Advanced Synthesis Of 3-Amino-4-Fluorophenylboronic Acid For Commercial Pharmaceutical Manufacturing
Advanced Synthesis Of 3-Amino-4-Fluorophenylboronic Acid For Commercial Pharmaceutical Manufacturing
The landscape of pharmaceutical intermediate manufacturing is constantly evolving, driven by the need for more efficient, cost-effective, and scalable synthetic routes for complex building blocks. A pivotal development in this domain is detailed in patent CN103626791A, which discloses a robust method for synthesizing 3-amino-4-fluorophenylboronic acid. This compound serves as a critical precursor in Suzuki-Miyaura cross-coupling reactions, widely utilized in the construction of biaryl scaffolds found in numerous active pharmaceutical ingredients (APIs). The disclosed methodology represents a significant departure from traditional pathways, addressing long-standing issues regarding raw material availability and process complexity. By leveraging a strategic sequence of Grignard borylation, controlled nitration, and catalytic hydrogenation, this technology offers a viable solution for producing high-purity intermediates essential for modern drug discovery and development pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to the innovations described in the patent data, the synthesis of structurally similar compounds often relied on the use of 3-amino-4-fluorophenylboronic acid pinacol esters as key intermediates. The conventional route typically involves the coupling of 2-fluoro-5-bromoaniline with pinacol diboron under palladium catalysis, followed by a subsequent deprotection step to yield the free boronic acid. This approach presents several inherent disadvantages that hinder large-scale commercial viability. Firstly, the starting material, 2-fluoro-5-bromoaniline, is notoriously expensive and difficult to source in bulk quantities, creating a bottleneck for supply chain continuity. Secondly, the requirement for a pinacol protecting group introduces an additional synthetic step—the hydrolysis of the ester—which not only increases processing time but also generates additional chemical waste. The cumulative effect of these factors results in a higher cost of goods sold (COGS) and a more complex operational footprint, making the conventional method less attractive for cost-sensitive pharmaceutical manufacturing.
The Novel Approach
In stark contrast, the novel approach outlined in the patent utilizes p-fluorobromobenzene as the primary starting material, a commodity chemical that is readily available and economically superior. The strategy bypasses the need for expensive aniline derivatives and protective group chemistry entirely. Instead, it employs a direct borylation via a Grignard intermediate, followed by a direct nitration and reduction sequence. This streamlined pathway significantly reduces the number of unit operations and eliminates the costly pinacol ligand. The simplicity of the operation allows for easier scale-up and reduces the environmental burden associated with waste disposal. By focusing on direct functionalization of the aromatic ring, this method achieves the desired substitution pattern with high efficiency, providing a suitable and economically favorable path for preparing 3-amino-4-fluorophenylboronic acid.
Mechanistic Insights into Grignard Borylation and Controlled Nitration
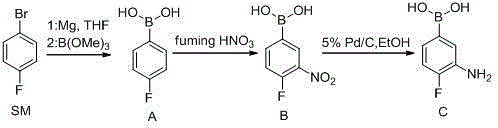
The core of this synthesis lies in the precise execution of the Grignard borylation step, which sets the foundation for the entire sequence. The process initiates with the formation of the Grignard reagent from p-fluorobromobenzene and magnesium turnings in tetrahydrofuran (THF). Critical to this step is the thermal management; the reaction is initiated at 50°C and maintained to ensure complete consumption of the magnesium, followed by cooling to -30°C before the addition of trimethyl borate. This low-temperature addition is vital to control the exothermic nature of the reaction between the highly reactive Grignard species and the borate ester, preventing side reactions such as homocoupling or decomposition. Following the reaction period, an acidic quench with hydrochloric acid hydrolyzes the borate ester to the free boronic acid, which is then isolated through extraction and slurry purification. This mechanistic precision ensures that Intermediate A (p-fluorophenylboronic acid) is obtained with a content of ≥97%, providing a clean substrate for the subsequent nitration.
The subsequent nitration and reduction steps demonstrate sophisticated control over chemoselectivity and functional group tolerance. In the nitration phase, Intermediate A is reacted with fuming nitric acid at strictly controlled temperatures ranging from -20°C to -55°C. This extreme cold is necessary because the boronic acid group is sensitive to oxidative conditions, and higher temperatures could lead to protodeboronation or over-nitration. The electrophilic aromatic substitution occurs selectively at the meta-position relative to the boronic acid group (ortho to the fluorine), yielding Intermediate B. Finally, the reduction of the nitro group to the amine is achieved via catalytic hydrogenation using 5% Pd/C in ethanol or methanol at 55°C. This heterogeneous catalysis is highly efficient, converting the nitro functionality without affecting the boronic acid moiety or the fluorine substituent, ultimately delivering the target molecule with an HPLC purity of 99.1% after recrystallization.
