Advanced Manufacturing of 2-Amino-5-Methylpyrazine: A Breakthrough in Safe Industrial Scale-Up
Advanced Manufacturing of 2-Amino-5-Methylpyrazine: A Breakthrough in Safe Industrial Scale-Up
The chemical landscape for heterocyclic intermediates is constantly evolving, driven by the dual needs for enhanced safety protocols and economic efficiency in large-scale manufacturing. A pivotal development in this sector is documented in patent CN111925333A, filed in late 2020, which introduces a robust and innovative preparation method for 2-amino-5-methylpyrazine. This compound serves as a critical building block in the synthesis of various pharmaceutical agents and fine chemicals, yet its historical production has been plagued by hazardous reagents and complex purification challenges. The disclosed technology offers a transformative approach, utilizing a multi-step sequence that begins with the condensation of 2-aminomalonamide and methylglyoxal. By systematically addressing the limitations of prior art, this methodology not only ensures high purity but also aligns perfectly with modern green chemistry principles, making it an ideal candidate for reliable pharmaceutical intermediate suppliers seeking to optimize their supply chains.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-amino-5-methylpyrazine has relied on pathways that present significant operational risks and logistical bottlenecks for procurement managers. One prevalent method involves the use of 5-methylpyrazine formic acid as a starting material, which undergoes reaction with oxalyl chloride followed by a Curtius rearrangement using sodium azide. The reliance on sodium azide is a major red flag for safety officers, as it introduces the risk of explosive hazards and requires specialized handling equipment that drives up capital expenditure. Furthermore, alternative routes utilizing 2-methylpyrazine often suffer from poor regioselectivity during chlorination, leading to difficult-to-separate isomers that compromise the overall yield. These conventional processes frequently necessitate high-pressure reactors for ammonia ammonolysis, imposing strict equipment requirements that limit the number of qualified manufacturers capable of executing the synthesis safely and efficiently.
The Novel Approach
In stark contrast, the methodology outlined in the patent data presents a streamlined and inherently safer alternative that bypasses these dangerous reagents entirely. The new route initiates with the condensation of widely available and inexpensive precursors, 2-aminomalonamide and methylglyoxal, to construct the pyrazine ring system directly. This strategic shift eliminates the need for hazardous azide chemistry and avoids the regioselectivity issues associated with direct chlorination of methylpyrazine. Instead, the process employs a controlled sequence involving phosphorylation, catalytic hydrogenation, and a final Hofmann degradation. This approach not only simplifies the operational workflow but also enhances the economic viability of the production line by utilizing standard reactor configurations. For supply chain heads, this translates to a more resilient sourcing strategy, as the dependency on exotic or dangerous raw materials is drastically reduced, ensuring continuity of supply even in volatile market conditions.
Mechanistic Insights into the Condensation and Degradation Pathway
The core of this synthetic innovation lies in the precise control of reaction conditions during the initial ring formation and the subsequent functional group transformations. The process begins with the reaction between 2-aminomalonamide and methylglyoxal, where the mass ratio is carefully optimized, typically around 5-6:4-5, to maximize the formation of 2-methyl-5-hydroxy-4-pyrazinamide. This condensation is conducted at low temperatures, specifically between 4°C and 6°C, under alkaline conditions to facilitate cyclization while minimizing side reactions. Following isolation, the hydroxyl group is activated using phosphorus oxychloride in the presence of N,N-dimethylaniline and xylene at elevated temperatures of 120°C to 130°C. This step is crucial as it simultaneously introduces a chlorine atom and converts the amide to a nitrile, generating 2-methyl-5-chloro-4-cyanopyrazine with high efficiency. The subsequent hydrogenation step utilizes a palladium-carbon catalyst to selectively remove the chlorine atom without reducing the nitrile group, demonstrating excellent chemoselectivity that is vital for maintaining product integrity.
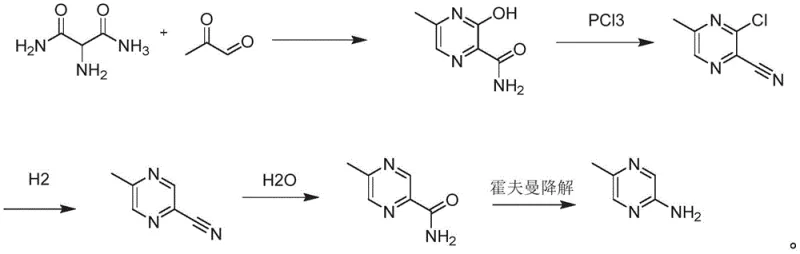
Following the hydrogenation, the nitrile group undergoes hydrolysis under alkaline conditions with hydrogen peroxide to yield the corresponding amide, 2-methyl-4-amide pyrazine. The final and perhaps most elegant step is the Hofmann degradation, where the amide is treated with bromine in an alkaline solution. This reaction proceeds through an isocyanate intermediate, which rearranges to lose a carbon atom and form the primary amine, 2-amino-5-methylpyrazine. The patent specifies a careful temperature gradient during this degradation, starting near 0°C and gradually heating to 70°C, which is essential for controlling the exothermic nature of the bromination and ensuring complete conversion. This mechanistic pathway offers R&D directors a clear advantage: the impurity profile is well-defined and manageable, as each step produces distinct intermediates that can be purified if necessary, although the high yields reported suggest that extensive purification may not always be required. The avoidance of transition metal catalysts in the final steps further simplifies the removal of heavy metal residues, a critical parameter for pharmaceutical grade intermediates.
