Revolutionizing Cobicistat Intermediate Production via Chiral Reductive Amination
The global demand for antiretroviral therapies continues to drive the need for efficient manufacturing of key pharmaceutical intermediates, particularly for boosting agents like cobicistat. Patent CN109912426B introduces a transformative synthetic strategy for producing (2R,5R)-1,6-diphenyl-2,5-hexanediamine, a critical chiral building block. Unlike conventional approaches that struggle with low yields or expensive starting materials, this invention leverages a clever reductive amination pathway using readily available diketone precursors. By shifting the paradigm from post-synthesis resolution to direct chiral induction, the technology offers a robust solution for cost reduction in pharmaceutical intermediates manufacturing. This report analyzes the technical merits of this route, providing actionable insights for R&D directors seeking purity and procurement teams focused on supply chain stability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 1,6-diphenyl-2,5-hexanediamine has been plagued by significant economic and technical inefficiencies. One prevalent route utilizes L-phenylalaninol as a chiral pool starting material; however, this amino alcohol is prohibitively expensive, and the multi-step sequence involving protection, oxidation, and coupling often results in suboptimal overall yields. Another common strategy involves the alkylation of adipic acid derivatives followed by Hofmann degradation. While the raw materials are cheap, this approach lacks inherent stereocontrol, producing a racemic mixture that necessitates a resolution step. This resolution process inherently discards more than half of the synthesized material as the unwanted enantiomer, creating a massive bottleneck in atom economy and waste management. Furthermore, the harsh reaction conditions required for alpha-alkylation often lead to difficult-to-remove impurities, complicating downstream purification and increasing the total cost of ownership for the final API.
The Novel Approach
The methodology disclosed in CN109912426B circumvents these historical bottlenecks by employing a symmetrical diketone, Compound II, as the central scaffold. This intermediate is easily constructed from succinic acid derivatives and benzyl Grignard reagents, ensuring a stable and low-cost supply chain foundation. The core innovation lies in the condensation of this diketone with specific chiral primary amines, such as (R)-methylbenzylamine or (R)-1-(1-naphthyl)ethylamine. This reaction forms a bis-imine intermediate where the chiral information from the amine auxiliary dictates the stereochemical outcome of the subsequent reduction. By utilizing bulky chiral amines, the process achieves high diastereoselectivity directly, eliminating the need for wasteful resolution steps. This streamlined logic not only shortens the synthetic route but also drastically simplifies the purification profile, making it an ideal candidate for commercial scale-up of complex pharmaceutical intermediates.
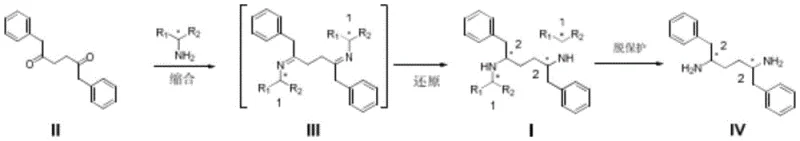
Mechanistic Insights into Chiral Reductive Amination
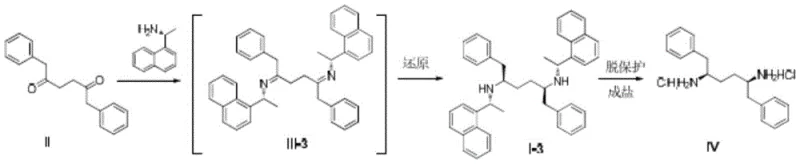
The heart of this technology is the stereoselective reduction of the bis-imine intermediate. When Compound II reacts with a chiral amine, it forms a Schiff base (Imine III) where the nitrogen atoms are bonded to the chiral auxiliaries. The spatial arrangement of these auxiliaries creates a specific chiral environment around the carbon-nitrogen double bonds. During the reduction phase, whether using metal borohydrides like sodium borohydride or catalytic hydrogenation with palladium on carbon, the hydride source attacks the imine carbon from the less hindered face. The patent data highlights a critical structure-activity relationship: the size of the substituent on the chiral amine profoundly impacts selectivity. For instance, using simple benzylamine results in a racemic mixture due to a lack of steric differentiation. However, switching to (R)-1-(1-naphthyl)ethylamine introduces significant steric bulk from the naphthyl ring system. This bulk effectively shields one face of the imine, forcing the reduction to occur with high fidelity on the opposite face, thereby locking in the desired (2R,5R) configuration with exceptional precision.
Impurity control is intrinsically built into this mechanism through the choice of the chiral auxiliary. In traditional resolution methods, the unwanted enantiomer is a structural twin of the product, making separation energy-intensive and yield-limiting. In this novel route, the "impurity" is essentially the diastereomer formed by incorrect facial attack, which is minimized by the steric bulk of the naphthyl group. Experimental data from the patent indicates that using (R)-1-(1-naphthyl)ethylamine can achieve a diastereomeric ratio of up to 96:4 favoring the target isomer. Following the reduction, the chiral auxiliary is removed via catalytic hydrogenolysis. This step is clean and efficient, typically using Pd-C under hydrogen pressure, which cleaves the benzyl-type C-N bonds to release the free diamine and recover the chiral amine auxiliary. This recovery potential further enhances the economic viability of the process, as the expensive chiral amine can potentially be recycled, contributing to reducing lead time for high-purity pharmaceutical intermediates by simplifying the overall material flow.
How to Synthesize (2R,5R)-1,6-Diphenyl-2,5-hexanediamine Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for laboratory and pilot-scale production. The process begins with the preparation of the symmetrical diketone precursor, followed by a one-pot or two-step reductive amination. The flexibility of the method allows for the use of various reducing agents, accommodating different facility capabilities. Whether utilizing chemical reduction with borohydrides for smaller batches or catalytic hydrogenation for larger scales, the core chemistry remains robust. The final deprotection step is equally straightforward, relying on standard hydrogenation equipment found in most fine chemical plants. For detailed operational parameters, safety guidelines, and specific stoichiometric ratios required for GMP compliance, please refer to the standardized synthesis guide below.
