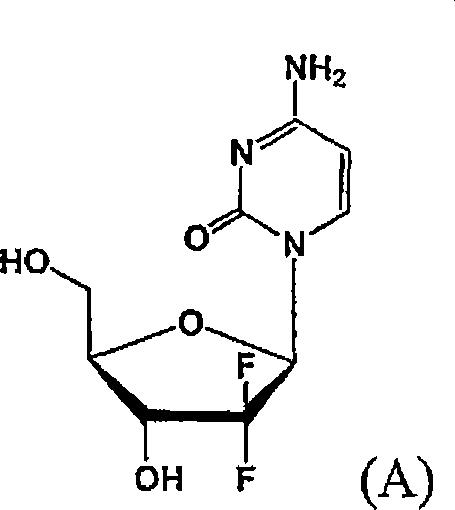
Scalable Synthesis of Solid-State Gemcitabine Intermediates via Novel Phosphate Strategy
Scalable Synthesis of Solid-State Gemcitabine Intermediates via Novel Phosphate Strategy
The pharmaceutical industry's demand for high-purity oncology therapeutics continues to drive innovation in synthetic methodology, particularly for complex nucleoside analogues like Gemcitabine. Patent CN1989148A introduces a transformative approach to synthesizing 1-alpha-halo-2,2-difluoro-2-deoxy-D-ribofuranose derivatives, which serve as critical precursors in the large-scale manufacturing of this vital anticancer agent. Unlike traditional routes that struggle with oily intermediates and poor stereocontrol, this invention leverages a unique biphenylcarbonyl protecting group strategy to produce solid-state intermediates amenable to simple recrystallization. This technological leap addresses long-standing bottlenecks in API production, offering a pathway to significantly enhanced purity profiles and operational efficiency for global supply chains.
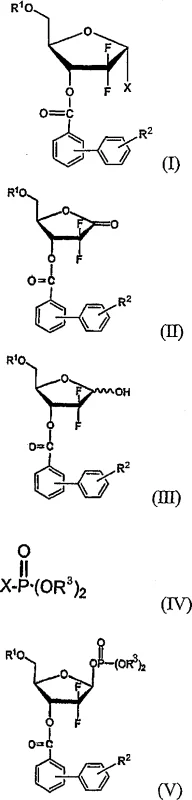
The core innovation lies in the structural modification of the ribofuranose backbone, specifically the introduction of a biphenylcarbonyl moiety at the 3-hydroxy position. This bulky aromatic group not only stabilizes the molecule into a crystalline solid but also exerts a profound influence on the stereoelectronic environment of the anomeric center. By shifting the physical state of the intermediate from an intractable oil to a manageable solid, the process eliminates the dependency on preparative column chromatography, a unit operation that is notoriously difficult to scale and economically burdensome in GMP environments. This fundamental change in physical properties translates directly into robust process reliability and cost predictability for commercial manufacturers.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Gemcitabine has relied on activated ribofuranose intermediates where the leaving group at the C-1 position is introduced via acetylation or sulfonation. As illustrated in traditional reaction schemes, these methods often suffer from inadequate stereoselectivity, yielding mixtures of alpha and beta anomers that require rigorous separation. The resulting 1-alpha-halo-furanoses are frequently isolated as oils, which complicates handling, storage, and subsequent glycosylation steps. Furthermore, the purification of these oily mixtures invariably demands column chromatography, a technique that consumes vast quantities of silica gel and solvents while generating significant hazardous waste. The overall yields for these conventional pathways hover around 45%, reflecting substantial material loss during purification and the inability to effectively suppress the formation of the undesired beta-anomer.
The Novel Approach
The methodology disclosed in CN1989148A circumvents these issues by employing a phosphate activation strategy coupled with biphenylcarbonyl protection. This approach generates a beta-enriched phosphate intermediate (Formula V) which serves as a superior precursor for the final halogenation step. The steric bulk of the biphenyl group facilitates the crystallization of the final alpha-halo product, enabling purification via recrystallization rather than chromatography. This shift allows for the isolation of the target alpha-anomer with exceptional purity levels exceeding 99.5%. Moreover, the overall process yield is boosted to a range of 65% to 75%, representing a dramatic improvement in atom economy and resource utilization. The ability to obtain a solid product simplifies logistics and quality control, making it an ideal candidate for multi-ton scale production.
Mechanistic Insights into Stereoselective Phosphate Activation
The success of this synthetic route hinges on the precise stereochemical control exerted during the phosphorylation and subsequent halogenation steps. The process begins with the reduction of a 1-oxaribose ketone to a lactol mixture, which is then subjected to phosphorylation using a reagent such as diphenyl chlorophosphate. Under basic conditions, this reaction proceeds with high stereoselectivity to favor the formation of the beta-phosphate anomer, achieving a beta-to-alpha ratio greater than 10. This selectivity is crucial because the subsequent nucleophilic substitution with a halide source proceeds via an SN2-like mechanism, resulting in inversion of configuration at the anomeric center. Consequently, the beta-phosphate is cleanly converted into the desired alpha-halo furanose.
