Advanced Synthesis of Nucleoside-5'-Phosphodiesters via Lithium Salt Condensation for Commercial Scale-up
Advanced Synthesis of Nucleoside-5'-Phosphodiesters via Lithium Salt Condensation for Commercial Scale-up
The development of robust synthetic routes for nucleoside analogs remains a critical frontier in medicinal chemistry, particularly for antiviral and oncology therapeutics where bioavailability is often limited by poor cellular uptake. Patent CN112279877B introduces a transformative methodology for synthesizing nucleoside-5'-phosphodiesters, addressing long-standing inefficiencies in prodrug manufacturing. By utilizing a diester phosphate lithium salt directly in a condensation reaction with protected nucleosides, this innovation circumvents the need for cumbersome ion exchange resin treatments and hazardous catalytic hydrogenation steps. For R&D directors and process chemists, this represents a significant leap forward in designing cleaner, more efficient pathways for high-value pharmaceutical intermediates. The technology not only streamlines the synthetic workflow but also enhances the safety profile of the manufacturing process, making it an attractive candidate for reliable nucleoside phosphate supplier partnerships aiming for GMP-compliant production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of nucleoside-5'-phosphodiesters has been plagued by operational complexities and safety hazards inherent to traditional phosphorylation strategies. One prominent legacy method involves the Mitsunobu reaction, which typically employs triphenylphosphine (PPh3) and diethyl azodicarboxylate (DEAD) to couple phosphodiesters with nucleosides. As illustrated in the reaction scheme below, while chemically feasible, this approach suffers from notoriously low synthesis yields and generates stoichiometric amounts of phosphine oxide byproducts that are difficult to remove, complicating downstream purification.
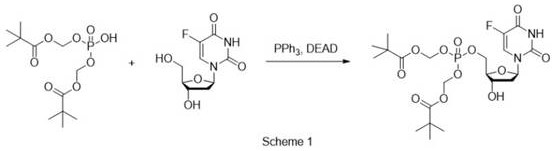
Furthermore, alternative routes reported by Imbach et al. utilize phosphorus oxychloride or require the synthesis of phosphoric diester triethylamine salts followed by catalytic debenzylation. These methods introduce severe safety liabilities, specifically the requirement for hydrogen gas and palladium on carbon (Pd/C) catalysts, which pose significant fire and explosion risks in large-scale reactors. Additionally, the conversion of phosphoric diesters to phosphorus oxychloride generates substantial quantities of corrosive hydrogen chloride gas, necessitating specialized corrosion-resistant equipment and rigorous scrubbing systems. These factors collectively drive up capital expenditure and operational risk, creating bottlenecks for the commercial scale-up of complex pharmaceutical intermediates.
The Novel Approach
In stark contrast to these legacy techniques, the method disclosed in CN112279877B leverages the unique reactivity of diester phosphate lithium salts to achieve direct condensation under mild conditions. This novel approach fundamentally alters the process landscape by eliminating the ion exchange resin step traditionally required to purify phosphoric acid diester precursors. Instead, the lithium salt is used directly, reacting efficiently with nucleosides in the presence of modern condensing agents like BOP-Cl (bis(2-oxo-3-oxazolidinyl)phosphinic chloride). The general reaction pathway demonstrates a streamlined convergence of the phosphate and nucleoside components without the need for hazardous hydrogenation or corrosive chlorinating agents.
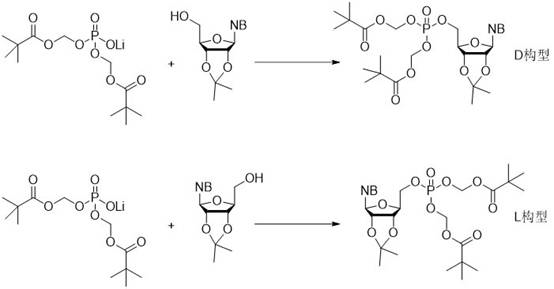
This strategic shift offers profound advantages for cost reduction in pharmaceutical intermediates manufacturing. By removing the ion exchange resin step, the process reduces solvent consumption, waste generation, and cycle time. Moreover, the avoidance of Pd/C hydrogenation not only mitigates safety risks but also removes the need for expensive catalyst recovery and validation processes associated with heavy metal removal. The result is a synthesis protocol that is not only chemically elegant but also industrially pragmatic, offering a clear path toward high-purity nucleoside phosphate production with a significantly reduced environmental footprint.
Mechanistic Insights into BOP-Cl Mediated Phosphorylation
The core of this technological advancement lies in the activation mechanism facilitated by the BOP-Cl condensing agent in conjunction with 3-nitro-1,2,4-triazole. In this system, the lithium salt of the diester phosphate acts as a nucleophile, attacking the activated phosphorus species generated in situ. The use of lithium counter-ions is critical; unlike bulky organic amines, lithium ions offer a favorable balance of solubility in polar aprotic solvents like tetrahydrofuran (THF) and reactivity, allowing the phosphate anion to remain sufficiently free for nucleophilic attack while maintaining stability. The reaction proceeds at ambient temperatures (0-30°C), which is crucial for preserving the stereochemical integrity of sensitive nucleoside substrates, particularly those with labile protecting groups.
Impurity control is inherently built into this mechanism through the choice of reagents and workup conditions. The use of 3-nitro-1,2,4-triazole as an additive enhances the leaving group ability during the activation step, driving the reaction to completion and minimizing the formation of unreacted starting materials. Post-reaction, the quenching with citric acid serves a dual purpose: it neutralizes basic residues like diisopropylethylamine (DIEA) and facilitates the partitioning of polar impurities into the aqueous phase. This careful orchestration of reaction parameters ensures that the final crude product requires minimal purification, often achievable through standard column chromatography, thereby supporting the production of high-purity pharmaceutical intermediates suitable for subsequent biological evaluation.
