Advanced Solid-Base Catalysis for Commercial Dexamethasone Palmitate Production
The pharmaceutical industry is constantly seeking robust, scalable, and environmentally sustainable pathways for the production of high-value corticosteroids. A pivotal advancement in this domain is detailed in patent CN110041391B, which discloses a novel synthetic method for Dexamethasone Palmitate, a potent anti-inflammatory agent widely used in the treatment of rheumatoid arthritis and asthma. This patent introduces a transesterification protocol that replaces hazardous acyl chlorides and toxic solvents with a green solid-base catalytic system. For R&D Directors and Procurement Managers evaluating a reliable API supplier, this technology represents a significant leap forward in process safety and operational efficiency. By utilizing methyl palmitate and a heterogeneous solid alkali catalyst, the method achieves high yields under mild conditions, effectively addressing the critical pain points of waste management and operator safety associated with legacy synthesis routes. The implications for commercial scale-up of complex steroids are profound, offering a pathway to high-purity API production with a drastically reduced environmental footprint.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Dexamethasone Palmitate has relied heavily on the Schotten-Baumann reaction conditions, which involve the use of palmitoyl chloride as the acylating agent. As illustrated in the reaction scheme below, this traditional approach necessitates the use of pyridine as both a solvent and an acid scavenger, alongside chlorinated solvents like chloroform or tetrahydrofuran. 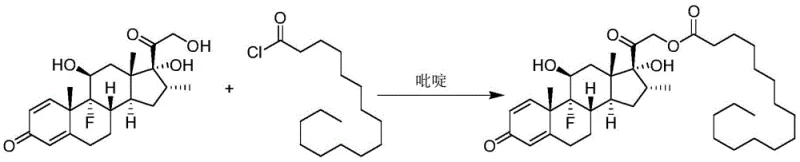 This legacy methodology presents severe drawbacks for modern pharmaceutical manufacturing. Pyridine is a known carcinogen with a noxious odor, creating significant occupational health hazards and requiring expensive abatement systems to protect the workforce. Furthermore, palmitoyl chloride is highly hygroscopic and unstable, complicating long-term storage and logistics for supply chain heads. The production of palmitoyl chloride itself generates substantial amounts of thionyl chloride waste and high-salinity wastewater, creating a heavy burden on environmental compliance teams. The purification process is equally cumbersome, often requiring multiple recrystallization steps to remove isomeric impurities formed due to the lack of regioselectivity in the harsh acyl chloride environment.
This legacy methodology presents severe drawbacks for modern pharmaceutical manufacturing. Pyridine is a known carcinogen with a noxious odor, creating significant occupational health hazards and requiring expensive abatement systems to protect the workforce. Furthermore, palmitoyl chloride is highly hygroscopic and unstable, complicating long-term storage and logistics for supply chain heads. The production of palmitoyl chloride itself generates substantial amounts of thionyl chloride waste and high-salinity wastewater, creating a heavy burden on environmental compliance teams. The purification process is equally cumbersome, often requiring multiple recrystallization steps to remove isomeric impurities formed due to the lack of regioselectivity in the harsh acyl chloride environment.
The Novel Approach
In stark contrast, the innovative method described in the patent utilizes a transesterification reaction between dexamethasone and methyl palmitate, catalyzed by a solid base and a phase transfer catalyst. 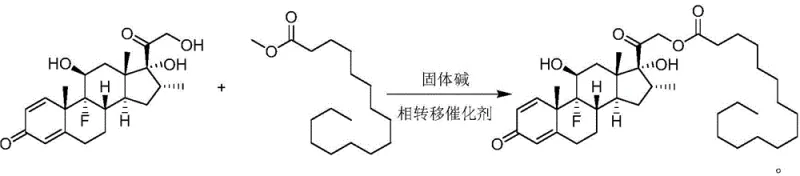 This approach fundamentally alters the reaction landscape by eliminating the need for corrosive acyl chlorides and toxic pyridine. The use of methyl palmitate, which acts as both a reactant and a solvent, simplifies the reaction matrix and allows for the recycling of unreacted starting materials, directly contributing to cost reduction in pharmaceutical manufacturing. The solid base catalyst, such as cesium oxide supported on gamma-alumina, provides a heterogeneous surface that facilitates easy separation via filtration. This not only streamlines the downstream processing but also enables the catalyst to be regenerated and reused, enhancing the overall sustainability of the process. The mild reaction conditions, typically ranging from 50°C to 80°C under vacuum, further reduce energy consumption and minimize the risk of thermal degradation of the sensitive steroid backbone.
