Advanced Catalytic Synthesis of Dichloro Diphthalimide Intermediates for Engineering Plastics
Introduction to Advanced Imide Monomer Synthesis
The landscape of high-performance engineering plastics is undergoing a significant transformation, driven by the demand for materials that can withstand extreme thermal and mechanical stresses. At the forefront of this innovation is the development of advanced monomers, specifically the dichloro diphthalimide intermediate, which serves as a critical building block for polyimide and polyetherimide polymers. A recent technological breakthrough, documented in patent CN114195697B, introduces a highly efficient synthetic route that overcomes the historical limitations of yield and purity associated with these complex molecules. This patent details a novel catalytic system that utilizes high-boiling solvents and specific acid catalysts to facilitate the condensation of chlorophthalic anhydride with various diamines. By addressing the fundamental challenges of reaction kinetics and byproduct formation, this method offers a robust pathway for the industrial production of high-purity dichloro diphthalimide intermediates. For R&D directors and procurement strategists, understanding the nuances of this patented process is essential for securing a reliable dichloro diphthalimide intermediate supplier capable of meeting the rigorous standards of the aerospace and electronics industries.
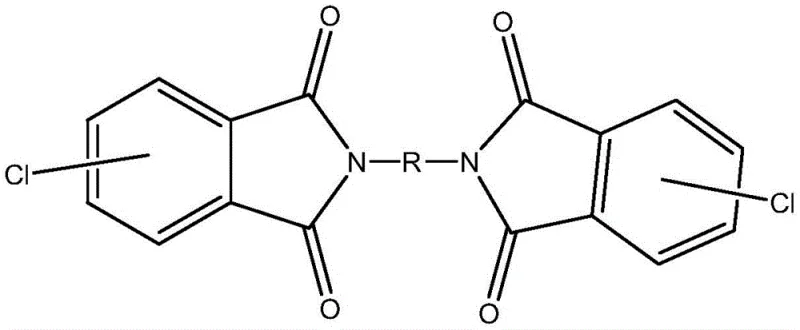
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of dichlorobisphthalimide intermediates has been plagued by significant technical hurdles that hindered large-scale commercialization and consistent quality. Prior art, such as the method described in patent CN108164452A, relied on inert solvents like paraffin without the addition of catalysts or dehydrating agents. This approach suffered from inherently slow reaction rates and low yields, primarily because the absence of a catalytic promoter failed to lower the activation energy barrier effectively. Furthermore, the lack of a dehydrating agent allowed water, a byproduct of the imidization reaction, to accumulate, which often reversed the reaction or led to hydrolysis. A critical operational failure in these older methods was the tendency of intermediate products or byproducts to agglomerate and form hard clumps within the paraffin solvent. This agglomeration made heat transfer and mass transfer extremely difficult, rendering the reaction process hard to control and posing safety risks during scale-up. Another existing method, disclosed in patent CN106232678A, utilized molten solvents like diphenyl sulfone but imposed draconian requirements on water content, mandating levels below 100ppm. Such strict moisture control necessitates expensive drying equipment and rigorous atmospheric controls, drastically increasing capital expenditure and operational complexity for any manufacturer attempting to adopt this technology.
The Novel Approach
In stark contrast to these cumbersome legacy techniques, the methodology outlined in CN114195697B introduces a sophisticated yet operationally simple solution that leverages acid catalysis and chemical dehydration. This novel approach employs high-boiling aprotic solvents such as DMAc, DMF, or NMP, which provide a homogeneous reaction medium that prevents the agglomeration issues seen in paraffin-based systems. The core innovation lies in the synergistic use of a high-boiling acid catalyst, such as sulfuric acid or phosphoric acid, alongside a dehydrating agent like acetic anhydride. The acid catalyst not only accelerates the reaction kinetics but also creates an acidic environment that suppresses the competing polycondensation reaction that leads to unwanted polyamide impurities. Simultaneously, the dehydrating agent actively scavenges the water produced during imidization, shifting the chemical equilibrium decisively towards the desired dichloro diphthalimide product. This dual-action mechanism ensures that the reaction proceeds smoothly at moderate temperatures between 50-200°C, eliminating the need for the extreme conditions or ultra-dry environments required by previous methods. The result is a process that yields a uniform solid suspension upon completion, which is exceptionally easy to filter and purify, thereby streamlining the entire downstream processing workflow.
