Advanced Synthesis of Alpha-Beta Deuterated Amines for Pharmaceutical Manufacturing
The pharmaceutical industry is increasingly recognizing the strategic value of deuterated drugs, which offer improved metabolic stability and pharmacokinetic profiles compared to their non-deuterated counterparts. Patent CN112430187B introduces a groundbreaking methodology for the synthesis of alpha,beta-deuterated amine compounds, addressing critical bottlenecks in current manufacturing capabilities. This innovation provides a robust pathway for producing high-purity pharmaceutical intermediates through a novel reductive deuteration of nitrile organic substances. The core of this technology lies in a two-step sequence: first, the conversion of nitrile compounds into alpha-deuterated nitrile intermediates using a base and a deuterium donor, followed by a highly selective reduction using divalent lanthanide transition metal compounds. This approach not only solves the limitations of existing synthesis methods regarding cost and deuteration rates but also opens new avenues for the efficient production of complex deuterated active pharmaceutical ingredients (APIs).
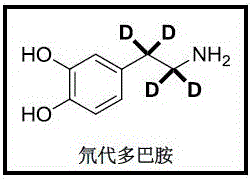
Historically, the synthesis of alpha,beta-deuterated amines has been plagued by significant technical and economic challenges. Conventional methods, such as the Ruthenium-catalyzed H/D exchange reported by Beller et al. (J. Am. Chem. Soc. 2012, 134, 12239), rely on expensive and toxic transition metal catalysts that are sensitive to air and moisture. These traditional processes often necessitate harsh reaction conditions, including high temperatures and pressures, leading to prolonged reaction times and suboptimal yields. Furthermore, the regioselectivity of H/D exchange is frequently poor, resulting in mixtures of isotopologues that require complex and costly purification steps. In contrast, the novel approach detailed in the patent utilizes a nitrile reduction strategy that operates under mild conditions, typically at room temperature, with short reaction times ranging from 15 minutes to 72 hours depending on the substrate. This shift from noble metal catalysis to lanthanide-mediated reduction represents a paradigm shift in deuterated chemical manufacturing, offering superior control over isotopic placement.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
The reliance on traditional transition metal catalysts for deuteration presents a multifaceted problem for large-scale pharmaceutical manufacturing. Catalysts based on metals like Ruthenium are not only prohibitively expensive but also pose significant environmental and safety hazards due to their toxicity and potential for residual contamination in the final drug product. The requirement for high-pressure hydrogenation or elevated temperatures increases the energy footprint of the process and demands specialized reactor infrastructure, which limits scalability. Additionally, the lack of precise regioselectivity in conventional H/D exchange means that deuterium atoms may incorporate at unintended positions on the molecular scaffold, compromising the therapeutic efficacy and metabolic profile of the deuterated drug. These inefficiencies translate directly into higher production costs and longer lead times, creating a barrier to the widespread adoption of deuterated therapeutics in the global market.
The Novel Approach
The methodology disclosed in CN112430187B overcomes these hurdles by employing a chemoselective reduction of nitriles using divalent lanthanide compounds. This process begins with the generation of an alpha-deuterated nitrile intermediate, which is subsequently reduced to the corresponding amine with high fidelity. The use of reagents such as Samarium Diiodide (SmI2) allows for the reaction to proceed at ambient temperatures, eliminating the need for energy-intensive heating or pressurization. Experimental data from the patent demonstrates exceptional performance, with yields reaching as high as 97% and deuteration rates exceeding 98% at specific sites. For instance, in the synthesis of compound 5d, a yield of 97% was achieved with alpha and beta deuteration rates of 95% and 98% respectively. This level of precision ensures that the final product meets the stringent purity specifications required for clinical applications, while the mild conditions facilitate a safer and more sustainable manufacturing environment.
Mechanistic Insights into SmI2-Catalyzed Reductive Deuteration
The mechanistic elegance of this synthesis lies in the unique reactivity of divalent lanthanide transition metal compounds, particularly Samarium Diiodide (SmI2), which acts as a potent single-electron transfer (SET) agent. In the second step of the reaction, the SmI2 reduces the carbon-nitrogen triple bond of the alpha-deuterated nitrile intermediate. This reduction proceeds through a radical anion species that is subsequently protonated (or rather, deuterated) by the deuterium source present in the system. The presence of a base in the initial step facilitates the rapid H/D exchange at the acidic alpha-proton of the nitrile, ensuring that the intermediate entering the reduction phase is already enriched with deuterium. The subsequent reduction of the nitrile group to an amine incorporates additional deuterium atoms at the beta-position, resulting in the characteristic alpha,beta-dideuterated motif. This cascade of events is highly efficient because the lanthanide catalyst coordinates specifically with the nitrile functionality, preventing side reactions with other sensitive functional groups that might be present on the aromatic ring or aliphatic chain.
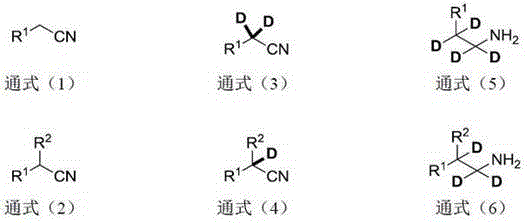
Impurity control is inherently built into this mechanism due to the high chemoselectivity of the lanthanide reagents. Unlike broad-spectrum hydrogenation catalysts that might reduce aromatic rings or other unsaturated bonds, SmI2 and related lanthanide halides preferentially target the nitrile group under the specified conditions. The patent data indicates that by-products are minimal, and the primary impurities arise from incomplete deuteration rather than structural degradation. This is evidenced by the high deuteration rates observed across a wide range of substrates, from simple alkyl nitriles to complex heterocyclic systems like benzothiophene and indole derivatives. The ability to quench the reaction simply by introducing air further simplifies the workup, as the oxidized lanthanide species can be easily separated during the aqueous extraction phase. This streamlined purification process minimizes the risk of introducing new impurities during isolation, ensuring a clean impurity profile that is critical for regulatory approval of deuterated drug substances.
