Advanced Palladium-Catalyzed Asymmetric Hydrogenation for Commercial Scale Chiral Intermediates
Advanced Palladium-Catalyzed Asymmetric Hydrogenation for Commercial Scale Chiral Intermediates
The pharmaceutical and agrochemical industries are constantly seeking more efficient pathways to access chiral building blocks that serve as the core frameworks for bioactive molecules. Patent CN111484459A introduces a groundbreaking methodology for the synthesis of chiral 3-trifluoromethyl-3,4-dihydroquinoxalinones via palladium-catalyzed asymmetric hydrogenation. This technology addresses the critical need for high-purity intermediates used in the development of potent drugs and pesticides, where the introduction of fluorine atoms often enhances metabolic stability and lipophilicity. The structural significance of this scaffold is evident in various bioactive compounds, as illustrated by the complex molecular architectures found in drug candidates like Kinin B1 and GW420867X.
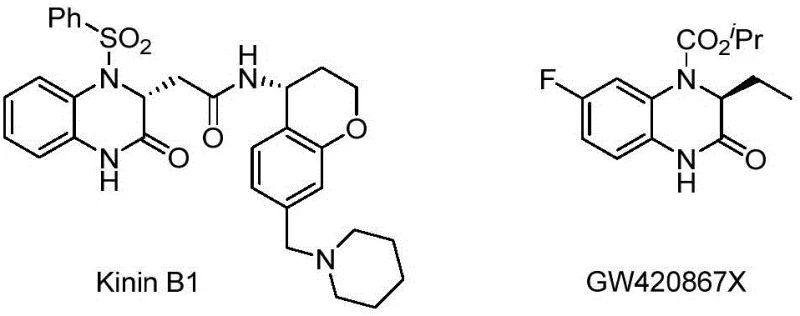
This patented approach leverages a homogeneous palladium catalytic system paired with specialized chiral bisphosphine ligands to achieve exceptional stereocontrol. By operating under relatively mild conditions ranging from 0 to 80 degrees Celsius and utilizing hydrogen pressures between 100 and 1000 psi, the process ensures high atom economy and environmental friendliness. For procurement specialists and supply chain managers, this represents a shift towards more sustainable and cost-effective manufacturing processes that reduce reliance on stoichiometric chiral reagents. The ability to produce these valuable intermediates with enantiomeric excess reaching up to 99 percent positions this technology as a vital asset for reliable pharmaceutical intermediate supplier networks aiming to optimize their production pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral dihydroquinoxalinones has been fraught with challenges related to step efficiency and waste generation. Traditional strategies often depended on the use of stoichiometric amounts of amino acid derivatives or chiral esters acting as chiral auxiliaries, which inherently increases the raw material costs and complicates the downstream purification processes. Furthermore, earlier methodologies such as the Hetero-Diels-Alder reaction, while effective, often require harsh conditions or multiple synthetic steps to install the necessary chirality, leading to lower overall yields and higher production lead times. The introduction of fluorine-containing groups into these scaffolds was particularly difficult using these legacy methods, limiting the exploration of fluorinated analogs that could offer superior biological profiles. Consequently, the industry faced significant bottlenecks in scaling up the production of these high-value intermediates without incurring prohibitive costs or generating excessive chemical waste.
The Novel Approach
The innovative method described in the patent circumvents these historical limitations by employing a direct asymmetric hydrogenation strategy using a palladium-chiral bisphosphine complex. This catalytic approach allows for the direct reduction of trifluoromethylquinoxalinone substrates to their corresponding chiral dihydroquinoxalinones with remarkable precision and efficiency. The general reaction scheme demonstrates the elegance of this transformation, where molecular hydrogen is activated by the palladium center to selectively reduce the imine bond while establishing the chiral center at the 3-position.
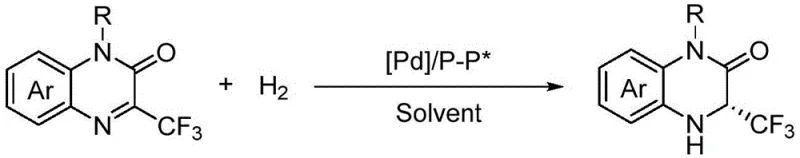
By eliminating the need for stoichiometric chiral sources, this novel approach drastically simplifies the synthetic route and improves the overall atom economy of the process. The use of fluorinated alcohol solvents such as 2,2,2-trifluoroethanol or hexafluoroisopropanol further enhances the reaction performance, providing a medium that stabilizes the transition state and promotes high enantioselectivity. For manufacturers focused on cost reduction in API manufacturing, this transition from stoichiometric to catalytic chirality induction represents a paradigm shift that aligns with green chemistry principles while delivering superior product quality.
