Advanced Synthesis of Vortioxetine Impurity Reference Standards for Global QC Labs
Introduction to High-Purity Impurity Standards
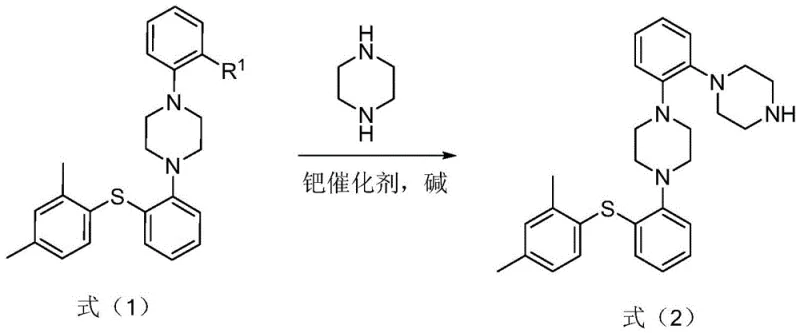
The rigorous demands of modern pharmaceutical quality control necessitate the availability of highly characterized impurity reference standards, particularly for complex antidepressants like Vortioxetine hydrobromide. Patent CN114075153A introduces a robust and scalable preparation method for the specific impurity 1-[2-[(2,4-dimethylphenyl)thio]phenyl]-4-[2-(1-piperazinyl)phenyl]piperazine, designated as Formula (2) within the document. This compound represents a critical byproduct formed during the synthesis of the active pharmaceutical ingredient, and its precise quantification is essential for meeting stringent regulatory safety profiles. The disclosed technology leverages advanced transition metal catalysis to overcome historical limitations in yield and purity, offering a reliable pathway for producing reference materials that ensure the safety and efficacy of the final drug product.
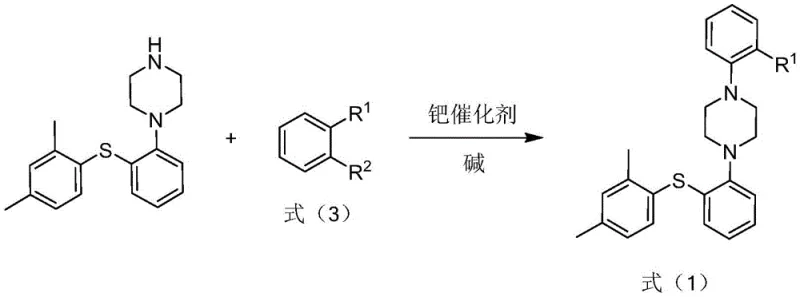
For research and development directors overseeing analytical method validation, the ability to source this impurity with guaranteed purity levels above 99% is paramount. The patent details a sophisticated two-step synthetic strategy that begins with the coupling of Vortioxetine (or its salts) with o-bromoiodobenzene to generate a key intermediate, Formula (1). This intermediate is subsequently reacted with piperazine under optimized palladium-catalyzed conditions to afford the target molecule. By strictly controlling reaction parameters such as temperature, solvent choice, and catalyst loading, the inventors have established a protocol that minimizes side reactions and maximizes the recovery of the desired impurity standard, thereby supporting comprehensive quality research and limit setting for intermediates and bulk drugs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this innovation, the synthesis of this specific Vortioxetine impurity was fraught with significant technical challenges that hindered efficient production. Existing literature, such as the route disclosed in J. Pharm. Biomed. Anal., described a synthetic pathway that proved inadequate for large-scale or high-purity applications. When attempted, these conventional methods resulted in substantial amounts of unreacted starting materials and a complex mixture of byproducts, making the isolation of the target compound extremely difficult. The low conversion rates meant that extensive and costly purification steps, such as repeated column chromatography, were required, which drastically reduced the overall yield and increased the operational complexity. Furthermore, the lack of clear optimization data in previous reports left manufacturers struggling with inconsistent batch quality and unreliable supply chains for this critical reference standard.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes a highly optimized palladium-catalyzed cross-coupling strategy that delivers superior performance metrics. The process employs a specific catalyst system consisting of bis(dibenzylideneacetone)palladium and a BINAP ligand, paired with sodium tert-butoxide as the base in mesitylene solvent. This combination facilitates a clean and efficient transformation, achieving yields as high as 82.2% for the final step and purities reaching 99.0%. The method allows for precise control over the reaction environment, typically operating between 130°C and 140°C, which ensures complete consumption of starting materials while suppressing the formation of unwanted side products. Additionally, the patent highlights the feasibility of a one-pot synthesis, where the second coupling step can be performed sequentially without isolating the intermediate, further streamlining the manufacturing process and reducing solvent waste.