Impurity control is inherently built into this mechanism through the physical properties of the intermediates. The biphenylcarbonyl protecting group increases the molecular weight and planarity of the scaffold, promoting strong intermolecular pi-stacking interactions that drive crystallization. This means that minor impurities, including the unwanted beta-halo anomer or residual phosphate species, remain in the mother liquor during recrystallization. The patent data indicates that a single recrystallization step is sufficient to upgrade the crude product from a mixture with an alpha/beta ratio of roughly 10:1 to a highly pure solid containing over 99.5% of the alpha-anomer. This level of purity is essential for the subsequent glycosylation with cytosine, ensuring that the final Gemcitabine API meets stringent regulatory specifications for stereochemical integrity.
How to Synthesize 1-alpha-halo-2,2-difluoro-2-deoxy-D-ribofuranose Efficiently
The synthesis protocol outlined in the patent provides a clear, three-step sequence that transforms a protected 1-oxaribose starting material into the high-value halogenated intermediate. The process is designed to be telescoped where possible, minimizing isolation steps and solvent exchanges. The initial reduction requires careful temperature control using lithium tri-tert-butoxyaluminum hydride to prevent over-reduction or side reactions. Following this, the phosphorylation step utilizes common reagents like triethylamine and diphenyl chlorophosphate in non-polar solvents such as toluene, which are easily removed. The final halogenation employs acidic conditions, such as HBr in acetic acid, to displace the phosphate group. For a detailed breakdown of the specific reaction conditions, stoichiometry, and workup procedures, please refer to the standardized synthesis guide below.
- Reduce the 1-oxaribose compound (Formula II) using lithium tri-tert-butoxyaluminum hydride to obtain the lactol compound (Formula III).
- React the lactol compound with a halophosphate (e.g., diphenyl chlorophosphate) in the presence of a base to form the beta-enriched phosphate intermediate (Formula V).
- Treat the phosphate intermediate with a halide source (e.g., HBr/acetic acid) followed by recrystallization to isolate the high-purity alpha-halo derivative (Formula I).
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition from an oil-based, chromatography-dependent process to a solid-state, recrystallization-based workflow offers profound economic and operational benefits. The elimination of column chromatography removes a major bottleneck in production throughput, significantly reducing the consumption of silica gel and organic solvents. This not only lowers direct material costs but also drastically reduces the volume of hazardous waste requiring disposal, aligning with modern environmental compliance standards. The higher overall yield further amplifies these savings by maximizing the output from each batch of starting materials, effectively lowering the cost per kilogram of the active intermediate.
- Cost Reduction in Manufacturing: The shift to recrystallization as the primary purification method eliminates the need for expensive chromatographic resins and the extensive labor associated with column packing and fraction collection. This simplification of the downstream processing stage leads to substantial cost savings in both operational expenditure and capital equipment requirements. Additionally, the improved yield means less raw material is wasted, optimizing the cost structure of the entire synthesis campaign.
- Enhanced Supply Chain Reliability: Solid intermediates are inherently more stable and easier to handle than their oily counterparts, reducing the risk of degradation during storage and transport. This physical robustness ensures consistent quality over time and simplifies inventory management. The reliance on commodity reagents like diphenyl chlorophosphate and standard halide sources further secures the supply chain against volatility, as these materials are widely available from multiple global vendors.
- Scalability and Environmental Compliance: Recrystallization is a well-understood unit operation that scales linearly from laboratory to plant scale without the engineering challenges associated with large-scale chromatography. This facilitates rapid technology transfer and capacity expansion to meet market demand. Furthermore, the reduction in solvent usage and waste generation supports sustainability goals, making the process more attractive for regulatory approval and long-term manufacturing licenses.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These insights are derived directly from the experimental data and comparative examples provided in the patent documentation, highlighting the practical advantages over legacy methods. Understanding these nuances is critical for technical teams evaluating the feasibility of adopting this new chemistry for commercial production.
Q: Why is the solid state of the 1-alpha-halo-furanose intermediate critical for manufacturing?
A: Conventional intermediates are often oily, requiring difficult and expensive column chromatography for purification. The novel biphenylcarbonyl-protected derivative described in CN1989148A is a solid, allowing for simple, cost-effective recrystallization to achieve >99.5% purity.
Q: How does the phosphate intermediate improve stereoselectivity compared to traditional methods?
A: The process utilizes a beta-enriched phosphate intermediate (Formula V) with a beta/alpha ratio greater than 10. Upon reaction with a halide source, this undergoes inversion to yield the desired alpha-halo anomer with an alpha/beta ratio of at least 10, significantly outperforming traditional acetate or sulfonate routes.
Q: What is the overall yield advantage of this new process?
A: The patented process achieves an overall yield of 65% to 75%, which is substantially higher than the approximately 45% yield typically obtained via conventional methods, primarily due to the elimination of losses associated with chromatographic purification.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1-alpha-halo-2,2-difluoro-2-deoxy-D-ribofuranose Supplier
NINGBO INNO PHARMCHEM stands at the forefront of fine chemical manufacturing, possessing the technical expertise to translate complex patent methodologies like CN1989148A into robust commercial processes. Our R&D team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab bench to reactor train is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to guarantee that every batch of 1-alpha-halo-2,2-difluoro-2-deoxy-D-ribofuranose meets the exacting standards required for oncology API synthesis.
We invite potential partners to engage with our technical procurement team to discuss how this advanced synthesis route can optimize your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic impact of switching to this solid-state intermediate. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your production volume requirements, ensuring a secure and cost-effective supply of this critical pharmaceutical building block.