How to Synthesize Nucleoside-5'-Phosphodiester Efficiently
The practical implementation of this synthesis route is designed for scalability and reproducibility in a standard chemical manufacturing environment. The process begins with the dissolution of the lithium diester phosphate and the protected nucleoside in a suitable solvent system, typically tetrahydrofuran, though acetonitrile or dichloromethane may also be employed depending on solubility profiles. The sequential addition of activators at controlled temperatures ensures a steady reaction rate, preventing exotherms that could degrade the product. Following the reaction period, a straightforward aqueous workup involving citric acid and brine washes effectively removes inorganic salts and urea byproducts derived from the condensing agent.
- Dissolve lithium diester phosphate and protected nucleoside in tetrahydrofuran (THF) or alternative solvents like acetonitrile or DMF.
- Add condensing agents including 3-nitro-1,2,4-triazole, diisopropylethylamine (DIEA), and BOP-Cl at 0-30°C, then stir at room temperature for 1-12 hours.
- Quench the reaction with citric acid solution, wash the organic phase with water and brine, dry over anhydrous sodium sulfate, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this lithium salt condensation technology translates directly into enhanced operational resilience and cost efficiency. The elimination of ion exchange resin treatment removes a significant variable from the supply chain, as resin availability, regeneration capacity, and disposal costs are no longer factors in the production equation. This simplification leads to a more predictable manufacturing timeline and reduces the dependency on specialized consumables that can sometimes face market shortages. Furthermore, the mild reaction conditions allow for the use of standard glass-lined or stainless steel reactors without the need for exotic alloys required to handle corrosive hydrogen chloride gas, thereby lowering capital barriers for contract manufacturing organizations.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the removal of the ion exchange resin purification step and the avoidance of catalytic hydrogenation. By bypassing the resin treatment, manufacturers save significantly on solvent volumes, resin replacement costs, and the labor hours associated with column packing and regeneration. Additionally, eliminating the Pd/C hydrogenation step removes the expense of precious metal catalysts and the rigorous analytical testing required to ensure residual metal levels meet regulatory limits. These cumulative savings result in a substantially lower cost of goods sold (COGS) for the final nucleoside phosphate intermediate.
- Enhanced Supply Chain Reliability: The reliance on stable, commercially available lithium salts rather than sensitive triethylamine salts or hazardous phosphorus oxychloride enhances supply security. Lithium salts are generally robust and easier to store and transport, reducing the risk of degradation during logistics. Moreover, the avoidance of hydrogen gas and pyrophoric catalysts simplifies facility safety compliance, allowing production to proceed in a wider range of manufacturing sites without requiring specialized high-pressure hydrogenation suites. This flexibility ensures consistent supply continuity even during periods of heightened regulatory scrutiny or facility maintenance.
- Scalability and Environmental Compliance: From an environmental perspective, this method offers a greener alternative to traditional phosphorylation routes. The absence of corrosive HCl gas generation means reduced burden on scrubber systems and lower risk of equipment corrosion-related leaks. The process generates less hazardous waste, as there are no phosphine oxides from Mitsunobu reactions or spent palladium catalysts to dispose of. This alignment with green chemistry principles facilitates easier permitting and environmental compliance, making the commercial scale-up of complex nucleoside intermediates more sustainable and socially responsible.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this nucleoside phosphate synthesis technology. These insights are derived directly from the experimental data and beneficial effects described in the patent literature, providing a clear understanding of the method's capabilities and limitations for potential partners.
Q: Why is the lithium salt of diester phosphate preferred over the triethylamine salt in this synthesis?
A: The use of lithium salt eliminates the need for ion exchange resin treatment during raw material preparation, which simplifies the process and avoids the complex purification operations associated with resin handling. Additionally, it bypasses the need for catalytic debenzylation using hydrogen and Pd/C, significantly reducing fire and explosion hazards.
Q: What types of nucleoside bases are compatible with this phosphorylation method?
A: This method is highly versatile and supports naturally occurring bases such as adenine, guanine, cytosine, and uracil, as well as their derivatives. It is effective for both D-configuration and L-configuration nucleosides, making it suitable for a wide range of antiviral and anticancer drug intermediates.
Q: How does this method improve safety compared to traditional phosphorus oxychloride routes?
A: Traditional methods often require converting phosphoric diesters to phosphorus oxychloride, which generates large amounts of corrosive hydrogen chloride gas. This novel route avoids that conversion entirely, preventing equipment corrosion and eliminating the generation of hazardous acidic gases, thereby enhancing overall plant safety.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Nucleoside Phosphate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and safe synthetic routes in the development of next-generation antiviral and anticancer therapies. Our technical team has extensively analyzed the lithium salt condensation methodology described in CN112279877B and is fully prepared to translate this laboratory-scale innovation into robust commercial processes. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can transition seamlessly from clinical trials to market launch. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of nucleoside phosphate intermediate meets the highest global regulatory standards.
We invite you to collaborate with us to leverage this advanced technology for your pipeline projects. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific molecule, demonstrating exactly how this resin-free, hydrogenation-free route can optimize your budget. We encourage you to contact us today to request specific COA data for similar compounds and comprehensive route feasibility assessments, ensuring that your supply chain is built on a foundation of scientific excellence and commercial reliability.