This approach fundamentally alters the reaction landscape by eliminating the need for corrosive acyl chlorides and toxic pyridine. The use of methyl palmitate, which acts as both a reactant and a solvent, simplifies the reaction matrix and allows for the recycling of unreacted starting materials, directly contributing to cost reduction in pharmaceutical manufacturing. The solid base catalyst, such as cesium oxide supported on gamma-alumina, provides a heterogeneous surface that facilitates easy separation via filtration. This not only streamlines the downstream processing but also enables the catalyst to be regenerated and reused, enhancing the overall sustainability of the process. The mild reaction conditions, typically ranging from 50°C to 80°C under vacuum, further reduce energy consumption and minimize the risk of thermal degradation of the sensitive steroid backbone.
Mechanistic Insights into Solid-Base Catalyzed Transesterification
The core of this technological breakthrough lies in the synergistic action of the solid base and the phase transfer catalyst. The solid base, loaded on a porous carrier like gamma-alumina or molecular sieves, provides strong basic sites that activate the hydroxyl group of the dexamethasone molecule. Unlike homogeneous bases which can be difficult to separate and may cause side reactions, the heterogeneous nature of the solid base ensures that the catalytic activity is confined to the surface. The addition of a phase transfer catalyst, such as polyethylene glycol (PEG 400-800) or quaternary ammonium salts, plays a critical role in bridging the interface between the solid catalyst and the organic reactants. This facilitates the transport of reactive species to the active sites, significantly accelerating the reaction rate and improving the overall conversion efficiency. The vacuum applied during the reaction serves to continuously remove methanol, a byproduct of the transesterification, thereby driving the equilibrium towards the formation of Dexamethasone Palmitate in accordance with Le Chatelier's principle.
From a quality control perspective, the steric hindrance provided by the solid support is a game-changer for impurity control. Dexamethasone contains three hydroxyl groups, and traditional methods often struggle with non-selective esterification, leading to di- or tri-ester byproducts that are difficult to separate. The specific pore structure and surface properties of the solid base catalyst in this patent exhibit high regioselectivity, preferentially targeting the primary hydroxyl group at the C21 position. This selectivity is crucial for achieving the stringent purity specifications required for a high-purity API. By minimizing the formation of structural impurities at the source, the need for extensive downstream purification is reduced, resulting in a more robust and predictable manufacturing process that ensures consistent batch-to-batch quality for the final drug product.
How to Synthesize Dexamethasone Palmitate Efficiently
Implementing this synthesis route requires precise control over reaction parameters to maximize yield and catalyst life. The process begins with the preparation of the reactor under an inert nitrogen atmosphere to prevent oxidation. Methyl palmitate is introduced first, followed by the addition of dexamethasone, the solid base catalyst, and the phase transfer agent. The mixture is then heated to the optimal temperature range while maintaining a specific vacuum degree to facilitate methanol removal.
- Prepare the reaction system by loading a solid base catalyst, such as cesium oxide supported on gamma-alumina, into a reactor under nitrogen protection.
- Add dexamethasone and methyl palmitate along with a phase transfer catalyst like polyethylene glycol, then heat to 50-80°C under vacuum.
