Scalable Synthesis of 17alpha-Estradiol: Advanced Stereoselective Inversion for Pharmaceutical Manufacturing
Scalable Synthesis of 17alpha-Estradiol: Advanced Stereoselective Inversion for Pharmaceutical Manufacturing
The pharmaceutical landscape is increasingly recognizing the distinct therapeutic potential of 17alpha-estradiol, a stereoisomer of the potent natural estrogen 17beta-estradiol. Unlike its beta-counterpart, 17alpha-estradiol exhibits negligible feminizing effects while retaining significant neuroprotective and metabolic benefits, positioning it as a critical candidate for treating age-related cognitive decline and metabolic syndrome. However, the commercial availability of this high-value intermediate has historically been constrained by inefficient synthetic routes plagued by low yields and complex impurity profiles. Patent CN101665526A introduces a groundbreaking methodology that addresses these bottlenecks through a novel stereoselective inversion strategy. This technical insight report analyzes the proprietary chemistry disclosed in the patent, highlighting how this approach enables the reliable production of high-purity 17alpha-estradiol intermediates suitable for demanding clinical applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 17alpha-estradiol has relied on the inversion of 17beta-precursors, a transformation that is chemically challenging due to the steric hindrance at the D-ring of the steroid nucleus. Prior art methods, such as those described in US Pat. 3,652,605, typically employ polar aprotic solvents like DMF and utilize alkali metal salts of carboxylic acids at elevated temperatures. While theoretically sound, practical replication of these legacy protocols reveals severe deficiencies. The reaction environment often promotes the formation of a unstable 17-position carbonium ion intermediate. This cationic species is highly prone to elimination reactions, leading to the formation of olefinic byproducts, or rearrangement pathways that generate structurally related impurities difficult to separate. Consequently, the actual isolated yield of the target 17alpha-isomer in these conventional processes often plummets to merely 20-30%, rendering the process economically unviable for large-scale manufacturing and creating significant waste disposal challenges.
The Novel Approach
The methodology disclosed in CN101665526A represents a paradigm shift in steroid stereochemistry control. By transitioning from polar protic or aprotic systems to non-polar organic solvents such as toluene or xylene, the inventors have fundamentally altered the reaction mechanism to favor direct nucleophilic substitution. The core innovation lies in the synergistic use of a Lewis acid, specifically inorganic lithium salts like lithium chloride, combined with an amine organic base and a short-chain carboxylic acid. This unique reagent system generates an organic ammonium carboxylate salt in situ, which possesses superior solubility in the non-polar medium. This solubility allows for the effective delivery of the carboxylate nucleophile to the sterically crowded C17 position without generating free carbocations. As demonstrated in the reaction scheme below, this approach facilitates a clean SN2 inversion of the 17beta-sulfonate to the 17alpha-ester with exceptional efficiency.
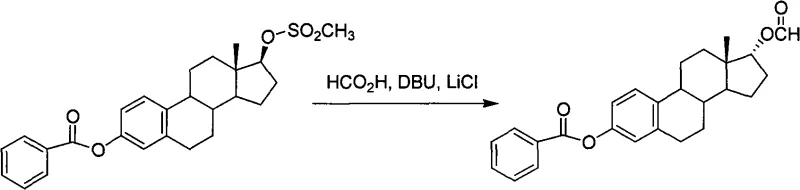
Furthermore, the process incorporates a sophisticated pH control strategy. The reaction is initiated at a neutral to slightly basic pH (6-8) to optimize the concentration of the active nucleophile, and subsequently adjusted to an acidic range (pH 2-3) towards the end of the reaction. This dynamic adjustment accelerates the consumption of residual starting material while preventing the degradation of the newly formed 17alpha-ester. The result is a robust process that consistently delivers yields exceeding 95% for the inversion step, a dramatic improvement over the single-digit or low-double-digit yields of previous generations.
Mechanistic Insights into Lewis Acid-Mediated Stereoinversion
To fully appreciate the technical superiority of this route, one must examine the mechanistic role of the Lewis acid and the amine base complex. In traditional SN2 reactions on steroid scaffolds, the bulky D-ring creates a significant energy barrier for the approaching nucleophile. The addition of lithium chloride acts as a Lewis acid that likely coordinates with the sulfonate leaving group, enhancing its lability without promoting full ionization to a carbocation. Simultaneously, the amine base (such as DBU or DMAP) reacts with the carboxylic acid to form a lipophilic ion pair. This ion pair effectively solubilizes the carboxylate anion in the non-polar toluene phase, increasing its local concentration near the substrate. The intermediate structure, generally represented as Formula A in the patent documentation, is a 3-hydroxyl-protected 17alpha-estradiol-17-carboxylic acid ester.
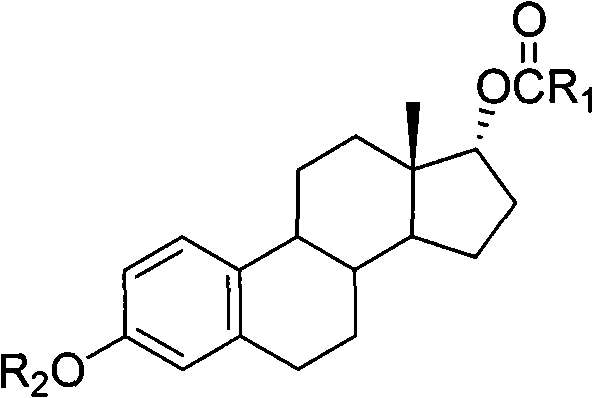
This specific intermediate architecture is crucial for impurity control. By maintaining the reaction in a non-ionizing solvent and avoiding strong mineral acids during the inversion step, the pathway for E2 elimination is effectively blocked. The stereochemical outcome is strictly governed by the backside attack of the carboxylate on the C17 carbon, ensuring complete inversion from the beta to the alpha configuration. The subsequent hydrolysis step, performed under mild alkaline conditions, cleaves both the 17-ester and the 3-protecting group (typically a benzoyl or acetyl group) simultaneously. This telescoped deprotection simplifies the workflow, eliminating the need for intermediate isolation and further reducing the opportunity for epimerization or degradation, thereby securing the high optical purity required for pharmaceutical grade 17alpha-estradiol.
