Advanced Synthesis of Nucleoside Diphosphite Amides for High-Purity Oligonucleotide Manufacturing
The landscape of antisense oligonucleotide therapeutics is rapidly evolving, driven by the urgent need for higher purity standards and more efficient synthetic routes. Patent CN107556355B, published on October 22, 2021, introduces a groundbreaking preparation method for nucleoside diphosphite amides, a critical class of intermediates in the solid-phase synthesis of antisense drugs. This technology addresses a persistent bottleneck in the industry: the formation of complex impurities during the monomer coupling stage. By utilizing a novel protecting group strategy based on 4-methoxy-4'-acetoxy trityl chloride, the invention offers a pathway to significantly enhance the quality profile of final oligonucleotide products. For R&D directors and procurement specialists alike, understanding this mechanistic shift is vital for securing a reliable nucleoside diphosphite amide supplier capable of meeting the stringent specifications required for clinical-grade manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of oligonucleotide monomers has relied heavily on 4,4'-bismethoxytrityl chloride (DMTCl) to protect the 5'-hydroxyl group of nucleosides. However, this conventional approach suffers from inherent stability issues. During the preparation and storage of DMTCl, a degradation product known as 4-methoxy-4'-hydroxytrityl chloride is frequently generated. When this impurity participates in the coupling reaction, it leads to the formation of structurally similar byproducts, specifically the nucleoside diphosphite amide monomer depicted in Formula (I). These impurities are notoriously difficult to separate from the desired product due to their similar physicochemical properties, thereby complicating downstream purification and jeopardizing the overall yield and safety of the therapeutic agent.
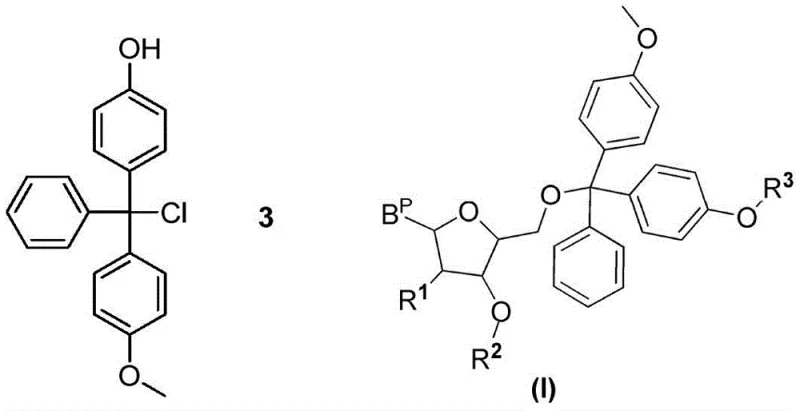
The Novel Approach
To circumvent these challenges, the patented methodology introduces a strategic modification to the trityl protecting group. Instead of using standard DMTCl directly, the process synthesizes a specialized intermediate: 4-methoxy-4'-acetoxy trityl chloride (Compound 1). This novel reagent is designed to prevent the formation of the problematic hydroxyl-containing impurities during the critical coupling phases. By incorporating an acetoxy group that can be selectively removed later, the synthesis ensures that only the desired mono-protected nucleoside is formed initially. This structural precision effectively eliminates the root cause of the impurity cascade, resulting in a much cleaner reaction profile and facilitating easier purification protocols for large-scale synthesis.
Mechanistic Insights into Selective Demethylation and Phosphitylation
The core innovation lies in the precise control of functional group transformations on the trityl backbone. The process begins with the selective demethylation of 4,4'-bis(methoxy)trityl chloride using boron tribromide (BBr3) at a strictly controlled temperature of 0°C. This low-temperature condition is paramount; it ensures that only one methoxy group is cleaved to form the phenol intermediate (Compound A) without causing hydrolysis of the trityl chloride moiety or over-demethylation. Following this, the phenolic hydroxyl is protected via acetylation using acetyl chloride under heating conditions, yielding Compound B. The final activation step involves chlorination to regenerate the reactive trityl chloride center, producing the key intermediate, 4-methoxy-4'-acetoxy trityl chloride (Compound 1).
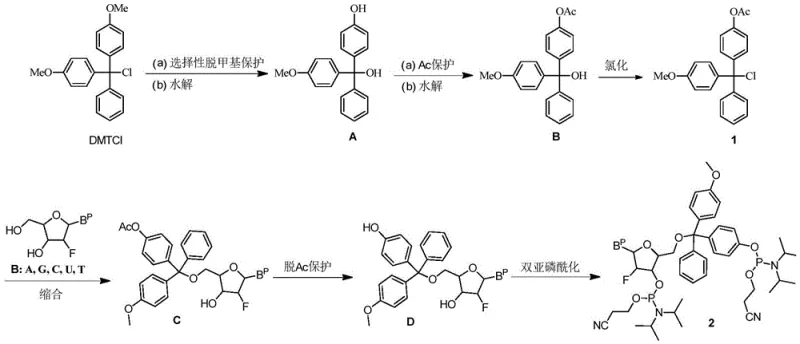
Subsequent condensation with 2'-halonucleosides proceeds with high regioselectivity at the 5'-position. The acetoxy group serves as a temporary mask that prevents side reactions, and it is subsequently removed under mild ammoniacal conditions to reveal the free hydroxyl necessary for the final phosphitylation. The double phosphorylation reaction, utilizing bis(diisopropylamino)(2-cyanoethoxy)phosphine, converts the diol intermediate into the target diphosphite amide. This multi-step sequence is engineered to maximize atom economy while minimizing the generation of hard-to-remove side products, offering a robust solution for cost reduction in oligonucleotide manufacturing where purity is the primary cost driver.
