Advanced Synthesis of D9-Clenbuterol Hydrochloride for High-Precision Analytical Standards
Advanced Synthesis of D9-Clenbuterol Hydrochloride for High-Precision Analytical Standards
The global demand for ultra-sensitive detection methods in food safety and pharmacokinetics has necessitated the development of robust synthetic routes for isotopically labeled internal standards. Patent CN109096126B introduces a groundbreaking methodology for the synthesis of Deuterium-labeled D9-Clenbuterol Hydrochloride, a critical reference standard used in GC-MS and LC-MS analysis to detect beta-agonist residues in animal-derived food products. This technology addresses the longstanding challenges of low yield and high cost associated with deuterated reagents by optimizing the reaction pathway to operate under mild conditions. By shifting from harsh thermal conditions to a room-temperature protocol, the process not only enhances the stability of key intermediates but also dramatically improves the atom economy of the expensive deuterated amine. For R&D directors and procurement specialists seeking a reliable API intermediate supplier, understanding the nuances of this improved synthesis is essential for securing a stable supply chain of high-purity analytical standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of deuterium-labeled clenbuterol has been plagued by inefficient utilization of costly isotopic raw materials and unstable reaction profiles. Prior art methodologies typically relied on heating reflux conditions to drive the nucleophilic substitution between the bromo-acetophenone precursor and D9-tert-butylamine. This thermal stress often led to the decomposition of the sensitive 4-amino-alpha-D9-tert-butylamino-3,5-dichloroacetophenone intermediate, resulting in poor reproducibility and significant side reactions. Furthermore, the conventional processes exhibited a dismal total conversion rate of the core D9-tert-butylamine reagent, often hovering around a mere 4.2%. Such inefficiency translates to exorbitant production costs, as over 95% of the expensive deuterated amine is wasted as byproduct salts or unreacted material, making the final product economically prohibitive for routine quality control laboratories. Additionally, the requirement for high temperatures complicates the scale-up process, introducing safety hazards and energy burdens that are unsustainable for modern green chemistry initiatives.
The Novel Approach
The innovative process disclosed in CN109096126B fundamentally reengineers the synthetic strategy to overcome these thermodynamic and kinetic barriers. By utilizing 3,5-dichloro-4-aminoacetophenone as the starting scaffold, the new route employs a strategic bromination followed by a room-temperature amination step that preserves the integrity of the deuterated label. The most significant breakthrough lies in the optimization of the amination conditions, where the reaction is conducted in a sealed container at 20-30°C, eliminating the need for thermal activation that previously degraded the product. This mild approach stabilizes the intermediate, allowing for a much cleaner reaction profile and facilitating easier downstream purification. Consequently, the total conversion rate of the valuable D9-tert-butylamine is elevated from 4.2% to an impressive 40%, representing a nearly tenfold improvement in raw material efficiency. This leap in efficiency is pivotal for cost reduction in pharmaceutical manufacturing, particularly for isotopic standards where reagent costs dominate the bill of materials.
Mechanistic Insights into the Three-Step Synthetic Route
The synthesis proceeds through a logical three-step sequence designed to maximize yield while minimizing isotopic dilution. The first stage involves the alpha-bromination of the acetophenone derivative. As illustrated in the reaction scheme below, 3,5-dichloro-4-aminoacetophenone reacts with a brominating agent, such as liquid bromine or copper bromide, to introduce the leaving group necessary for subsequent nucleophilic attack. The patent specifies that this can be achieved with high selectivity, yielding the 3,5-dichloro-4-amino-alpha-bromoacetophenone intermediate in approximately 62% yield. The choice of solvent, ranging from ethyl acetate to chloroform, plays a crucial role in solubilizing the reactants and managing the exotherm during bromine addition.