- Filter the solid catalyst while hot, cool the filtrate to crystallize the product, and recycle the mother liquor for methyl palmitate recovery.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this solid-base catalyzed process offers tangible strategic advantages beyond mere technical feasibility. The elimination of pyridine and palmitoyl chloride removes two of the most problematic raw materials from the supply chain, reducing dependency on hazardous chemical suppliers and simplifying regulatory compliance regarding transportation and storage. The ability to recycle the solid catalyst and the methyl palmitate solvent creates a closed-loop system that significantly lowers the consumption of raw materials per kilogram of product. This efficiency translates directly into substantial cost savings over the lifecycle of the product, making it a highly attractive option for long-term commercial contracts. Furthermore, the simplified work-up procedure, which involves only filtration and crystallization, reduces the operational time and labor costs associated with production.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven by the dual functionality of methyl palmitate as both reagent and solvent, which allows for the recovery and reuse of unreacted material, thereby minimizing raw material waste. Additionally, the heterogeneous catalyst can be filtered and reused multiple times without significant loss of activity, eliminating the recurring cost of purchasing stoichiometric amounts of homogeneous bases or acid scavengers. The removal of expensive and hazardous waste treatment protocols associated with pyridine and chlorinated solvents further contributes to a leaner cost structure, ensuring competitive pricing for the final API without compromising on quality standards.
- Enhanced Supply Chain Reliability: By replacing hygroscopic and unstable palmitoyl chloride with stable methyl palmitate, the risk of raw material degradation during storage and transit is virtually eliminated. This stability ensures a consistent supply of high-quality inputs, reducing the likelihood of production delays caused by substandard raw materials. The robustness of the solid catalyst system also means that the manufacturing process is less sensitive to minor fluctuations in operating conditions, providing a more reliable production schedule. For supply chain heads, this translates to reduced lead time for high-purity steroids and a more predictable inventory management strategy, crucial for meeting the demands of the global pharmaceutical market.
- Scalability and Environmental Compliance: The green chemistry principles embedded in this method align perfectly with increasingly stringent global environmental regulations. The absence of carcinogenic solvents and the reduction of high-salt wastewater simplify the permitting process for new manufacturing facilities and reduce the liability associated with environmental discharge. The process is inherently scalable, as the heterogeneous catalysis system is well-suited for large-scale reactor operations without the mixing and heat transfer limitations often seen in viscous homogeneous systems. This scalability ensures that the technology can grow with market demand, supporting the commercial scale-up of complex steroids from pilot plant to multi-ton production without the need for fundamental process redesign.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent literature, providing a clear understanding of the process capabilities.
Q: How does the solid-base method improve purity compared to traditional pyridine methods?
A: The solid-base catalyst possesses significant steric hindrance, which selectively targets the primary hydroxyl group at the C21 position while preventing esterification at other hydroxyl sites. This high selectivity minimizes byproduct formation, eliminating the need for multiple recrystallizations required in conventional methods to remove isomeric impurities.
Q: What are the supply chain advantages of using methyl palmitate over palmitoyl chloride?
A: Methyl palmitate is significantly more stable and less hygroscopic than palmitoyl chloride, simplifying storage and logistics. Furthermore, it serves a dual role as both a reactant and a solvent, allowing for efficient recycling of unreacted material, which drastically reduces raw material consumption and waste generation.
Q: Is the solid catalyst reusable for industrial scale-up?
A: Yes, experimental data indicates that the solid base catalyst can be recovered via simple filtration and reused for multiple cycles with only marginal decreases in yield. This heterogeneity allows for easy separation from the product stream, supporting continuous or semi-continuous manufacturing processes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dexamethasone Palmitate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic routes to meet the evolving needs of the global pharmaceutical industry. Our technical team has extensively evaluated the solid-base catalyzed transesterification pathway and possesses the expertise to implement this technology at an industrial scale. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab to plant is seamless and efficient. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that verify every batch against the highest international standards, guaranteeing that the Dexamethasone Palmitate we supply meets the exacting requirements of your formulation teams.
We invite you to collaborate with us to optimize your supply chain for this critical anti-inflammatory agent. By leveraging our technical capabilities, you can achieve significant operational efficiencies and secure a stable source of high-quality material. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing excellence can support your product development and commercialization goals.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