How to Synthesize Nucleoside Diphosphite Amide Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for producing these high-value intermediates with consistent quality. The procedure leverages standard organic synthesis techniques such as solvent extraction, crystallization, and column chromatography, making it highly adaptable for industrial scale-up. The detailed steps involve careful monitoring of reaction progress via HPLC to ensure complete conversion at each stage, particularly during the selective demethylation and final phosphitylation steps. For a comprehensive breakdown of the specific reagents, stoichiometry, and workup procedures required to replicate this high-yield process, please refer to the standardized guide below.
- Selective demethylation of 4,4'-bismethoxytrityl chloride using boron tribromide at 0°C followed by hydrolysis.
- Acetyl protection of the hydroxyl group and subsequent chlorination to form 4-methoxy-4'-acetoxy trityl chloride.
- Condensation with 2'-halonucleosides, deprotection, and double phosphorylation to yield the final diphosphite amide.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthesis route translates into tangible operational benefits beyond mere chemical elegance. The primary advantage is the drastic simplification of the purification workflow. By preventing the formation of the specific impurity shown in Formula (I) at the source, manufacturers can reduce the number of chromatographic passes required to achieve pharmaceutical-grade purity. This reduction in processing steps directly correlates to lower solvent consumption, reduced waste generation, and shorter production cycles, all of which contribute to substantial cost savings in the final price of the active pharmaceutical ingredient.
- Cost Reduction in Manufacturing: The elimination of difficult-to-separate impurities means that less material is lost during the rigorous purification stages typically required for oligonucleotide monomers. In traditional processes, significant yield loss occurs when trying to separate closely related byproducts. By utilizing the AcO-MMT-Cl intermediate, the process achieves higher effective yields of the desired isomer, thereby optimizing the utilization of expensive nucleoside starting materials and reducing the overall cost of goods sold (COGS) without compromising on quality standards.
- Enhanced Supply Chain Reliability: The reagents used in this pathway, such as boron tribromide, acetyl chloride, and standard phosphitylating agents, are commercially available bulk chemicals with stable supply chains. Unlike proprietary catalysts that may face sourcing bottlenecks, this method relies on established commodity chemicals. This ensures that production schedules remain robust and less susceptible to raw material shortages, providing a reliable nucleoside diphosphite amide supplier with the ability to maintain continuous output even during market fluctuations.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing crystallization steps (e.g., for Compound 1) that are easily transferable from kilogram to tonne scales. Furthermore, the improved selectivity reduces the volume of hazardous waste solvents associated with extensive recycling and purification. This aligns with modern green chemistry principles and environmental regulations, making the commercial scale-up of complex oligonucleotide intermediates more sustainable and compliant with increasingly strict global environmental standards.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and benefits of this patented synthesis method. These insights are derived directly from the experimental data and mechanistic descriptions provided in the patent documentation, offering clarity on how this technology integrates into existing manufacturing frameworks.
Q: How does this new synthesis method improve impurity control compared to traditional DMTCl methods?
A: Traditional DMTCl often contains 4-methoxy-4'-hydroxytrityl chloride impurities which lead to unwanted side products during oligonucleotide synthesis. This patent introduces a novel intermediate, 4-methoxy-4'-acetoxy trityl chloride, which allows for precise control over the protecting group introduction, significantly reducing the formation of difficult-to-remove impurities like those shown in Formula (I).
Q: What are the key reaction conditions for the selective demethylation step?
A: The process utilizes boron tribromide (BBr3) in dichloromethane at a controlled temperature of 0°C. This low-temperature condition is critical for achieving selective mono-demethylation without degrading the trityl chloride structure, ensuring high regioselectivity before subsequent acetylation.
Q: Can this method be scaled for commercial production of antisense drugs?
A: Yes, the patent describes a robust pathway using standard reagents like acetyl chloride and pyridine. The reported yields (e.g., 70-85% for the final phosphitylation step) and the use of crystallization for purification indicate that the process is amenable to scale-up for industrial manufacturing of oligonucleotide intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Nucleoside Diphosphite Amide Supplier
As the demand for antisense therapies continues to surge, the need for high-purity, reliably sourced intermediates has never been greater. NINGBO INNO PHARMCHEM stands at the forefront of this sector, leveraging advanced synthetic methodologies like the one described in CN107556355B to deliver superior products. Our facility boasts extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the volumetric demands of global pharmaceutical partners. With our stringent purity specifications and rigorous QC labs, we guarantee that every batch of nucleoside diphosphite amide meets the exacting standards required for clinical and commercial oligonucleotide synthesis.
We invite you to collaborate with us to optimize your supply chain and reduce your manufacturing costs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific project needs. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can accelerate your drug development timeline.