Mechanistic Insights into Pd-Catalyzed Buchwald-Hartwig Amination
The core of this technological breakthrough lies in the meticulous application of the Buchwald-Hartwig amination mechanism, a powerful tool for forming carbon-nitrogen bonds. In the second step of the synthesis, the aryl halide moiety of Formula (1) undergoes oxidative addition with the zero-valent palladium catalyst, forming a reactive organopalladium intermediate. The presence of the bulky and electron-rich BINAP ligand is crucial here, as it stabilizes the palladium center and accelerates the subsequent coordination and deprotonation of the piperazine nucleophile. This is followed by reductive elimination, which releases the final product, Formula (2), and regenerates the active catalyst species. The choice of sodium tert-butoxide as the base is mechanistically significant, as it effectively generates the reactive amido species necessary for the transmetallation step without promoting competing elimination pathways that could degrade the sensitive thioether linkage present in the molecule.
From an impurity control perspective, this catalytic system offers exceptional selectivity. The steric hindrance provided by the ligand prevents non-specific coupling at other potential sites on the aromatic rings, ensuring that the piperazine attaches exclusively at the intended position ortho to the sulfur linkage. This high regioselectivity is vital for producing a reference standard that accurately mimics the specific impurity profile found in the drug substance. Moreover, the use of mesitylene as a high-boiling solvent allows the reaction to proceed at elevated temperatures (130-140°C) required to overcome the activation energy barrier of the sterically hindered substrates, ensuring rapid kinetics and high conversion. The result is a clean reaction profile that simplifies downstream processing, as the crude product requires only crystallization from ethanol to achieve analytical grade purity, eliminating the need for labor-intensive chromatographic separations.
How to Synthesize 1-[2-[(2,4-dimethylphenyl)thio]phenyl]-4-[2-(1-piperazinyl)phenyl]piperazine Efficiently
The synthesis of this complex heterocyclic impurity requires precise adherence to the optimized conditions outlined in the patent to ensure reproducibility and high quality. The process involves dissolving the precursor compounds in an inert organic solvent and heating them under a nitrogen atmosphere to prevent catalyst oxidation. The addition of the palladium source and ligand must be carefully timed to maximize catalytic turnover, while the stoichiometric ratio of base to substrate is critical for driving the equilibrium towards product formation. Detailed standardized operating procedures for this synthesis, including specific molar ratios and work-up protocols, are provided in the structured guide below to assist technical teams in replicating these results.
- React Vortioxetine (or salt) with o-bromoiodobenzene using Pd(dba)2, BINAP ligand, and sodium tert-butoxide in mesitylene at 130-140°C to form Formula (1).
- React the resulting Formula (1) intermediate with anhydrous piperazine under similar palladium catalysis conditions to yield the final impurity Formula (2).
- Purify the crude product via crystallization from ethanol to achieve high purity (>99%) suitable for reference standards.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this patented synthesis route offers tangible benefits that extend beyond mere technical feasibility. The primary advantage lies in the drastic simplification of the purification workflow. By achieving such high crude purity through selective catalysis, the need for expensive and time-consuming preparative chromatography is largely eliminated. This reduction in processing steps translates directly into lower manufacturing costs and shorter production cycles, allowing for more responsive fulfillment of orders for reference standards. Furthermore, the raw materials required, such as Vortioxetine salts and o-bromoiodobenzene, are commercially available and stable, reducing the risk of supply disruptions associated with exotic or hard-to-source reagents.