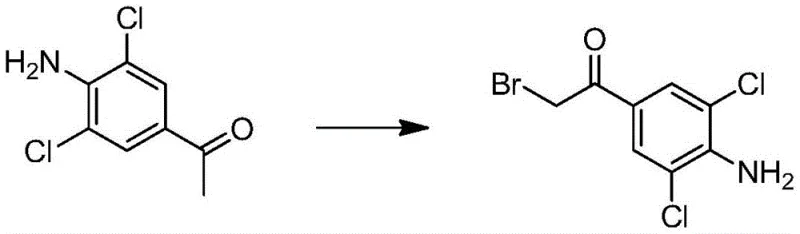
Following bromination, the critical deuterium incorporation occurs via nucleophilic substitution. The alpha-bromo intermediate reacts with D9-tert-butylamine in a polar aprotic solvent like tetrahydrofuran or acetonitrile. Unlike previous methods that required heat, this reaction proceeds efficiently at room temperature over 4 to 10 hours. The mechanism involves the displacement of the bromide ion by the deuterated amine, forming the stable ketone intermediate shown in the following figure. The use of a sealed vessel prevents the loss of volatile amine and maintains the stoichiometry required for high conversion. Following the reaction, the mixture is filtered to remove the hydrobromide salt byproduct, and the filtrate is acidified to isolate the stable isotope-labeled ketone hydrochloride salt with yields reaching up to 96%.
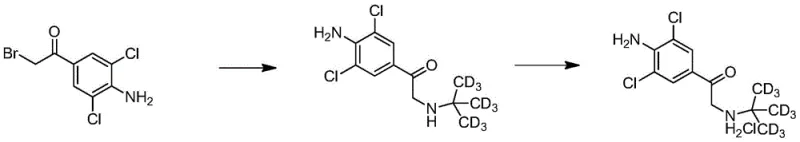
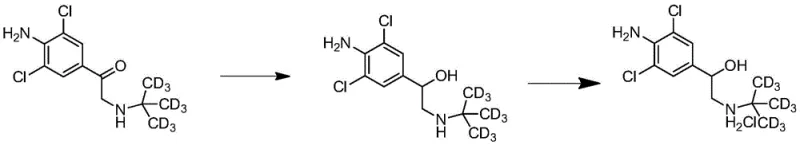
The final transformation involves the stereoselective reduction of the ketone carbonyl group to the corresponding benzylic alcohol. The isolated ketone hydrochloride is dissolved and neutralized to a pH of roughly 7 before the addition of a reducing agent such as sodium borohydride. Controlling the internal temperature below 5°C during the addition of the hydride source is critical to prevent over-reduction or side reactions. As depicted in the final reaction scheme, the hydride attacks the carbonyl carbon, generating the chiral alcohol center. Subsequent workup involves adjusting the pH to alkaline conditions to free the base, extracting into an organic phase, and finally converting it back to the hydrochloride salt for isolation. This final step delivers the target D9-Clenbuterol Hydrochloride with a purity of 98.98% and an overall yield of roughly 64% for this specific step, ensuring the final product meets the rigorous specifications required for mass spectrometry internal standards.
How to Synthesize D9-Clenbuterol Hydrochloride Efficiently
Implementing this synthesis requires precise control over reaction parameters, particularly regarding temperature and pH management during the reduction phase. The protocol outlined in the patent provides a scalable framework that avoids the use of exotic catalysts, relying instead on commodity chemicals like sodium borohydride and copper bromide. The detailed operational procedure emphasizes the importance of thorough washing and recrystallization steps to remove residual halides and unreacted amines, which could otherwise interfere with the isotopic purity of the final standard. For process chemists looking to replicate this high-purity API intermediate, adherence to the specified molar ratios—specifically the 1:1.2 to 1:2 ratio of bromo-intermediate to deuterated amine—is vital to balance cost against conversion efficiency. The complete standardized operating procedure, including specific workup instructions and safety precautions for handling bromine and deuterated reagents, is detailed in the technical guide below.
- Perform bromination on 3,5-dichloro-4-aminoacetophenone using liquid bromine or copper bromide to obtain the alpha-bromo intermediate.
- React the bromo-intermediate with D9-tert-butylamine at room temperature in a sealed container to form the deuterated amino-ketone.