How to Synthesize 17alpha-Estradiol Efficiently
The synthesis of 17alpha-estradiol via this patented route involves a streamlined sequence that begins with the activation of commercially available 17beta-estradiol derivatives. The process is designed for operational simplicity, utilizing standard reactor equipment and readily available reagents. The initial step involves the protection of the 3-hydroxyl group and the conversion of the 17-hydroxyl group into a good leaving group, such as a mesylate or triflate. Following this activation, the critical stereoinversion is performed in a non-polar solvent system under reflux conditions. Detailed standardized operating procedures, including precise molar ratios of the Lewis acid catalyst and the specific temperature ramps required to maximize yield while minimizing side reactions, are outlined in the technical guide below.
- Preparation of 3-hydroxyl-protected 17beta-estradiol-17-sulfonate starting materials using sulfonyl chlorides and organic bases.
- Stereoselective inversion reaction in non-polar solvents using Lewis acids, amine bases, and carboxylic acids to form 17alpha-estradiol-17-carboxylates.
- Hydrolysis of the 17alpha-ester intermediate under alkaline conditions to yield high-purity 17alpha-estradiol.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthesis route offers compelling strategic advantages beyond mere technical elegance. The shift away from polar aprotic solvents like DMF to non-polar solvents like toluene significantly simplifies solvent recovery and recycling processes. Toluene is easier to distill and reuse compared to high-boiling polar solvents, leading to substantial reductions in raw material costs and waste treatment expenses. Moreover, the elimination of transition metal catalysts, which are often required in alternative coupling strategies, removes the need for expensive and time-consuming heavy metal scavenging steps. This simplification of the downstream processing train directly translates to shorter batch cycle times and lower operational expenditures.
- Cost Reduction in Manufacturing: The most significant economic driver of this technology is the drastic improvement in reaction yield. By pushing the inversion step yield from a historical average of roughly 30% to over 95%, the effective cost of goods sold (COGS) for the active pharmaceutical ingredient is markedly reduced. This efficiency gain means that less starting material is required to produce the same amount of final product, optimizing the utilization of expensive steroid precursors. Additionally, the high selectivity of the reaction minimizes the formation of difficult-to-remove impurities, reducing the load on chromatographic purification columns and extending their operational lifespan, which further drives down manufacturing costs.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as lithium chloride, triethylamine, and formic acid ensures a robust and resilient supply chain. Unlike specialized catalysts that may have single-source suppliers or long lead times, the reagents for this process are globally available in bulk quantities. This commoditization of inputs mitigates the risk of supply disruptions. Furthermore, the process operates at atmospheric pressure and moderate temperatures (80-120°C), which allows for production in standard glass-lined or stainless steel reactors found in most multipurpose chemical facilities, ensuring that production capacity can be easily scaled or shifted between sites without specialized infrastructure investments.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this process aligns well with modern green chemistry principles. The use of non-polar solvents reduces the generation of hazardous aqueous waste streams often associated with polar solvent washes. The high atom economy of the inversion step means less chemical waste is generated per kilogram of product. The ability to recycle the toluene solvent and the absence of toxic heavy metals in the final product stream simplify regulatory compliance and wastewater treatment, facilitating faster environmental permitting for new production lines and ensuring long-term operational continuity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and claims presented in the patent literature, providing a factual basis for evaluating the feasibility of integrating 17alpha-estradiol into your development pipeline. Understanding these nuances is essential for R&D teams planning scale-up activities and quality assurance teams establishing specification limits.
Q: How does this synthesis method prevent the formation of elimination byproducts?
A: The method utilizes a specific combination of Lewis acids and amine organic bases in non-polar solvents to facilitate an SN2 nucleophilic substitution. This effectively suppresses the formation of the 17-position carbonium ion intermediate, which is the primary cause of elimination and rearrangement side products in conventional acidic or polar protic conditions.
Q: What are the purity specifications achievable with this novel route?
A: By avoiding the harsh conditions of prior art methods that lead to significant impurity profiles, this process achieves HPLC purity levels exceeding 96-98% for the final 17alpha-estradiol product after recrystallization, significantly reducing the burden on downstream purification.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the process employs common non-polar solvents like toluene and avoids expensive transition metal catalysts. The reaction conditions are robust, with yields for the inversion step reaching near-quantitative levels in optimized embodiments, making it highly viable for metric-ton scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 17alpha-Estradiol Supplier
The synthesis of 17alpha-estradiol described in CN101665526A represents a mature and highly efficient pathway for producing this valuable hormonal intermediate. At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless. Our facility is equipped with rigorous QC labs capable of verifying stringent purity specifications, including chiral HPLC analysis to guarantee the correct stereochemical configuration of the 17-alpha hydroxyl group. We understand that consistency is key in pharmaceutical manufacturing, and our process controls are designed to deliver batch-after-batch reproducibility.
We invite potential partners to engage with our technical team to discuss how this advanced synthesis route can be tailored to your specific project needs. Whether you require custom kilo-lab quantities for preclinical studies or metric-ton volumes for commercial launch, we are prepared to provide a Customized Cost-Saving Analysis that quantifies the economic benefits of switching to this superior method. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us support your journey in bringing next-generation hormone therapies to market.