- Cost Reduction in Manufacturing: The elimination of complex purification stages significantly lowers the operational expenditure associated with producing this impurity standard. Traditional methods often required multiple rounds of chromatography, which consume vast quantities of solvents and silica gel, driving up both material costs and waste disposal fees. By relying on a high-yield crystallization process instead, the new method reduces solvent consumption and labor hours, leading to substantial cost savings per gram of product. Additionally, the high atom economy of the coupling reaction ensures that expensive starting materials are converted efficiently into the desired product rather than being lost as waste, further optimizing the cost structure for large-scale production runs.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route ensures a consistent and reliable supply of high-purity impurity standards, which is critical for pharmaceutical companies maintaining their quality control laboratories. The use of common industrial solvents like mesitylene and toluene, along with stable catalyst systems, means that production is not dependent on fragile or specialized supply chains. This stability allows suppliers to maintain healthy inventory levels and offer shorter lead times to global clients. The ability to perform the synthesis in a one-pot manner also reduces the turnaround time between batches, enabling manufacturers to scale up production rapidly in response to fluctuating market demand without compromising on quality or delivery schedules.
- Scalability and Environmental Compliance: From an environmental and scalability standpoint, this process aligns well with green chemistry principles by minimizing waste generation. The high selectivity of the reaction reduces the formation of hazardous byproducts, simplifying effluent treatment and lowering the environmental footprint of the manufacturing facility. The reaction conditions, while requiring elevated temperatures, operate within the safe limits of standard glass-lined or stainless steel reactors commonly found in fine chemical plants, facilitating easy scale-up from kilogram to multi-ton quantities. This scalability ensures that the supply of this critical reference material can grow in tandem with the increasing global production of Vortioxetine, securing the supply chain for years to come.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this Vortioxetine impurity standard. These answers are derived directly from the experimental data and specifications provided in the patent documentation, ensuring accuracy and relevance for industry professionals. Understanding these details helps stakeholders make informed decisions regarding the sourcing and utilization of this critical quality control material.
Q: What is the primary advantage of this synthesis method over prior art?
A: The patented method significantly improves yield (up to 82.2%) and purity (99.0%) compared to literature methods which suffered from low conversion and difficult purification.
Q: Can the synthesis be performed in a single pot?
A: Yes, the patent discloses a one-pot variation (Example 10) where the second reagent is added directly after the first reaction completes, streamlining the workflow.
Q: What catalyst system is recommended for optimal results?
A: The patent prefers a system comprising bis(dibenzylideneacetone)palladium [Pd(dba)2] as the catalyst and 1,1'-binaphthyl-2,2'-bis-diphenylphosphine (BINAP) as the ligand.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Vortioxetine Impurity Supplier
As the global demand for high-quality antidepressant medications continues to rise, the need for precise impurity profiling becomes increasingly critical for regulatory compliance and patient safety. NINGBO INNO PHARMCHEM stands at the forefront of this sector, leveraging advanced catalytic technologies like the one described in CN114075153A to deliver reference standards of unparalleled purity. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the rigorous volume requirements of major pharmaceutical manufacturers. With our stringent purity specifications and rigorous QC labs, we guarantee that every batch of Vortioxetine impurity supplied meets the highest international standards for analytical accuracy and reliability.
We invite procurement directors and R&D leaders to collaborate with us to optimize their supply chains for critical reference materials. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis that identifies opportunities to reduce expenses without compromising on quality. We encourage you to contact our technical procurement team today to request specific COA data for our current inventory and to discuss route feasibility assessments for your specific project needs. Let us be your trusted partner in ensuring the quality and safety of your pharmaceutical products through superior chemical solutions.
