Advanced Synthesis of Stable Isotope Labeled Chlorpropaline for Precision Pharmaceutical Analysis
Advanced Synthesis of Stable Isotope Labeled Chlorpropaline for Precision Pharmaceutical Analysis
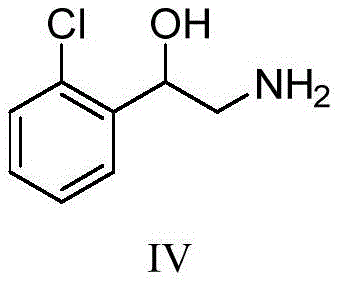
The pharmaceutical and agrochemical industries increasingly rely on precise analytical methods to ensure food safety and regulatory compliance, particularly regarding beta-2 receptor agonists like chlorpropaline. Patent CN113149851A introduces a groundbreaking preparation method for stable isotope labeled chlorpropaline, addressing the critical shortage of domestic high-quality internal standards for Isotope Dilution Mass Spectrometry (IDMS). This technology leverages a streamlined four-step synthetic pathway starting from inexpensive 2-bromo-2'-chloroacetophenone, utilizing an improved Gabriel synthesis followed by hydrolysis, reduction, and final reductive amination with labeled acetone. By shifting the isotope labeling step to the final stage of the synthesis, the process maximizes atom economy and minimizes isotopic dilution, ensuring the final product achieves an isotopic abundance exceeding 98%. This innovation not only breaks the reliance on imported standards but also provides a robust, scalable solution for producing high-purity reference materials essential for accurate trace residue detection in complex biological matrices.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chlorpropaline and its isotopically labeled analogues has been plagued by inefficient multi-step routes that suffer from low overall yields and arduous purification challenges. Prior literature, such as the methods described in Yakugaku Zasshi (1978), relied on alpha-formyl o-chloroacetophenone which required difficult separation processes and resulted in suboptimal yields due to side reactions during amination. Furthermore, existing patents like CN108911995A and related academic works often necessitated the pre-synthesis of deuterated isopropylamine through five or more reaction steps, drastically inflating production costs and extending lead times. These conventional approaches frequently encountered issues with byproduct formation during the amination process, leading to complex impurity profiles that were difficult to resolve without expensive chromatographic techniques. The cumulative effect of these inefficiencies was a high-cost supply chain heavily dependent on foreign manufacturers, creating significant vulnerabilities for domestic testing laboratories requiring reliable internal standards for mass spectrometry.
The Novel Approach
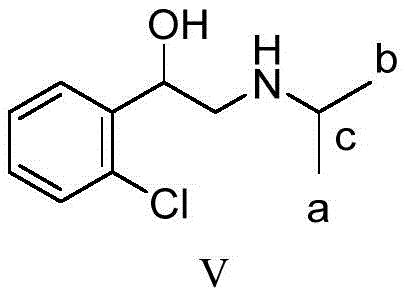
The patented methodology revolutionizes this landscape by employing a rational design that postpones the introduction of the expensive isotope label until the final synthetic step, thereby preserving isotopic integrity and maximizing yield. By utilizing 2-bromo-2'-chloroacetophenone as a readily available starting material and reacting it with sodium diformylamide, the process establishes the carbon-nitrogen bond early in a highly controlled nucleophilic substitution. This strategic sequence allows for the bulk of the molecular skeleton to be constructed using natural abundance reagents, reserving the costly deuterated acetone solely for the terminal reductive amination. The result is a concise four-step protocol that eliminates the need for pre-labeling the amine component, significantly simplifying the operational workflow. This approach not only enhances the reproducibility of the synthesis but also ensures that the isotopic abundance remains above 98% without the dilution phenomena often observed in earlier labeling strategies.
Mechanistic Insights into Improved Gabriel Synthesis and Reductive Amination
The core of this synthetic success lies in the modified Gabriel amination strategy, where sodium diformylamide acts as a masked ammonia equivalent to install the nitrogen functionality with high regioselectivity. In the initial step, the bromine atom of the acetophenone derivative is displaced by the diformylamide anion in a polar aprotic solvent like acetonitrile, forming a stable N,N-diformyl protected intermediate that prevents over-alkylation. Subsequent acidic hydrolysis under reflux conditions efficiently cleaves the formyl groups to reveal the primary amine as a hydrochloride salt, a transformation that proceeds with exceptional conversion rates exceeding 96%. The following reduction of the ketone moiety using sodium borohydride is carefully controlled to generate the chiral amino-alcohol intermediate without affecting the aromatic chloride, demonstrating excellent chemoselectivity. Finally, the reductive amination with isotope-labeled acetone introduces the deuterated isopropyl group, completing the molecular architecture while maintaining the structural integrity of the sensitive beta-hydroxy amine motif.
Impurity control is inherently built into this route through the use of crystalline intermediates and selective reaction conditions that minimize side products. The hydrolysis step, for instance, precipitates the amine salt directly from the reaction mixture, allowing for a simple filtration that removes organic soluble impurities before the reduction phase. During the final reductive amination, the use of specific reducing agents like sodium borodeuteride ensures that the deuterium label is incorporated exclusively at the desired positions on the isopropyl group, avoiding scrambling or loss of the label to the solvent. This precision is critical for IDMS applications, where the mass difference between the analyte and the internal standard must be distinct and stable. The process consistently delivers a final product with purity greater than 98%, as confirmed by ESI-MS and NMR analysis, ensuring that the reference material meets the rigorous specifications required for quantitative trace analysis in food safety monitoring.
