Scalable Synthesis of NHPI-Functionalized Polycarboxylic Ligands for Advanced MOF Catalysis
Scalable Synthesis of NHPI-Functionalized Polycarboxylic Ligands for Advanced MOF Catalysis
The landscape of industrial oxidation catalysis is undergoing a significant transformation, driven by the urgent need for sustainable, reusable, and highly efficient catalytic systems. Patent CN109608381B introduces a groundbreaking methodology for synthesizing N-hydroxyphthalimide (NHPI) functionalized polycarboxylic organic ligands, which serve as the critical structural building blocks for advanced Metal-Organic Frameworks (MOFs). Unlike traditional homogeneous catalysts that suffer from separation difficulties and single-use limitations, these novel ligands enable the construction of porous frameworks where the catalytic NHPI groups are covalently locked within the pore channels. This technological leap addresses the persistent challenge of catalyst leaching while maintaining the high activity associated with small-molecule NHPI derivatives. For R&D directors and procurement specialists in the fine chemical sector, this patent represents a viable pathway to producing high-purity intermediates that facilitate green oxidation processes under mild conditions, potentially replacing hazardous stoichiometric oxidants with molecular oxygen.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the immobilization of NHPI onto solid supports has been plagued by significant technical inefficiencies that hinder large-scale commercial adoption. Traditional strategies often involve physically confining NHPI molecules within the pores of existing MOFs or adsorbing them onto cross-linked polymer microspheres. While these methods attempt to impart heterogeneous characteristics to the catalyst, they fundamentally fail to prevent the gradual leaching of the active NHPI species during catalytic cycles. This leaching not only deactivates the catalyst over time, necessitating frequent replacement and increasing operational costs, but also contaminates the final product stream, creating downstream purification burdens. Furthermore, surface-only immobilization techniques typically result in low solid loading densities, meaning the catalytic efficiency per unit mass of the material is suboptimal. These inherent flaws in conventional immobilization technologies create a bottleneck for pharmaceutical and agrochemical manufacturers seeking reliable, long-life oxidation catalysts that comply with stringent purity standards.
The Novel Approach
The synthesis method disclosed in CN109608381B circumvents these legacy issues by integrating the NHPI functionality directly into the organic ligand backbone prior to MOF assembly. By designing a polycarboxylic acid ligand that inherently contains the N-hydroxyphthalimide moiety, the resulting MOF structure possesses catalytic sites that are chemically inseparable from the framework itself. This covalent integration ensures that the active nitroxyl radical sites remain stable and accessible within the pore channels throughout repeated reaction cycles. The patent highlights a streamlined three-step synthetic route that avoids the complex multi-step derivatizations typical of older methods. Specifically, the process utilizes a direct cyclization strategy that bypasses the energy-intensive dehydration of dicarboxylic acids into anhydrides, a standard prerequisite in classical imide synthesis. This simplification not only accelerates the production timeline but also drastically improves the final product purity, making it an ideal candidate for high-value applications in fine chemical manufacturing.
Mechanistic Insights into Pd-Catalyzed Coupling and Direct Cyclization
The core of this innovative synthesis lies in a robust sequence of organometallic and oxidative transformations designed for maximum atom economy and yield. The process initiates with a Suzuki-Miyaura cross-coupling reaction, where 1,4-dibromo-2,3-dimethylbenzene reacts with methyl-substituted phenylboronic acids in the presence of a palladium catalyst, specifically tetrakis(triphenylphosphine)palladium(0). This step constructs the rigid terphenyl skeleton essential for forming the porous MOF structure. Following the coupling, the methyl groups on the aromatic rings are aggressively oxidized to carboxylic acids using potassium permanganate in a pyridine-water solvent system. This oxidation is critical as it generates the coordination sites required for metal node assembly. The final and most mechanistically distinct step involves the direct reaction of the resulting polycarboxylic acid with hydroxylamine hydrochloride. Unlike traditional pathways that require isolating an anhydride intermediate, this method facilitates direct imide ring closure in pyridine, leveraging the proximity of the carboxyl groups to form the five-membered NHPI ring efficiently.
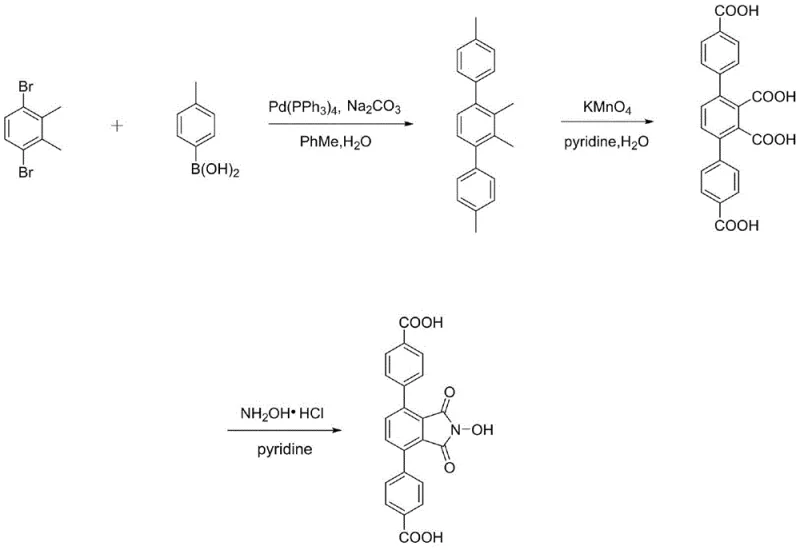
This direct cyclization mechanism is particularly advantageous for scaling operations because it eliminates a discrete processing unit operation, thereby reducing solvent consumption and waste generation. The patent data confirms that this route consistently delivers yields exceeding 96% for the final ligand, with the intermediate oxidation step achieving up to 97% conversion. Such high efficiency suggests that side reactions, such as over-oxidation or incomplete cyclization, are effectively suppressed under the specified conditions of 90°C reflux in pyridine. For technical teams evaluating process feasibility, this implies a narrow impurity profile and a simplified purification protocol involving merely acidification and centrifugation, rather than complex chromatographic separations.