How to Synthesize 3-Amino-4-Fluorophenylboronic Acid Efficiently
Implementing this synthesis route requires adherence to specific operational parameters to maximize yield and safety, particularly regarding the handling of pyrophoric Grignard reagents and strong oxidizers like fuming nitric acid. The patent provides a clear framework for executing these transformations, emphasizing the importance of stoichiometry and temperature control at each stage. For research and development teams looking to replicate or scale this process, understanding the nuances of the workup procedures—such as the specific pH adjustments and solvent slurries—is key to isolating high-quality intermediates. The following guide outlines the standardized procedural flow derived from the patent examples, serving as a foundational reference for process optimization.
- Prepare p-fluorophenylboronic acid (Intermediate A) by reacting p-fluorobromobenzene with magnesium to form a Grignard reagent, followed by reaction with trimethyl borate and acidic hydrolysis.
- Perform nitration on Intermediate A using fuming nitric acid at controlled low temperatures (-20°C to -55°C) to generate 3-nitro-4-fluorophenylboronic acid (Intermediate B).
- Conduct catalytic hydrogenation of Intermediate B using Pd/C in methanol at 55°C to reduce the nitro group, yielding the final 3-amino-4-fluorophenylboronic acid product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this novel synthesis route offers tangible strategic benefits that extend beyond mere technical feasibility. The primary advantage lies in the fundamental shift of the raw material base. By replacing scarce and expensive aniline derivatives with abundant p-fluorobromobenzene, the manufacturing process becomes inherently more resilient to market fluctuations. This change mitigates the risk of supply disruptions that often plague specialized fine chemical sectors. Furthermore, the elimination of the pinacol ester protection and deprotection cycle translates directly into a reduction in processing time and solvent consumption. Fewer steps mean fewer opportunities for yield loss and lower overall energy consumption per kilogram of product, contributing to a leaner and more sustainable manufacturing profile that aligns with modern green chemistry initiatives.
- Cost Reduction in Manufacturing: The economic impact of this route is driven by the substantial difference in raw material costs. P-fluorobromobenzene is a bulk commodity chemical, whereas 2-fluoro-5-bromoaniline is a specialized, high-cost intermediate. Additionally, removing the pinacol diboron reagent and the associated hydrolysis step eliminates significant material costs and waste treatment expenses. The simplified workflow reduces labor hours and equipment occupancy time, leading to a drastic improvement in overall production efficiency and a lower cost basis for the final API intermediate.
- Enhanced Supply Chain Reliability: Sourcing reliability is a critical metric for any long-term supply agreement. The reliance on easily obtainable starting materials ensures that production schedules are not held hostage by the lead times of exotic reagents. The robustness of the Grignard and hydrogenation steps, which are well-understood unit operations in the fine chemical industry, further guarantees consistent output. This stability allows for better inventory planning and reduces the need for excessive safety stock, optimizing working capital for both the supplier and the client.
- Scalability and Environmental Compliance: From a scale-up perspective, the process utilizes standard reactors and filtration equipment, avoiding the need for specialized high-pressure or cryogenic infrastructure beyond standard industrial capabilities. The waste stream is simplified by the absence of pinacol byproducts, making effluent treatment more straightforward and cost-effective. This environmental compatibility facilitates smoother regulatory approvals and supports the growing demand for eco-friendly pharmaceutical manufacturing processes, ensuring long-term operational continuity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of 3-amino-4-fluorophenylboronic acid. These insights are derived directly from the patented methodology and are intended to clarify the operational advantages and quality standards associated with this specific synthetic route. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this intermediate into their broader drug substance manufacturing strategies.
Q: Why is the Grignard borylation route preferred over the pinacol ester route for this compound?
A: The Grignard route utilizes p-fluorobromobenzene, which is significantly more accessible and cost-effective than the expensive 2-fluoro-5-bromoaniline required for the pinacol ester pathway. Furthermore, this direct method eliminates the additional hydrolysis step needed to convert pinacol esters to free boronic acids, thereby simplifying the process and reducing waste.
Q: What are the critical control parameters during the nitration step?
A: Temperature control is paramount during nitration. The reaction must be maintained between -20°C and -55°C using fuming nitric acid to ensure regioselectivity and prevent the decomposition of the sensitive boronic acid moiety. Deviation from this range can lead to impurities or reduced yields.
Q: What purity levels can be achieved with this synthesis method?
A: According to the patent data, the final product obtained after recrystallization and vacuum drying achieves an HPLC purity of 99.1%. The process includes rigorous purification steps such as solvent slurring and recrystallization to ensure the removal of catalyst residues and organic impurities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Amino-4-Fluorophenylboronic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of new therapeutic agents depends on the reliability and quality of the supply chain. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with precision and consistency. Our facilities are equipped with stringent purity specifications and rigorous QC labs capable of verifying the structural integrity and purity of complex intermediates like 3-amino-4-fluorophenylboronic acid. We are committed to delivering products that meet the highest international standards, supporting your R&D and commercial goals with unwavering dependability.
We invite you to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic advantages of switching to this more efficient manufacturing method. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that drive value and efficiency in your pharmaceutical development pipeline.