Mechanistic Insights into Palladium-Catalyzed Asymmetric Hydrogenation
The success of this hydrogenation protocol relies heavily on the intricate interplay between the palladium metal center and the chiral environment provided by the bisphosphine ligands. The catalytic cycle begins with the formation of an active palladium-hydride species, which then coordinates with the imine substrate of the quinoxalinone ring. The chiral ligand, such as the highly effective (R)-SegPhos, creates a specific steric pocket that directs the approach of the hydride to one face of the imine bond, thereby controlling the absolute configuration of the resulting stereocenter. The optimization data reveals that ligands with specific bite angles and electronic properties, like those shown in the structural comparison of SegPhos, SynPhos, and BINAP derivatives, are crucial for achieving maximum turnover and selectivity.
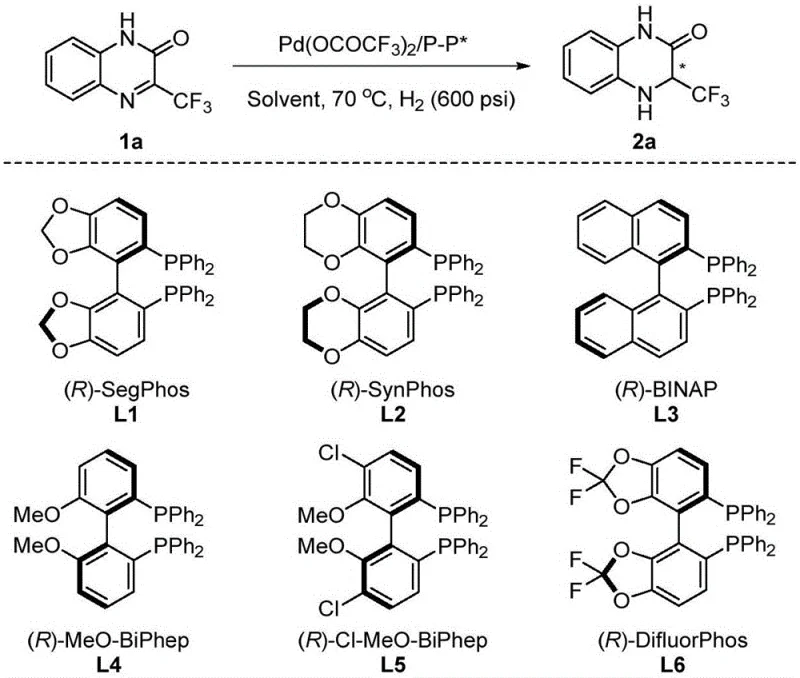
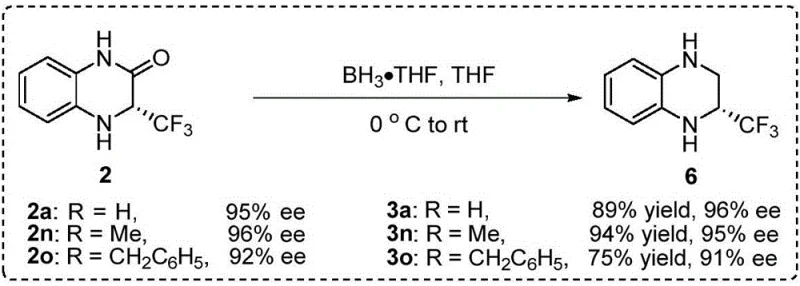
Impurity control is another critical aspect where this mechanistic understanding pays dividends, as the high specificity of the catalyst minimizes the formation of side products or over-reduced species. The patent data indicates that nuclear magnetic resonance spectroscopy detects no significant side reactions, suggesting that the catalyst system is highly chemoselective for the target imine reduction even in the presence of other functional groups. This level of purity is essential for pharmaceutical applications where strict regulatory standards dictate the impurity profile of active ingredients. Furthermore, the versatility of the system allows for the reduction of the resulting amide to tetrahydroquinoxalines using borane reagents, expanding the utility of this platform for synthesizing diverse nitrogen-containing heterocycles.
How to Synthesize Chiral 3-Trifluoromethyl-3,4-Dihydroquinoxalinone Efficiently
Implementing this synthesis route requires careful attention to catalyst preparation and reaction conditions to ensure reproducibility and high optical purity. The process begins with the in situ generation of the active catalyst by mixing the palladium precursor with the chiral ligand in a volatile solvent like acetone, followed by concentration to remove the coordinating solvent. This pre-formed catalyst is then introduced to the substrate dissolved in the specialized fluorinated alcohol solvent under an inert atmosphere to prevent catalyst deactivation. Detailed standardized synthesis steps see the guide below.
