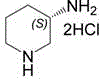
Advanced Aqueous Resolution Strategy for High-Purity S-3-Aminopiperidine Dihydrochloride Production
Advanced Aqueous Resolution Strategy for High-Purity S-3-Aminopiperidine Dihydrochloride Production
The pharmaceutical industry continuously demands more efficient and environmentally benign synthetic routes for chiral intermediates, and patent CN114591219A represents a significant breakthrough in the preparation of S-3-aminopiperidine dihydrochloride. This specific chemical entity serves as a critical building block for various high-value active pharmaceutical ingredients, yet its production has historically been plagued by low yields and excessive solvent usage. The disclosed methodology introduces a novel aqueous-based resolution process that leverages the differential solubility of diastereomeric salts to achieve exceptional stereochemical control. By shifting the reaction medium from traditional organic solvents to water, this technology not only aligns with green chemistry principles but also simplifies the downstream processing requirements significantly. For R&D directors and process chemists, this patent offers a robust pathway to access high-purity materials without the burden of complex chromatographic separations or hazardous waste streams.
Furthermore, the strategic implementation of this synthesis route addresses the growing regulatory pressure on pharmaceutical manufacturers to minimize their carbon footprint and reduce the use of Class 2 and Class 3 solvents. The ability to produce S-3-aminopiperidine dihydrochloride with an enantiomeric excess exceeding 99.9% using such a straightforward protocol demonstrates the maturity and scalability of the underlying chemistry. As a reliable pharmaceutical intermediate supplier, understanding these nuanced improvements in synthetic design is crucial for maintaining a competitive edge in the global market. The following analysis delves deep into the mechanistic advantages and commercial implications of adopting this water-mediated resolution strategy for large-scale manufacturing operations.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the separation of racemic 3-aminopiperidine into its individual enantiomers has relied heavily on organic solvent systems, such as methanol, which present significant operational and economic drawbacks. Prior art, including methods described in patents like US2013172562, often suffers from suboptimal resolution yields and fails to incorporate effective strategies for the recovery of expensive chiral resolving agents. When methanol is used as the primary solvent, the process generates substantial volumes of flammable and toxic waste that require specialized incineration or treatment facilities, thereby inflating the overall cost of goods sold. Moreover, the inability to efficiently recycle the resolving agent means that each batch consumes a fresh quantity of high-cost chiral acids, creating a linear cost structure that does not benefit from economies of scale. These inefficiencies create bottlenecks in supply chains, particularly when demand for the S-enantiomer surges, as the production capacity is limited by the availability and cost of the resolving agent.
The Novel Approach
In stark contrast, the methodology outlined in CN114591219A revolutionizes the production landscape by utilizing water as the exclusive reaction and crystallization medium for the initial resolution step. This fundamental shift eliminates the fire hazards associated with large volumes of organic solvents and drastically reduces the environmental impact of the manufacturing process. The novel approach capitalizes on the distinct solubility profiles of the diastereomeric salts formed between racemic 3-aminopiperidine and D-dibenzoyl tartaric acid in an aqueous environment. By carefully controlling temperature and concentration parameters, the process selectively precipitates the desired S-configured salt while keeping the unwanted R-isomer in solution, achieving high optical purity in a single operation. Furthermore, the design of this process inherently facilitates the recovery of the resolving agent in a later stage, transforming what was once a consumable expense into a reusable asset, which is a game-changer for cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Aqueous Diastereomeric Salt Resolution
The core of this innovative synthesis lies in the precise manipulation of acid-base interactions and solubility equilibria within an aqueous system. When racemic 3-aminopiperidine is introduced to D-dibenzoyl tartaric acid (D-DBTA) in water, two distinct diastereomeric salts are formed: the S-aminopiperidine-D-DBTA salt and the R-aminopiperidine-D-DBTA salt. Due to the specific stereochemical arrangement of the S-enantiomer paired with the D-tartaric acid derivative, this particular salt exhibits significantly lower solubility in water compared to its R-counterpart. This differential solubility is the driving force behind the separation, allowing the S-salt to crystallize out of the solution preferentially while the R-salt remains dissolved in the mother liquor. The process is further refined through a recrystallization step, where the crude wet salt is redissolved in water and slowly cooled, effectively purging any remaining R-isomer impurities and elevating the enantiomeric excess to near-perfect levels.
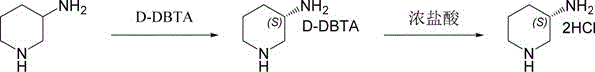
Following the isolation of the high-purity S-DBTA salt, the final transformation involves an acidification step using concentrated hydrochloric acid. This step serves a dual purpose: it protonates the amine to form the stable dihydrochloride salt of the target product and simultaneously regenerates the free D-dibenzoyl tartaric acid. Because the regenerated resolving agent is insoluble under these specific acidic conditions, it precipitates as a solid filter cake that can be easily separated from the product-containing filtrate. This mechanistic elegance ensures that the chiral information is preserved throughout the cycle and that the resolving agent is recovered in a form suitable for direct reuse in subsequent batches. For technical teams, understanding this cycle is vital for optimizing mass balance and ensuring consistent product quality across multiple production runs without the accumulation of deleterious by-products.