How to Synthesize Alpha-Beta Deuterated Amines Efficiently
The operational simplicity of this protocol makes it accessible for both laboratory research and industrial production. The process generally involves protecting the reactor with argon to prevent oxidation of the sensitive lanthanide reagents, followed by the sequential addition of base, deuterium donor, and the nitrile substrate. After the initial exchange period, the lanthanide solution is introduced, and the mixture is stirred for a short duration before quenching. Detailed standardized synthesis steps for specific compounds are provided in the technical guide below, ensuring reproducibility and consistency across different batches.
- Prepare the reactor under argon protection and add a base (e.g., triethylamine) and a deuterium donor reagent (e.g., heavy water).
- Add the nitrile compound dissolved in an organic solvent and stir at room temperature for 24 to 72 hours to form the alpha-deuterated nitrile intermediate.
- Introduce a divalent lanthanide transition metal compound solution (e.g., SmI2 in THF), stir for 15 to 60 minutes, quench with air, and purify to obtain the target alpha,beta-deuterated amine.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers tangible benefits in terms of cost structure and supply reliability. The shift away from precious metal catalysts to abundant lanthanides fundamentally alters the cost dynamics of deuterated intermediate production. By eliminating the need for expensive Ruthenium or Rhodium complexes, manufacturers can achieve significant cost reduction in deuterated drug manufacturing without compromising on quality. Furthermore, the mild reaction conditions reduce energy consumption and extend the lifespan of reactor equipment, contributing to lower overheads. The robustness of the method also means that production schedules are less likely to be disrupted by equipment failures or safety incidents associated with high-pressure operations.
- Cost Reduction in Manufacturing: The replacement of noble metal catalysts with divalent lanthanide compounds like SmI2 drastically reduces the raw material costs associated with catalysis. Since these lanthanide reagents are more abundant and easier to handle, the overall cost of goods sold (COGS) for the deuterated intermediate is lowered. Additionally, the high yields and selectivity minimize the loss of expensive deuterated starting materials, ensuring that every gram of deuterium source is utilized effectively. The simplified workup procedure, which avoids complex chromatographic separations in many cases, further reduces solvent usage and labor costs, leading to substantial economic efficiency.
- Enhanced Supply Chain Reliability: The reliance on readily available nitrile precursors and common organic solvents ensures a stable supply chain that is less vulnerable to geopolitical fluctuations affecting rare metal markets. The operational simplicity of the process, which does not require specialized high-pressure autoclaves, allows for greater flexibility in manufacturing site selection. This decentralization capability enhances supply continuity, as production can be scaled across multiple facilities without significant capital investment in specialized infrastructure. The short reaction times also improve throughput, allowing suppliers to respond more rapidly to fluctuating market demands.
- Scalability and Environmental Compliance: The environmental profile of this method is superior to traditional hydrogenation techniques, as it generates fewer toxic by-products and avoids the use of high-pressure hydrogen gas. The ability to quench the reaction with air and perform simple aqueous extractions simplifies waste management and reduces the burden on effluent treatment plants. This alignment with green chemistry principles facilitates easier regulatory compliance and supports corporate sustainability goals. The process has been demonstrated to be scalable, with potential for commercial scale-up of complex pharmaceutical intermediates from kilogram to multi-ton quantities while maintaining high purity standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this deuteration technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing a clear understanding of the method's capabilities and limitations for potential partners.
Q: How does this method improve regioselectivity compared to traditional H/D exchange?
A: Unlike traditional transition metal-catalyzed H/D exchange which often lacks selectivity, this nitrile reduction method specifically targets the alpha-position of the cyano group and the carbon-nitrogen triple bond, ensuring high deuteration rates at the desired alpha and beta positions without affecting other parts of the molecule.
Q: What are the cost advantages of using lanthanide catalysts over noble metals?
A: This process utilizes divalent lanthanide compounds like Samarium Diiodide (SmI2) instead of expensive and toxic noble metals like Ruthenium or Rhodium. This substitution significantly lowers raw material costs and simplifies the removal of metal residues, reducing downstream purification expenses.
Q: Is this synthesis method scalable for industrial production?
A: Yes, the method operates under mild conditions (mostly room temperature and atmospheric pressure) and uses simple workup procedures like air quenching and solvent extraction. These factors make it highly suitable for commercial scale-up from kilogram to tonnage production without requiring specialized high-pressure equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alpha-Beta Deuterated Amines Supplier
At NINGBO INNO PHARMCHEM, we understand the critical role that high-quality intermediates play in the development of next-generation deuterated therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to market availability is seamless. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that utilize advanced analytical techniques to verify isotopic enrichment and chemical purity. Our expertise in handling sensitive organometallic reactions allows us to optimize this lanthanide-catalyzed process for maximum efficiency and safety on an industrial scale.
We invite you to collaborate with us to leverage this innovative technology for your drug development pipeline. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific project needs. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can accelerate your timeline and reduce your overall development costs. Let us be your partner in bringing high-value deuterated medicines to patients worldwide.