- Reduce the ketone group using sodium borohydride under controlled pH conditions, followed by acidification to isolate the final hydrochloride salt.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain perspective, the adoption of this patented methodology offers substantial strategic advantages beyond mere technical feasibility. The primary driver for value creation is the drastic improvement in the utilization of D9-tert-butylamine, a specialized and expensive reagent. By increasing the conversion efficiency from single digits to 40%, manufacturers can significantly lower the variable cost per gram of the final product. This efficiency gain mitigates the risk of supply volatility associated with deuterated chemicals, as less raw material is required to meet the same production targets. Furthermore, the elimination of high-temperature reflux conditions reduces energy consumption and lowers the thermal load on reactor systems, contributing to a more sustainable and cost-effective manufacturing footprint. These factors combined make the process highly attractive for commercial scale-up of complex isotopic intermediates.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the optimization of the most expensive input material. In traditional synthesis, the low conversion of D9-tert-butylamine meant that the majority of the reagent cost was lost to waste streams. By engineering a pathway that captures 40% of the amine into the final product, the effective cost of goods sold is drastically reduced without compromising quality. Additionally, the ability to perform the key coupling step at room temperature eliminates the need for energy-intensive heating mantles or oil baths, further reducing utility costs. The simplified purification process, which relies on filtration and crystallization rather than complex chromatography, also lowers the consumption of solvents and stationary phases, driving down operational expenditures significantly.
- Enhanced Supply Chain Reliability: Stability is a critical factor in the supply of reference standards. The conventional method's reliance on heating unstable intermediates often led to batch-to-batch variability and potential supply disruptions due to failed runs. This new approach, operating under mild conditions, ensures consistent reaction kinetics and higher reproducibility. The robustness of the room-temperature amination step means that production can be scheduled with greater certainty, reducing lead time for high-purity isotopic standards. Moreover, the use of common solvents like ethanol and ethyl acetate, rather than specialized high-boiling polar aprotic solvents that might be subject to supply constraints, ensures that the manufacturing process remains resilient against raw material shortages.
- Scalability and Environmental Compliance: Scaling isotopic syntheses often presents challenges regarding waste management, particularly when dealing with halogenated byproducts. This process generates D9-tert-butylamine hydrobromide as a solid byproduct which can be easily filtered off, simplifying the waste stream compared to liquid effluents that require complex treatment. The high purity of the crude product (98.98%) minimizes the need for extensive reprocessing, thereby reducing the overall solvent waste volume. The avoidance of heavy metal catalysts in the reduction step (using borohydrides instead) aligns with strict environmental regulations regarding metal residues in pharmaceutical intermediates. This compliance facilitates smoother regulatory filings and reduces the burden on environmental health and safety teams during plant operations.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and quality attributes of this synthesis route. Understanding these details is crucial for quality assurance teams validating the method for GMP production of reference standards. The answers are derived directly from the experimental data and beneficial effects reported in the patent documentation, ensuring accuracy and relevance for technical decision-makers.
Q: How does this patent improve the economic efficiency of D9-Clenbuterol production?
A: The patented process significantly increases the total conversion rate of the expensive D9-tert-butylamine raw material from a historical low of 4.2% to approximately 40%, drastically reducing raw material waste and overall production costs.
Q: What are the critical reaction conditions for the amination step?
A: Unlike prior art requiring heating reflux which causes instability, this method utilizes mild room temperature conditions (20-30°C) in a sealed container, ensuring the stability of the intermediate and preventing decomposition.
Q: What purity levels can be achieved with this synthesis route?
A: Through optimized purification steps including recrystallization and pH-controlled extraction, the process yields D9-Clenbuterol Hydrochloride with a purity exceeding 98.98%, meeting stringent requirements for analytical reference standards.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable D9-Clenbuterol Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality isotopic standards play in ensuring food safety and regulatory compliance globally. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art NMR and Mass Spectrometry instrumentation to verify isotopic enrichment and chemical purity. Our facility is designed to handle complex deuterated chemistry safely, adhering to international EHS standards while maintaining the flexibility to customize batch sizes according to your specific analytical requirements.
We invite procurement leaders and R&D directors to collaborate with us to optimize their supply chain for analytical standards. By leveraging our expertise in process optimization, we can offer a Customized Cost-Saving Analysis tailored to your volume needs, demonstrating how our manufacturing efficiencies translate into tangible value for your organization. Please contact our technical procurement team today to request specific COA data, route feasibility assessments, and competitive quotations for D9-Clenbuterol Hydrochloride and other specialized intermediates.