How to Synthesize 2-Amino-5-Methylpyrazine Efficiently
Implementing this synthesis requires a disciplined approach to reaction parameters, particularly regarding temperature control and reagent addition rates. The patent data provides a comprehensive framework for executing the five-stage process, emphasizing the importance of maintaining specific pH levels and thermal profiles to achieve the reported high yields. For technical teams looking to replicate or scale this process, the key lies in the precise management of the exothermic condensation and the subsequent high-temperature chlorination. The following guide outlines the standardized operational sequence derived from the patent examples, serving as a foundational reference for process engineers aiming to establish this capability in a GMP environment.
- Condense 2-aminomalonamide with methylglyoxal under alkaline conditions at low temperature to form the pyrazinamide core.
- Activate the hydroxyl group using phosphorus oxychloride to generate the chloro-cyano intermediate.
- Perform catalytic hydrogenation to remove chlorine, followed by hydrolysis and final Hofmann degradation to yield the amine.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this novel synthesis route offers profound benefits that extend beyond mere technical feasibility, directly impacting the bottom line and operational reliability of chemical manufacturing enterprises. The elimination of sodium azide and high-pressure ammonia systems removes significant safety liabilities, which in turn lowers insurance costs and reduces the regulatory burden associated with handling explosive or toxic substances. This shift allows manufacturers to operate with greater flexibility and reduced downtime, as the safety protocols required are less stringent than those for traditional azide chemistry. Furthermore, the use of commodity chemicals like methylglyoxal and 2-aminomalonamide ensures that raw material sourcing is stable and cost-effective, shielding the supply chain from the volatility often seen with specialized starting materials. This stability is crucial for long-term contracting and capacity planning, providing procurement managers with the confidence to secure supply agreements without fear of sudden raw material shortages.
- Cost Reduction in Manufacturing: The economic advantages of this process are driven primarily by the simplification of the reaction sequence and the use of inexpensive, readily available feedstocks. By avoiding the multi-step protection and deprotection strategies often required in older methodologies, the overall material throughput is improved, leading to substantial cost savings in raw material consumption. Additionally, the high yields achieved at each stage minimize waste generation and reduce the load on downstream purification units, further driving down the cost of goods sold. The removal of expensive noble metal catalysts from the final steps, relying instead on recoverable palladium-carbon in the middle stage, optimizes catalyst usage and recovery costs. These factors combine to create a highly competitive cost structure that allows suppliers to offer aggressive pricing while maintaining healthy margins.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route significantly enhances supply chain resilience by decoupling production from scarce or geographically concentrated raw materials. Since the key precursors are bulk chemicals produced by multiple vendors globally, the risk of supply disruption is minimized. The process itself is designed for scalability, utilizing standard unit operations such as filtration, distillation, and crystallization that are common in most fine chemical facilities. This universality means that production can be easily transferred between sites or scaled up to meet surging demand without requiring bespoke engineering solutions. For supply chain heads, this translates to shorter lead times and a more predictable delivery schedule, ensuring that downstream pharmaceutical production lines remain uninterrupted.
- Scalability and Environmental Compliance: Environmental sustainability is increasingly a prerequisite for doing business, and this method aligns well with green chemistry initiatives by reducing the generation of hazardous waste. The absence of azide waste streams simplifies effluent treatment and lowers the environmental compliance costs associated with wastewater disposal. The process is inherently scalable, as demonstrated by the patent examples which utilize standard laboratory glassware that translates linearly to industrial reactor sizes. The ability to run the reaction at atmospheric pressure for most steps, except for the controlled hydrogenation, reduces the energy footprint and equipment complexity. This ease of scale-up ensures that manufacturers can rapidly respond to market demands, moving from pilot batches to multi-ton production runs with minimal technical friction.
Frequently Asked Questions (FAQ)
To assist technical and commercial stakeholders in evaluating this technology, we have compiled a set of frequently asked questions based on the specific technical disclosures and beneficial effects detailed in the patent documentation. These answers address common concerns regarding safety, raw material availability, and the feasibility of industrial implementation. Understanding these nuances is essential for making informed decisions about integrating this intermediate into your supply portfolio.
Q: Why is this new synthesis route safer than traditional methods?
A: Traditional routes often rely on hazardous sodium azide for Curtius rearrangement or high-pressure ammonia ammonolysis. This novel method eliminates explosive azides and uses standard hydrogenation and degradation techniques, significantly improving operational safety.
Q: What are the key raw materials for this process?
A: The process utilizes readily available and cost-effective starting materials, specifically 2-aminomalonamide and methylglyoxal, avoiding the need for difficult-to-source precursors like 5-methyl-2-pyrazinecarboxylic acid.
Q: Is this method suitable for large-scale commercial production?
A: Yes, the patent explicitly highlights the method's suitability for industrial scale-up due to simple operations, high yields across multiple steps, and the use of standard reactor equipment without extreme pressure requirements.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Amino-5-Methylpyrazine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of pharmaceutical development and commercial manufacturing. Our team of expert chemists has thoroughly analyzed the technological potential of the route described in CN111925333A and is fully prepared to leverage this knowledge for our clients. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that whether you require material for clinical trials or full-scale commercialization, our capacity is ready to meet your needs. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that employ state-of-the-art analytical techniques to verify every batch, guaranteeing that the 2-amino-5-methylpyrazine supplied meets the highest industry standards.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis route can benefit your specific projects. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your volume requirements, helping you optimize your budget without compromising on quality. We encourage you to reach out today to request specific COA data and route feasibility assessments, allowing us to demonstrate our capability to deliver this valuable intermediate with the reliability and efficiency your supply chain demands.
