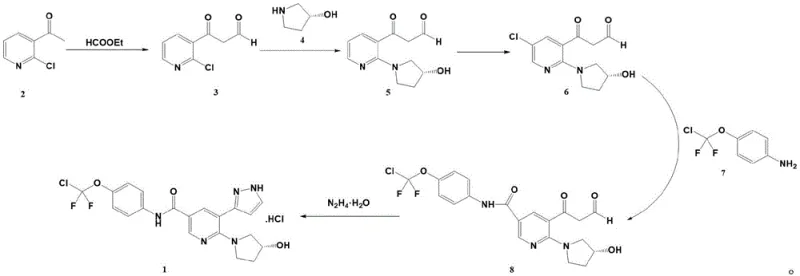
From an impurity control perspective, this route offers distinct advantages due to its reliance on crystallization-driven purification. In each step, from the initial condensation to the final pyrazole formation, the reaction conditions are tuned to favor the precipitation of the desired product while leaving soluble impurities in the mother liquor. For instance, the N-alkylation step utilizes potassium carbonate and tetrabutylammonium bromide in acetonitrile, conditions that promote high conversion rates (reported yields up to 90.6%) while minimizing side reactions. The avoidance of column chromatography means that the impurity profile is managed through solubility differences rather than adsorption dynamics, leading to a more consistent and reproducible quality profile. This mechanistic robustness ensures that the final Asciminib hydrochloride achieves high HPLC purity, meeting the stringent specifications required for pharmaceutical applications without the need for resource-intensive polishing steps.
How to Synthesize Asciminib Hydrochloride Efficiently
The synthesis of this complex oncology intermediate requires precise control over reaction parameters to maximize yield and safety. The patented procedure details specific temperature ranges, molar ratios, and solvent systems optimized for each transformation. For example, the initial condensation is best performed by slowly adding ethyl formate to a cooled mixture of the starting material and sodium hydride in THF, followed by heating to reflux. Similarly, the carbonylation step demands strict nitrogen protection and controlled introduction of carbon monoxide gas to ensure efficient coupling. While the general workflow is straightforward, adhering to the specific stoichiometric ratios and crystallization protocols described in the patent is critical for success. The detailed standardized synthesis steps see the guide below for a comprehensive breakdown of the operational parameters.
- Perform ketoester condensation of 2-chloro-3-acetylpyridine with ethyl formate using a strong base to obtain the beta-keto aldehyde intermediate.
- Execute N-alkylation with (S)-3-hydroxypyrrolidine under phase transfer catalysis to install the pyrrolidine moiety.
- Conduct selective chlorination using N-chlorosuccinimide (NCS) followed by palladium-catalyzed carbonylative amidation.
- Finalize the synthesis via Knorr pyrazole cyclization with hydrazine hydrate and subsequent salt formation with hydrochloric acid.
Commercial Advantages for Procurement and Supply Chain Teams
For stakeholders focused on the bottom line and supply continuity, this synthetic route offers transformative benefits over legacy methods. The elimination of hazardous reagents like thionyl chloride removes the need for specialized corrosion-resistant reactors and extensive gas scrubbing systems, directly lowering capital expenditure and maintenance costs. Furthermore, the shift from chromatography to recrystallization significantly reduces solvent consumption and waste disposal fees, contributing to substantial cost savings in pharmaceutical intermediates manufacturing. The use of cheap, commercially available starting materials ensures that the supply chain remains resilient against raw material shortages, providing a stable foundation for long-term production planning.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the removal of expensive purification technologies. By replacing column chromatography with recrystallization, manufacturers can drastically reduce the volume of solvents required and the associated energy costs for solvent recovery. Additionally, the avoidance of precious metal scavengers or complex workups associated with corrosive reagents further streamlines the cost structure. This efficiency allows for a more competitive pricing model for high-purity Asciminib intermediates, enabling downstream API producers to optimize their own margins without compromising on quality standards.
- Enhanced Supply Chain Reliability: Supply chain stability is bolstered by the use of commodity chemicals as starting materials. 2-chloro-3-acetylpyridine and other key reagents are widely available in the global market, reducing the risk of bottlenecks associated with custom-synthesized building blocks. The robustness of the reaction conditions, which do not require cryogenic temperatures or ultra-high pressures, means that production can be easily distributed across multiple manufacturing sites if necessary. This flexibility ensures consistent delivery schedules and reduces lead time for high-purity pharmaceutical intermediates, a critical factor for drug developers managing tight clinical trial timelines.
- Scalability and Environmental Compliance: The environmental footprint of this process is significantly smaller than that of traditional routes. The absence of toxic gas emissions and the reduction in hazardous waste generation align perfectly with modern green chemistry principles and regulatory requirements. This compliance simplifies the permitting process for new manufacturing facilities and reduces the liability associated with environmental incidents. Moreover, the scalability of recrystallization processes is well-understood in the chemical industry, allowing for seamless transition from pilot plant batches to multi-ton commercial production without the need for re-engineering purification workflows.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity on safety, purity, and operational feasibility. Understanding these aspects is crucial for technical teams evaluating the adoption of this methodology for their own production lines.
Q: How does this synthesis route improve safety compared to conventional methods?
A: This method eliminates the use of highly corrosive reagents like thionyl chloride and oxalyl chloride, as well as strong acids like trifluoroacetic acid, significantly reducing equipment corrosion risks and toxic gas emissions.
Q: What purification strategy is employed to ensure high purity without chromatography?
A: The process relies exclusively on recrystallization for purification at each step, avoiding time-consuming and solvent-intensive column chromatography, which facilitates industrial mass production.
Q: Is the starting material commercially viable for large-scale production?
A: Yes, the route utilizes 2-chloro-3-acetylpyridine as the starting material, which is a cheap, readily available, and commonly used reagent, ensuring a stable and cost-effective supply chain.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Asciminib Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and safe manufacturing pathways for life-saving oncology medications. Our team of expert chemists has extensively analyzed the route disclosed in CN114369085B and possesses the technical capability to implement this advanced synthesis at scale. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab to plant is smooth and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of Asciminib intermediate meets the highest international standards for pharmaceutical use.
We invite pharmaceutical partners to collaborate with us to leverage this superior synthetic route for their supply chains. By optimizing the process parameters and leveraging our bulk purchasing power for raw materials, we can offer a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments, allowing you to make informed decisions that enhance both the quality and economics of your API production.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