How to Synthesize Stable Isotope Labeled Chlorpropaline Efficiently
The synthesis of this high-value analytical standard is designed for scalability and operational simplicity, making it accessible for both laboratory-scale preparation and commercial manufacturing. The process begins with the dissolution of the bromo-acetophenone starting material in acetonitrile, followed by the addition of sodium diformylamide and heating under reflux to drive the substitution to completion. Detailed standardized operating procedures for each of the four reaction stages, including specific workup protocols like pH adjustment and solvent extraction, are essential to maintain the high yield and purity profile described in the patent documentation. Operators must pay close attention to the stoichiometry of the reducing agents and the temperature control during the exothermic reduction steps to ensure safety and consistency. For a comprehensive guide on the exact molar ratios, reaction times, and purification techniques required to replicate this high-efficiency pathway, please refer to the technical execution steps outlined below.
- Perform nucleophilic substitution of 2-bromo-2'-chloroacetophenone with sodium diformylamide to generate the protected amine intermediate.
- Conduct acidic hydrolysis of the intermediate to remove formyl protecting groups and obtain the amine salt.
- Reduce the ketone group using a borohydride reducing agent to form the amino-alcohol intermediate.
- Execute reductive amination with stable isotope labeled acetone to introduce the deuterated isopropyl group.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, this patented synthesis route offers substantial economic benefits by fundamentally restructuring the cost drivers associated with isotope-labeled compound production. By eliminating the need for multi-step pre-synthesis of labeled amines, the process drastically reduces the consumption of expensive deuterated reagents, confining their use to a single, high-yield final step. This strategic material utilization translates directly into lower raw material costs per gram of finished product, allowing for more competitive pricing models for end-users in the analytical testing sector. Furthermore, the reliance on commodity chemicals like 2-bromo-2'-chloroacetophenone and common solvents ensures a stable supply chain that is less susceptible to the volatility often seen with specialized labeled building blocks. The simplified four-step sequence also reduces labor hours and utility consumption, contributing to a leaner manufacturing footprint that aligns with modern cost-reduction initiatives in fine chemical production.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts and the reduction of synthetic steps from five or more down to four significantly lowers the operational expenditure associated with this API intermediate. By avoiding complex purification sequences and expensive catalytic systems, the process achieves a streamlined workflow that minimizes waste generation and solvent usage. This efficiency gain allows manufacturers to offer high-purity isotope standards at a fraction of the cost of imported alternatives, providing immediate value to budget-conscious quality control laboratories. The high total yield of over 66% further amplifies these savings by maximizing the output from every batch of starting material processed.
- Enhanced Supply Chain Reliability: Utilizing widely available industrial feedstocks rather than niche imported precursors mitigates the risk of supply disruptions and long lead times. The robustness of the reaction conditions, which tolerate standard laboratory equipment and do not require exotic high-pressure or cryogenic setups, facilitates easier technology transfer to multiple manufacturing sites. This geographic flexibility ensures a continuous supply of critical reference materials, safeguarding national food safety monitoring programs against external market fluctuations. The ability to produce various labeled variants, such as D-labeled or 13C-labeled compounds, using the same core platform adds further resilience to the supply chain by allowing rapid adaptation to changing analytical requirements.
- Scalability and Environmental Compliance: The process is inherently scalable, having been designed with large-scale production in mind through the use of simple unit operations like filtration and crystallization. The avoidance of heavy metal catalysts simplifies wastewater treatment and reduces the environmental burden associated with hazardous waste disposal, aligning with increasingly stringent global environmental regulations. This green chemistry profile not only reduces compliance costs but also enhances the corporate sustainability credentials of the manufacturer. The high reproducibility of the method ensures that scaling from kilogram to tonne quantities can be achieved without compromising the critical isotopic abundance or chemical purity specifications.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this stable isotope labeled compound, derived directly from the patent's experimental data and background analysis. Understanding these details helps stakeholders appreciate the technical superiority and practical utility of this new manufacturing method in the context of modern analytical chemistry. The answers provided reflect the specific advantages of the four-step route over traditional methods, focusing on yield, purity, and operational feasibility. For further technical discussions or custom synthesis requests, our team is prepared to provide detailed documentation and support.
Q: Why is stable isotope labeled chlorpropaline critical for residue detection?
A: It serves as an internal standard in Isotope Dilution Mass Spectrometry (IDMS), effectively eliminating matrix effects and recovery differences caused by complex sample pretreatment in meat and urine analysis.
Q: How does this patented method improve upon conventional synthesis routes?
A: Unlike prior art requiring 5+ steps with difficult purification, this method utilizes a concise 4-step route with cheap starting materials, achieving over 66% total yield and >98% isotopic abundance.
Q: What represent the key cost advantages of this manufacturing process?
A: The process avoids expensive transition metal catalysts and complex multi-step isotope labeling of the amine precursor, significantly reducing raw material costs and operational complexity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Stable Isotope Labeled Chlorpropaline Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality internal standards play in ensuring the accuracy of residue detection in the food and pharmaceutical industries. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the demanding volume requirements of global testing agencies. We are committed to delivering products with stringent purity specifications and rigorous QC labs verification, guaranteeing that every batch of isotope-labeled chlorpropaline meets the >98% isotopic abundance and >98% chemical purity benchmarks set by the patent. Our state-of-the-art facilities are equipped to handle the specific solvent and reagent requirements of this synthesis, ensuring consistent quality and supply continuity for our partners.
We invite procurement managers and R&D directors to contact our technical procurement team to discuss how this optimized synthesis route can benefit your analytical workflows. By partnering with us, you gain access to a Customized Cost-Saving Analysis that demonstrates the tangible economic advantages of switching to our domestically produced standards. We encourage you to request specific COA data and route feasibility assessments to verify the superior performance of our materials in your specific IDMS applications. Let us help you secure a reliable, cost-effective supply of this essential reference material today.