How to Synthesize S-3-Aminopiperidine Dihydrochloride Efficiently
The practical execution of this synthesis requires careful attention to temperature control and stoichiometric ratios to maximize both yield and optical purity. The process begins with the dissolution of the racemic amine in water, followed by the controlled addition of the resolving agent at elevated temperatures to ensure complete salt formation before inducing crystallization. Detailed standard operating procedures regarding cooling rates, stirring speeds, and filtration techniques are essential to replicate the high success rates reported in the patent examples. Operators must adhere strictly to the specified weight ratios of water to substrate, as deviations can alter the solubility equilibrium and compromise the resolution efficiency. The following guide outlines the critical operational phases necessary to achieve the reported benchmarks of purity and yield in a commercial setting.
- Dissolve racemic 3-aminopiperidine in water and react with D-dibenzoyl tartaric acid (D-DBTA) at 35-55°C to form the diastereomeric salt precipitate.
- Recrystallize the wet S-DBTA salt intermediate in water to enhance chiral purity, achieving an ee value exceeding 99%.
- Treat the purified intermediate with concentrated hydrochloric acid to release the final S-3-aminopiperidine dihydrochloride product and recover the D-DBTA resolving agent.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this aqueous resolution technology translates into tangible strategic benefits that extend far beyond simple chemical yield improvements. The most significant advantage is the drastic reduction in raw material costs achieved through the closed-loop recovery of the D-dibenzoyl tartaric acid resolving agent. In traditional processes, the resolving agent is often lost or degraded, representing a major recurring expense; however, this method allows for its quantitative recovery and reuse, effectively amortizing the cost of this high-value chiral auxiliary over hundreds of production cycles. Additionally, the elimination of bulk organic solvents like methanol reduces the logistical burden associated with the storage, handling, and disposal of hazardous chemicals, leading to lower insurance premiums and reduced regulatory compliance costs. These factors combine to create a more resilient and cost-efficient supply chain capable of withstanding market volatility in raw material pricing.
- Cost Reduction in Manufacturing: The ability to recycle the chiral resolving agent fundamentally alters the cost structure of producing S-3-aminopiperidine dihydrochloride. By recovering the D-DBTA from the acidification filtrate, manufacturers avoid the continuous purchase of expensive chiral acids, resulting in substantial long-term savings. Furthermore, the use of water as a solvent eliminates the need for costly solvent recovery distillation units and reduces energy consumption associated with heating and cooling volatile organic compounds. This streamlined approach minimizes waste treatment fees and lowers the overall cost of goods sold, making the final product more competitive in price-sensitive markets.
- Enhanced Supply Chain Reliability: Relying on water as the primary reaction medium significantly de-risks the supply chain by removing dependence on petrochemical-derived solvents whose prices and availability can fluctuate wildly. Water is universally available, inexpensive, and non-flammable, ensuring that production can continue uninterrupted even during periods of solvent shortages or transport disruptions. The robustness of the aqueous process also simplifies facility requirements, allowing for production in a wider range of manufacturing sites without the need for explosion-proof infrastructure, thereby increasing overall production capacity and flexibility to meet urgent customer demands.
- Scalability and Environmental Compliance: The simplicity of the work-up procedure, which primarily involves filtration and crystallization rather than complex extractions or chromatography, makes this process highly scalable from pilot plant to multi-ton commercial production. The reduction in hazardous waste generation aligns perfectly with increasingly strict environmental regulations globally, facilitating easier permitting and community acceptance of manufacturing facilities. This environmental stewardship not only protects the company from potential fines but also enhances its brand reputation among eco-conscious pharmaceutical partners who prioritize sustainable sourcing in their vendor selection criteria.
Frequently Asked Questions (FAQ)
The technical nuances of this resolution process often raise specific questions regarding operational parameters and quality control metrics among potential partners. Understanding the specifics of solvent recovery, purity thresholds, and scalability is essential for integrating this technology into existing manufacturing portfolios. The answers provided below are derived directly from the experimental data and claims within the patent documentation, offering a transparent view of the process capabilities. These insights are designed to address the common concerns of technical buyers and quality assurance teams evaluating new supply sources for critical chiral intermediates.
Q: What is the primary advantage of using water as a solvent in this resolution process?
A: Using water eliminates the need for volatile organic solvents like methanol, significantly reducing environmental waste and safety hazards while lowering disposal costs associated with hazardous waste treatment.
Q: Can the chiral resolving agent be recovered and reused in this method?
A: Yes, the D-dibenzoyl tartaric acid (D-DBTA) resolving agent precipitates as a filter cake during the acidification step and can be dried and directly recycled for subsequent batches, drastically reducing raw material costs.
Q: What level of chiral purity can be achieved with this aqueous resolution technique?
A: The process utilizes a double crystallization strategy that consistently achieves an enantiomeric excess (ee) value greater than 99.9%, meeting the stringent quality requirements for advanced pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable S-3-Aminopiperidine Dihydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and robust manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising results seen in patent literature can be reliably reproduced on an industrial scale. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instruments to verify the enantiomeric excess and chemical purity of every batch. Our commitment to quality assurance means that clients receive materials that consistently meet the demanding standards required for the synthesis of next-generation pharmaceuticals and fine chemicals.
We invite you to engage with our technical procurement team to discuss how this advanced aqueous resolution technology can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits of switching to this greener, more efficient manufacturing route. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that optimize both your supply chain security and your bottom line. Let us collaborate to bring high-quality S-3-aminopiperidine dihydrochloride to your development pipeline with speed and precision.
