Scalable Biocatalytic Synthesis of Chiral Epoxides for Advanced Pharmaceutical Manufacturing
Scalable Biocatalytic Synthesis of Chiral Epoxides for Advanced Pharmaceutical Manufacturing
The pharmaceutical industry is currently witnessing a paradigm shift towards sustainable and highly selective synthetic methodologies, particularly for the production of chiral building blocks essential for beta-blocker drugs and other active pharmaceutical ingredients. Patent CN110564755B introduces a groundbreaking biocatalytic approach for the preparation of (S)-3-phenyl-1,2-epoxypropane and its derivatives, utilizing a novel alanine-phosphoribitol ligase (HhAPL) derived from Herbaspirillum huttiense. This technology addresses critical bottlenecks in traditional chemical synthesis by offering a route that combines exceptional enantioselectivity with environmentally benign reaction conditions. For R&D directors and procurement specialists, this represents a significant opportunity to streamline supply chains for high-value chiral intermediates while adhering to increasingly stringent environmental regulations regarding heavy metal usage and organic solvent waste.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of optically pure 1,2-epoxypropane compounds has relied heavily on chemical methods such as the Sharpless asymmetric epoxidation or chemical resolution of racemates. While effective, the Sharpless protocol necessitates the use of stoichiometric amounts of titanium tetraisopropoxide and chiral tartrate esters, leading to substantial production costs and the generation of hazardous metal-containing waste streams that require complex and expensive removal processes. Furthermore, alternative biocatalytic routes using styrene monooxygenases have shown promise but suffer from significant drawbacks, including low diastereoselectivity for certain substrate olefins and poor enantioselectivity (often less than 86% ee) when acting on terminally nonconjugated olefins without ortho-hydroxyl substitution. These limitations result in lower overall yields, increased raw material consumption, and a larger environmental footprint, making them less attractive for modern, cost-sensitive commercial manufacturing.
The Novel Approach
The methodology disclosed in patent CN110564755B overcomes these historical challenges by employing a recombinant Escherichia coli strain expressing the HhAPL gene to catalyze the asymmetric epoxidation of allylbenzene derivatives. This whole-cell biocatalytic system operates efficiently in an aqueous potassium phosphate buffer system, eliminating the need for precious metal catalysts and toxic organic solvents typically associated with chemical oxidation. The process demonstrates remarkable versatility, successfully converting a wide range of substrates including methyl-substituted allylbenzenes, methoxy derivatives, and even challenging substrates like 2-allylphenol with high conversion rates. By shifting the reaction medium to water and utilizing renewable biocatalysts, this approach not only simplifies the workup procedure but also aligns perfectly with green chemistry principles, offering a robust platform for the sustainable production of high-purity chiral epoxides.

Mechanistic Insights into HhAPL-Catalyzed Asymmetric Epoxidation
The core of this technological advancement lies in the unique catalytic properties of the HhAPL enzyme, which functions as a highly efficient monooxygenase mimic within the cellular environment of E. coli. Unlike traditional chemical oxidants that rely on harsh electrophilic oxygen transfer, the HhAPL enzyme facilitates a stereospecific oxygen insertion into the carbon-carbon double bond of the allylbenzene substrate. The genetic engineering strategy involves codon optimization of the HhAPL gene for E. coli expression, ensuring high levels of soluble protein production that drive the reaction kinetics. The enzyme exhibits a profound preference for the formation of the (S)-enantiomer, achieving enantiomeric excess values exceeding 99% for the parent allylbenzene substrate. This high level of stereocontrol is attributed to the precise spatial arrangement of the active site residues, which restrict the substrate orientation during the oxygen transfer event, thereby minimizing the formation of the unwanted (R)-isomer and reducing the burden on downstream chiral purification steps.
Beyond simple epoxidation, the HhAPL system demonstrates sophisticated kinetic resolution capabilities when presented with racemic secondary alcohol substrates such as (±)-1-phenyl-2-propenol. In these reactions, the enzyme selectively epoxidizes one enantiomer of the racemic mixture while leaving the other largely untouched, effectively acting as a dynamic kinetic resolution agent. This dual functionality allows for the simultaneous production of chiral epoxides and the recovery of unreacted chiral alcohols, maximizing atom economy and resource utilization. The mechanism involves a highly specific interaction between the hydroxyl group of the substrate and the enzyme's binding pocket, which dictates the diastereoselectivity of the epoxidation event. This capability is particularly valuable for synthesizing complex intermediates where multiple chiral centers need to be established with high fidelity, providing R&D teams with a powerful tool for constructing intricate molecular architectures found in next-generation therapeutics.
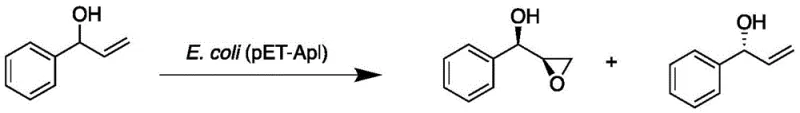
How to Synthesize (S)-3-phenyl-1,2-epoxypropane Efficiently
The implementation of this biocatalytic route involves a streamlined fermentation and bioconversion process that is readily adaptable to existing industrial infrastructure. The synthesis begins with the construction of the expression vector pET-Apl, followed by the transformation of E. coli host cells and subsequent fermentation to generate the biocatalyst. The actual epoxidation reaction is conducted under mild physiological conditions, typically between 25°C and 40°C, using a biphasic system where n-octane serves as a co-solvent to enhance substrate solubility without compromising enzyme activity. This operational simplicity reduces the need for specialized high-pressure or cryogenic equipment, lowering the barrier to entry for commercial adoption. For detailed standard operating procedures regarding cell culture conditions, induction protocols, and downstream extraction methods, please refer to the comprehensive guide below.