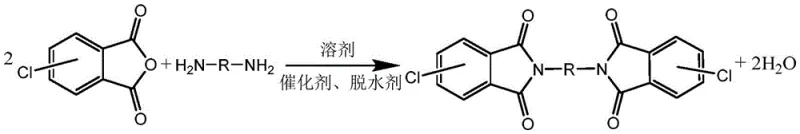
Mechanistic Insights into Acid-Catalyzed Imidization
To fully appreciate the technical superiority of this synthesis route, one must delve into the mechanistic role of the acid catalyst and the dehydrating agent within the reaction matrix. The imidization reaction between chlorophthalic anhydride and a diamine is fundamentally a nucleophilic acyl substitution followed by cyclodehydration. In the absence of a catalyst, the nucleophilicity of the amine group can be inconsistent, and the elimination of water is often the rate-limiting step. The introduction of a strong acid catalyst protonates the carbonyl oxygen of the anhydride, making the carbonyl carbon significantly more electrophilic and thus more susceptible to nucleophilic attack by the amine. This reduction in activation energy allows the reaction to proceed rapidly even at lower temperatures, minimizing thermal degradation of sensitive functional groups. Moreover, the acidic environment plays a crucial role in impurity control; by protonating free amine groups, the acid temporarily reduces their nucleophilicity, preventing them from reacting with other anhydride molecules to form linear polyamide chains instead of the cyclic imide structure. This selective suppression of polyamide formation is critical for achieving the high purity levels (>99%) required for high-performance polymer applications.
Complementing the catalytic action is the function of the dehydrating agent, which serves as a chemical sink for the water molecule released during the ring-closing step. In traditional thermal imidization, removing water often requires high vacuum or azeotropic distillation, which can be energy-intensive. Here, agents like acetic anhydride react chemically with the generated water to form acetic acid. Interestingly, this hydrolysis of the dehydrating agent generates additional acid in situ, effectively increasing the concentration of the catalyst as the reaction progresses. This autocatalytic effect ensures that the reaction rate is maintained or even accelerated towards completion, preventing the stalling that often occurs in equilibrium-limited reactions. The resulting reaction mixture forms a uniform suspension of the product, which precipitates out of the solvent upon cooling due to its low solubility at lower temperatures. This physical behavior is advantageous for isolation, as it allows for simple filtration rather than complex extraction or distillation processes, further enhancing the feasibility of cost reduction in engineering plastic manufacturing.
How to Synthesize Dichloro Diphthalimide Efficiently
Implementing this patented synthesis route requires precise control over reaction parameters to maximize yield and purity while ensuring operational safety. The process begins with the charging of chlorophthalic anhydride, the chosen solvent, the acid catalyst, and the dehydrating agent into a reactor equipped with a nitrogen inlet and temperature control. The diamine component can be added either all at once or via a controlled dosing strategy to manage exotherms, depending on the specific reactivity of the diamine selected. Maintaining a nitrogen atmosphere is critical throughout the 5 to 20-hour reaction window to prevent oxidation and moisture ingress, which could compromise the catalyst's efficacy. Upon completion, the reaction mixture is cooled to induce precipitation, followed by filtration to isolate the crude solid. The detailed standardized synthesis steps, including specific molar ratios, temperature ramps, and purification protocols like recrystallization and alcohol washing, are outlined in the guide below to ensure reproducibility.
- Charge chlorophthalic anhydride, solvent (e.g., DMAc), acid catalyst, and dehydrating agent into a reactor under nitrogen atmosphere.
- Heat the mixture to 50-200°C and add the diamine compound gradually or all at once, maintaining reaction for 5-20 hours.