- Prepare the active catalyst by stirring palladium trifluoroacetate and a chiral bisphosphine ligand such as (R)-SegPhos in acetone at room temperature, followed by vacuum concentration.
- Dissolve the trifluoromethylquinoxalinone substrate in hexafluoroisopropanol (HFIP) under a nitrogen atmosphere and add the prepared catalyst solution.
- Introduce hydrogen gas at pressures between 100-1000 psi and maintain the reaction temperature between 0-80°C for 15-48 hours to achieve high enantioselectivity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this palladium-catalyzed technology offers tangible benefits that extend beyond mere technical feasibility. The shift from stoichiometric chiral auxiliaries to a catalytic system fundamentally alters the cost structure of the synthesis, removing the burden of purchasing expensive single-use chiral starting materials. This transition leads to substantially reduced raw material costs and simplifies the inventory management of critical reagents, as the catalyst loading is significantly lower than the molar equivalents required in traditional methods. Additionally, the high selectivity of the reaction minimizes the need for complex chromatographic separations, which are often the most time-consuming and solvent-intensive steps in fine chemical manufacturing.
- Cost Reduction in Manufacturing: The elimination of stoichiometric chiral sources and the high atom economy of the hydrogenation reaction drive down the overall cost of goods sold. By utilizing molecular hydrogen as the reductant, the process avoids the generation of stoichiometric waste byproducts associated with chemical reducing agents, leading to significant savings in waste disposal and treatment costs. The robustness of the catalyst system also implies longer operational lifetimes and potential for recycling, further enhancing the economic viability of large-scale production runs.
- Enhanced Supply Chain Reliability: The starting materials for this synthesis, specifically the trifluoromethylquinoxalinones, are readily accessible and can be produced from commodity chemicals, ensuring a stable supply base. The mild reaction conditions reduce the risk of thermal runaways or equipment failures, contributing to consistent batch-to-batch quality and reliable delivery schedules. This stability is crucial for maintaining continuous production lines for critical pharmaceutical intermediates where supply interruptions can have cascading effects on downstream drug manufacturing.
- Scalability and Environmental Compliance: The use of fluorinated alcohol solvents, while specialized, allows for efficient solvent recovery and reuse due to their distinct physical properties, aligning with modern environmental regulations. The process operates at moderate pressures and temperatures that are compatible with standard industrial hydrogenation reactors, facilitating easy scale-up from laboratory to commercial tonnage without requiring exotic equipment. This scalability ensures that the technology can meet growing market demands for chiral fluorinated intermediates while maintaining a reduced environmental footprint.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this asymmetric hydrogenation technology. These insights are derived directly from the experimental data and scope defined in the patent documentation to provide clarity on process capabilities. Understanding these details helps stakeholders evaluate the fit of this technology within their existing manufacturing frameworks.
Q: What represents the primary advantage of this Pd-catalyzed method over traditional synthetic routes?
A: Unlike traditional methods relying on stoichiometric chiral auxiliaries or complex Hetero-Diels-Alder reactions, this palladium-catalyzed asymmetric hydrogenation offers superior atom economy and eliminates the need for expensive chiral starting materials, significantly simplifying the purification process.
Q: How does the choice of solvent impact the enantioselectivity in this reaction?
A: The use of fluorinated alcohol solvents like hexafluoroisopropanol (HFIP) is critical, as they enhance the reaction rate and enantioselectivity through specific hydrogen-bonding interactions with the substrate and catalyst complex, often yielding over 99% ee.
Q: Is this synthesis method scalable for industrial production of API intermediates?
A: Yes, the process utilizes mild reaction conditions (0-80°C) and robust catalyst systems that are highly suitable for scale-up, allowing for the commercial production of complex chiral intermediates with consistent quality and reduced environmental impact.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Trifluoromethyl-3,4-Dihydroquinoxalinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic technologies like the one described in CN111484459A for the production of high-value chiral intermediates. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods are successfully translated into robust industrial processes. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of chiral 3-trifluoromethyl-3,4-dihydroquinoxalinone meets the exacting standards required by global pharmaceutical clients.
We invite you to contact our technical procurement team to discuss how we can integrate this efficient hydrogenation route into your supply chain. Request a Customized Cost-Saving Analysis today to understand the specific economic benefits for your project, and ask for specific COA data and route feasibility assessments tailored to your target molecules. Let us partner with you to deliver high-purity pharmaceutical intermediates with the reliability and quality your business demands.