- Construct the expression vector pET-Apl by inserting the codon-optimized HhAPL gene into the pET24(a) vector and transform it into E. coli BL21 cells.
- Cultivate the recombinant cells in TB medium with kanamycin, induce expression with IPTG at 16°C, and harvest the wet cells via centrifugation.
- Resuspend the cells in potassium phosphate buffer, add the allylbenzene substrate with n-octane co-solvent, and react at 30-40°C to obtain the target chiral epoxide.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this HhAPL-mediated biocatalytic process offers transformative benefits for procurement managers and supply chain leaders focused on cost reduction and risk mitigation. The elimination of expensive transition metal catalysts such as titanium and vanadium, which are staples in traditional Sharpless epoxidation, directly translates to significant raw material cost savings. Furthermore, the absence of heavy metals in the reaction mixture removes the necessity for rigorous and costly metal scavenging steps during purification, thereby shortening the overall production cycle time and reducing the consumption of specialized adsorbents. The use of water as the primary reaction medium also drastically lowers the volume of volatile organic compounds (VOCs) emitted, simplifying regulatory compliance and reducing waste disposal fees associated with hazardous solvent incineration.
- Cost Reduction in Manufacturing: The transition to a whole-cell biocatalytic system fundamentally alters the cost structure of chiral epoxide production by removing the dependency on precious metal reagents and chiral ligands that drive up the bill of materials in chemical synthesis. The high turnover number of the HhAPL enzyme ensures that a small amount of biocatalyst can convert large quantities of substrate, improving the catalyst-to-product ratio and lowering the unit cost of goods sold. Additionally, the simplified downstream processing, which avoids complex metal removal and extensive chromatographic separations often required for chemically synthesized racemates, leads to substantial operational expenditure reductions. These efficiencies compound at scale, making the biocatalytic route increasingly economically attractive as production volumes increase from pilot to commercial scales.
- Enhanced Supply Chain Reliability: Relying on fermentation-derived enzymes rather than finite mineral resources or complex chemical syntheses for catalysts enhances the resilience of the supply chain against raw material price volatility and geopolitical disruptions. The E. coli expression system is a well-established, robust platform that can be rapidly scaled up using standard fermenters, ensuring consistent and reliable supply of the biocatalyst. Moreover, the stability of the recombinant cells allows for the potential storage of frozen biomass, creating a buffer inventory that can be deployed on demand to meet fluctuating market needs. This flexibility ensures that manufacturers can maintain continuous production schedules without being held hostage by the lead times associated with sourcing specialized chemical reagents or resolving supply bottlenecks for rare earth metals.
- Scalability and Environmental Compliance: The inherent safety profile of conducting reactions in aqueous buffers at near-ambient temperatures significantly reduces the operational risks associated with exothermic chemical oxidations that often require strict thermal control to prevent runaway reactions. This safety margin facilitates easier scale-up from laboratory benchtop to multi-ton industrial reactors without the need for extensive re-engineering of safety systems. From an environmental standpoint, the process generates significantly less hazardous waste, aligning with global sustainability goals and corporate social responsibility mandates. The reduced environmental footprint not only mitigates regulatory risks but also enhances the brand value of the final pharmaceutical products by appealing to eco-conscious stakeholders and consumers who prioritize green manufacturing practices.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this biocatalytic technology. These insights are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing a clear understanding of the process capabilities and limitations. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What are the advantages of this enzymatic method over Sharpless epoxidation?
A: Unlike Sharpless epoxidation which requires expensive titanium catalysts and generates heavy metal waste, this biocatalytic method uses whole-cell E. coli in an aqueous buffer, eliminating metal contamination risks and significantly reducing downstream purification costs.
Q: Can this process handle substrates with ortho-hydroxyl groups?
A: Yes, the HhAPL enzyme demonstrates high diastereoselectivity and activity towards substrates like 2-allylphenol, overcoming the limitations of styrene monooxygenases which often show poor selectivity for such structurally complex olefins.
Q: Is this method suitable for large-scale commercial production?
A: Absolutely. The process utilizes robust E. coli fermentation technology and mild reaction conditions (30-40°C, atmospheric pressure), making it highly scalable from kilogram to multi-ton production without the safety hazards associated with peracid oxidants.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (S)-3-phenyl-1,2-epoxypropane Supplier
As the demand for high-purity chiral intermediates continues to surge in the pharmaceutical sector, partnering with a technically proficient CDMO is essential for translating innovative patent technologies into commercial reality. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of (S)-3-phenyl-1,2-epoxypropane delivered meets the highest quality standards required for API synthesis. We understand the critical nature of supply continuity and are committed to providing a stable, high-quality source of these vital building blocks.
We invite procurement directors and R&D leaders to engage with our technical procurement team to discuss how this biocatalytic route can be tailored to your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain a detailed understanding of the economic benefits specific to your volume needs. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that optimize both your product quality and your bottom line. Let us be your strategic partner in navigating the complexities of modern chiral synthesis.