- Condense 1,6-diphenyl-3-hexanedione (Compound II) with a chiral primary amine such as (R)-1-(1-naphthyl)ethylamine to form the bis-imine intermediate.
- Perform stereoselective reduction of the imine bonds using metal borohydrides or catalytic hydrogenation to establish the desired (2R,5R) configuration.
- Execute catalytic hydrogenolysis using Pd-C to remove the chiral auxiliary groups, yielding the target high-purity diamine hydrochloride salt.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this synthetic route represents a strategic opportunity to optimize costs and secure supply continuity. The primary advantage stems from the decoupling of the process from expensive chiral pool materials like L-phenylalaninol. By relying on commodity chemicals such as succinic esters and benzyl halides for the backbone, and widely available chiral amines for induction, the raw material risk profile is significantly lowered. This diversification of the supply base ensures that production is not held hostage by the volatility of niche amino alcohol markets. Furthermore, the elimination of the resolution step fundamentally alters the cost structure. In resolution-based processes, the theoretical maximum yield is capped at 50% unless dynamic kinetic resolution is employed, which adds complexity. This direct induction method bypasses that cap, effectively doubling the potential output per unit of backbone material input compared to racemic routes, leading to substantial cost savings in manufacturing.
- Cost Reduction in Manufacturing: The economic model of this process is strengthened by the high atom economy and the avoidance of wasteful separation techniques. Traditional methods that generate racemates require discarding or recycling the unwanted enantiomer, which incurs significant processing costs and material loss. By achieving high diastereoselectivity upfront, this method minimizes the burden on purification units like preparative HPLC or repeated crystallizations. Additionally, the potential to recover and reuse the chiral amine auxiliary after the deprotection step creates a closed-loop system for the most expensive reagent. This circular approach to reagent usage means that the effective cost per kilogram of the final diamine is drastically reduced, offering a competitive edge in pricing for the final cobicistat API.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of robust, non-sensitive intermediates. The diketone precursor (Compound II) is chemically stable and can be stockpiled without significant degradation, allowing manufacturers to buffer against upstream fluctuations. Unlike routes requiring cryogenic conditions or highly sensitive organometallic reagents that demand specialized logistics, this synthesis operates under mild conditions (often ambient temperature for condensation and moderate pressure for hydrogenation). This operational simplicity reduces the dependency on specialized equipment and highly trained personnel, making it easier to qualify multiple contract manufacturing organizations (CMOs). Consequently, buyers can establish a multi-vendor strategy more easily, ensuring that the supply of this critical HIV drug intermediate remains uninterrupted even during global logistical disruptions.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, the route is exceptionally clean. The avoidance of heavy metal catalysts for chirality induction (relying instead on organic chiral amines) simplifies the impurity profile regarding residual metals, a critical quality attribute for pharmaceutical regulators. The waste stream is primarily composed of organic salts and recovered amines, which are easier to treat than the complex mixtures generated by resolution mother liquors. The process is amenable to large-scale batch processing, as the exotherms associated with the reduction steps are manageable with standard cooling systems. This scalability ensures that as the demand for cobicistat grows, the manufacturing capacity can be expanded linearly without encountering the nonlinear cost increases often associated with chromatographic separations or low-yield coupling reactions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology. They are derived from the specific experimental data and comparative analysis provided in the patent documentation, aiming to clarify the practical benefits for potential licensees and manufacturing partners.
Q: How does this new method improve upon traditional resolution processes?
A: Traditional methods using adipic acid derivatives often produce racemic mixtures requiring resolution, which theoretically wastes over 50% of the material. This patent utilizes chiral amines like (R)-1-(1-naphthyl)ethylamine to induce stereochemistry directly during synthesis, achieving high diastereomeric ratios (up to 96:4) without the need for wasteful separation steps.
Q: What are the key starting materials for this synthesis?
A: The process begins with Compound II, a symmetrical diketone derived from succinyl chloride or succinate esters reacting with benzyl Grignard reagents. These precursors are commodity chemicals, significantly lowering the raw material cost compared to routes relying on expensive L-phenylalaninol.
Q: Can the chirality be controlled effectively without expensive ligands?
A: Yes. Instead of costly chiral ligands that require complex recovery, this method uses stoichiometric chiral amines which act as both reactants and chiral inducers. The bulky naphthyl group provides superior steric hindrance, ensuring high selectivity during the reduction phase.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cobicistat Intermediate Supplier
The technological advancements detailed in CN109912426B underscore the importance of having a manufacturing partner who understands both the chemistry and the commercial implications of process optimization. NINGBO INNO PHARMCHEM stands at the forefront of this evolution, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facilities are equipped with the necessary hydrogenation reactors and purification trains to execute this reductive amination route with precision. We adhere to stringent purity specifications and operate rigorous QC labs to ensure that every batch of (2R,5R)-1,6-diphenyl-2,5-hexanediamine meets the exacting standards required for antiretroviral drug synthesis. Our commitment to quality ensures that the high diastereomeric ratios achieved in the lab are faithfully reproduced at the industrial scale.
We invite global pharmaceutical companies and procurement leaders to engage with us for a Customized Cost-Saving Analysis tailored to your specific volume requirements. By leveraging this efficient synthetic route, we can help you significantly lower the cost of goods for your cobicistat supply chain. Please contact our technical procurement team to request specific COA data from our pilot runs and comprehensive route feasibility assessments. Let us collaborate to secure a sustainable and cost-effective supply of this vital life-saving medicine intermediate.