How to Synthesize NHPI-Functionalized Polycarboxylic Ligands Efficiently
The synthesis of these high-performance ligands follows a logical progression from simple commodity chemicals to complex functionalized architectures. The process begins with the assembly of the carbon framework, followed by functional group interconversion, and concludes with heterocycle formation. Each stage is optimized to ensure that the intermediate products are sufficiently pure to proceed without extensive purification, which is vital for cost-effective manufacturing. The detailed standardized synthesis steps, including precise molar ratios, temperature profiles, and workup procedures, are outlined in the guide below to assist process engineers in replicating these results.
- Perform Suzuki-Miyaura coupling between 1,4-dibromo-2,3-dimethylbenzene and methylphenylboronic acid using Pd(PPh3)4 catalyst.
- Oxidize the resulting polymethyl terphenyl compound using potassium permanganate in pyridine/water to generate polycarboxylic acids.
- React the polycarboxylic acid directly with hydroxylamine hydrochloride in pyridine to form the N-hydroxyphthalimide ring.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis route offers tangible strategic benefits beyond mere technical performance. The elimination of the anhydride formation step represents a significant reduction in process complexity, which directly translates to lower capital expenditure requirements for reactor trains and reduced utility consumption. By consolidating the synthesis into fewer steps with high yields, manufacturers can achieve substantial cost savings in raw material utilization and waste disposal. Furthermore, the reliance on readily available starting materials such as dibromoxylene and phenylboronic acids ensures a stable supply chain that is less susceptible to the volatility often associated with exotic reagents. This supply security is crucial for maintaining continuous production schedules in the fast-paced pharmaceutical and agrochemical sectors.
- Cost Reduction in Manufacturing: The streamlined process architecture significantly lowers the cost of goods sold (COGS) by removing energy-intensive dehydration steps and reducing solvent turnover. The high yields reported in the patent minimize the loss of valuable intermediates, ensuring that the majority of input mass is converted into saleable product. Additionally, the simplified workup procedure, which relies on precipitation and centrifugation rather than column chromatography, is inherently more scalable and economically viable for tonnage production.
- Enhanced Supply Chain Reliability: Utilizing commodity-grade precursors like methylphenylboronic acids and common inorganic bases reduces dependency on specialized suppliers with long lead times. The robustness of the palladium-catalyzed coupling and permanganate oxidation steps means that the process is tolerant to minor variations in feedstock quality, further stabilizing the supply chain. This reliability allows procurement teams to negotiate better terms and secure long-term contracts with confidence, knowing that production bottlenecks are minimized.
- Scalability and Environmental Compliance: The synthesis route is designed with green chemistry principles in mind, utilizing water as a co-solvent in the oxidation step and avoiding hazardous chlorinating agents often used in anhydride preparation. The ability to isolate the final product via simple pH adjustment and filtration makes the process easily adaptable from kilogram-scale R&D batches to multi-ton commercial production. This scalability ensures that the technology can grow with market demand without requiring fundamental process re-engineering, supporting sustainable expansion goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this NHPI ligand synthesis technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing a clear understanding of the operational advantages.
Q: What is the primary advantage of covalently bonding NHPI to the ligand backbone?
A: Covalent bonding prevents the leaching of the catalytic active site (NHPI) from the Metal-Organic Framework (MOF) structure during reaction cycles, ensuring superior reusability and heterogeneous stability compared to physically confined catalysts.
Q: What yields can be expected from this synthesis route?
A: The patent data indicates exceptionally high yields across all steps, with the coupling step achieving approximately 85-88%, oxidation reaching 94-97%, and the final cyclization step yielding 96-97% of the pure target ligand.
Q: How does this method simplify the traditional imide formation process?
A: This novel route eliminates the conventional requirement to dehydrate the dicarboxylic acid into an anhydride prior to reaction. Instead, it allows for direct cyclization with hydroxylamine hydrochloride, significantly reducing process complexity and improving product purity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable NHPI Ligand Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of covalently functionalized MOF ligands in next-generation catalytic applications. As a leading CDMO partner, we possess the technical expertise to translate the methodologies described in CN109608381B from laboratory curiosity to industrial reality. Our facilities are equipped to handle complex organometallic couplings and aggressive oxidations safely and efficiently. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. Our rigorous QC labs enforce stringent purity specifications, guaranteeing that every batch of NHPI-functionalized ligand meets the exacting standards required for high-performance MOF synthesis.
We invite you to collaborate with us to optimize your catalytic processes and reduce your overall manufacturing footprint. By leveraging our process development capabilities, you can achieve a Customized Cost-Saving Analysis tailored to your specific production volumes. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you secure a competitive advantage through superior chemical innovation and reliable supply chain execution.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