- Cool the reaction, filter the solid suspension, recrystallize the filter cake, wash with alcohol or water, and dry to obtain the pure intermediate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the transition to this catalyzed synthesis method represents a strategic opportunity to optimize costs and secure a more resilient supply of critical monomers. The elimination of complex drying requirements and the ability to use standard high-boiling solvents significantly lowers the barrier to entry for manufacturing, allowing for a broader base of qualified suppliers. This diversification reduces supply chain risk and mitigates the potential for bottlenecks that often occur with specialty chemicals requiring unique processing conditions. Furthermore, the simplicity of the workup procedure, characterized by straightforward filtration and washing, translates directly into reduced labor hours and lower utility consumption per kilogram of product. These operational efficiencies compound over time, offering substantial cost savings without compromising the stringent quality specifications demanded by the aerospace and electronics sectors.
- Cost Reduction in Manufacturing: The implementation of this acid-catalyzed process eliminates the need for expensive inert solvents like paraffin or the rigorous moisture control systems required by molten salt methods, leading to a drastic simplification of the production infrastructure. By utilizing recyclable solvents such as DMAc and generating the catalyst in situ through the hydrolysis of the dehydrating agent, the consumption of fresh raw materials is significantly minimized. The high conversion rates and suppression of byproducts mean that less material is wasted, and the yield of the desired intermediate is maximized, directly lowering the cost of goods sold. Additionally, the ability to recycle the reaction filtrate directly into subsequent batches creates a closed-loop system that further drives down solvent procurement costs and waste disposal fees.
- Enhanced Supply Chain Reliability: The robustness of this synthesis method, which tolerates a wider range of operating conditions compared to prior art, ensures consistent production output and reduces the likelihood of batch failures. The use of commercially available and stable raw materials, such as chlorophthalic anhydride and common diamines, means that supply is not dependent on niche or single-source vendors. This accessibility allows for better inventory planning and reduces the lead time for high-purity dichloro diphthalimide intermediates, enabling manufacturers to respond more agilely to market demands. The simplified purification process also shortens the overall production cycle time, allowing for faster turnover and more frequent delivery schedules to meet just-in-time manufacturing requirements.
- Scalability and Environmental Compliance: The formation of a uniform solid suspension rather than a viscous melt or agglomerated mass makes this process inherently scalable from pilot plant to full commercial production without significant re-engineering. The reduced generation of hazardous waste, coupled with the ability to recover and reuse solvents and wash liquids, aligns with increasingly strict environmental regulations and corporate sustainability goals. The absence of heavy metal catalysts or toxic reagents simplifies the handling of effluent streams, reducing the burden on wastewater treatment facilities. This environmental compatibility not only ensures regulatory compliance but also enhances the brand reputation of the end-product manufacturers who prioritize green chemistry principles in their supply chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of dichloro diphthalimide intermediates based on the insights from patent CN114195697B. These answers are derived directly from the experimental data and process descriptions provided in the patent documentation to ensure accuracy and relevance for industry professionals. Understanding these details is crucial for evaluating the feasibility of integrating this intermediate into your specific polymer formulation or procurement strategy.
Q: How does the acid catalyst improve the synthesis of dichloro diphthalimide intermediates?
A: The acid catalyst lowers the activation energy required for the imidization reaction and creates an acidic environment that inhibits the formation of polyamide byproducts, ensuring higher purity and yield compared to non-catalyzed methods.
Q: What are the advantages of using a dehydrating agent in this process?
A: The dehydrating agent, such as acetic anhydride, removes water generated during the reaction, driving the equilibrium towards product formation. Additionally, its hydrolysis can regenerate the acid catalyst, enhancing process efficiency.
Q: Can the solvent be recycled in this synthesis method?
A: Yes, the filtrate obtained after the reaction can be directly recycled into the next batch or processed for solvent recovery, significantly reducing waste and raw material costs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dichloro Diphthalimide Intermediate Supplier
As the global demand for high-performance engineering plastics continues to surge, securing a partner with deep technical expertise and proven manufacturing capabilities is paramount. NINGBO INNO PHARMCHEM stands at the forefront of this industry, leveraging advanced synthetic methodologies like the one described in CN114195697B to deliver superior quality intermediates. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements regardless of the project stage. We operate with stringent purity specifications and utilize rigorous QC labs to guarantee that every batch of dichloro diphthalimide intermediate meets the exacting standards required for aerospace and electronic applications.
We invite you to collaborate with us to explore how this optimized synthesis route can benefit your specific product line. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your current supply chain needs. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our capabilities can drive efficiency and innovation in your manufacturing processes.
