Advanced Synthesis of Ractopamine Hydrochloride-D6 for High-Precision Mass Spectrometry Standards
Advanced Synthesis of Ractopamine Hydrochloride-D6 for High-Precision Mass Spectrometry Standards
The global demand for ultra-sensitive detection of beta-agonists in food supply chains and athletic doping control has necessitated the development of superior internal standards for mass spectrometry. Patent CN113061094A introduces a groundbreaking preparation method for Ractopamine Hydrochloride-D6, addressing the critical limitations of existing synthetic routes. This technology leverages a strategic combination of hydrogen-deuterium (H-D) exchange and reductive amination to install six deuterium atoms efficiently. Unlike previous methods that struggled with low yields or insufficient mass differentiation, this novel approach ensures an isotopic abundance greater than 96% and chemical purity exceeding 98%. For analytical laboratories and regulatory bodies, the availability of such high-fidelity standards is paramount for enforcing strict residue limits defined by organizations like the World Anti-Doping Agency (WADA) and the European Union.
The significance of this patent extends beyond mere chemical synthesis; it represents a vital tool for public health safety. Ractopamine, while used in veterinary medicine, poses severe health risks when consumed via contaminated meat products, ranging from muscle tremors to life-threatening cardiac arrhythmias. Accurate quantification at trace levels is only possible through Isotope Dilution Mass Spectrometry (IDMS), which requires an internal standard that behaves identically to the analyte during extraction but is distinguishable by mass. The method disclosed in CN113061094A provides a robust, reproducible pathway to generate this essential reagent, utilizing cost-effective starting materials and avoiding the prohibitive expenses associated with fully labeled precursor molecules found in older literature.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art synthetic strategies for deuterated ractopamine have been plagued by significant economic and technical inefficiencies that hinder widespread adoption in quality control laboratories. For instance, earlier patents such as CN102786426A describe routes relying on the condensation of pre-labeled octopamine derivatives with deuterated raspberry ketone. This approach is fundamentally flawed for large-scale production because the starting materials themselves are exorbitantly expensive isotopically labeled compounds, driving up the cost of the final standard to unsustainable levels. Furthermore, these traditional coupling reactions often suffer from poor atom economy and complex purification requirements, resulting in overall yields that are commercially unviable for the high-volume demand of the food safety testing industry.
Another critical deficiency in legacy methods, exemplified by patent CN104387282A, is the insufficient degree of isotopic labeling. Some existing processes introduce only a single deuterium atom (1D) into the ractopamine structure. In the context of high-resolution mass spectrometry, a mass difference of merely one unit is inadequate because the natural isotopic distribution of carbon-13 in the unlabeled analyte creates background noise that overlaps with the labeled standard. This spectral interference compromises the accuracy of quantification, rendering such mono-deuterated standards useless for authoritative arbitration testing. Consequently, there has been a persistent market gap for a synthesis method that can deliver multi-deuterated analogues with high mass shifts at a fraction of the historical cost.
The Novel Approach
The methodology presented in CN113061094A revolutionizes the production landscape by decoupling the labeling process from the purchase of expensive labeled precursors. Instead, it employs a late-stage functionalization strategy where a readily available, non-labeled ketone precursor (Compound I) undergoes a rigorous H-D exchange reaction. By utilizing strong alkaline conditions with sodium deuteroxide in heavy water, the process efficiently replaces five hydrogen atoms at the alpha-positions of the ketone with deuterium. This is followed by a reductive amination step that introduces the sixth deuterium atom using a deuterated reducing agent. This modular approach drastically reduces raw material costs while simultaneously simplifying the supply chain, as the bulk chemicals required are commodity items rather than specialized isotopic reagents.
Moreover, this novel route offers exceptional flexibility in controlling the isotopic profile of the final product. The process parameters, such as the molar ratio of base to substrate and the reaction temperature, allow for precise tuning of the deuterium incorporation, ensuring consistent batch-to-batch reproducibility. The ability to achieve a total yield of approximately 44% over just two to three synthetic steps is a remarkable improvement over the multi-step, low-yield sequences of the past. This efficiency not only lowers the barrier to entry for producing high-quality reference materials but also ensures that analytical laboratories can secure a continuous supply of certified standards without facing the volatility associated with niche labeled chemical markets.
Mechanistic Insights into Base-Catalyzed H-D Exchange and Reductive Amination
The core innovation of this synthesis lies in the mechanistic elegance of the hydrogen-deuterium exchange reaction performed on the aromatic ketone scaffold. In the first stage, Compound I is subjected to basic conditions using sodium deuteroxide (NaOD) in a solvent system comprising 1,4-dioxane and heavy water (D2O). 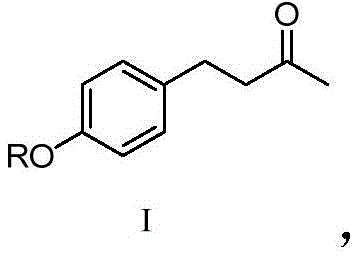 Under these conditions, the alpha-protons adjacent to the carbonyl group become acidic and are rapidly abstracted by the deuteroxide anion. The resulting enolate intermediate is then quenched by deuterium from the heavy water solvent, effectively swapping hydrogen for deuterium.
Under these conditions, the alpha-protons adjacent to the carbonyl group become acidic and are rapidly abstracted by the deuteroxide anion. The resulting enolate intermediate is then quenched by deuterium from the heavy water solvent, effectively swapping hydrogen for deuterium. 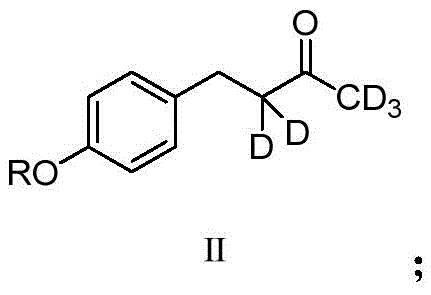 This equilibrium process is driven to completion by the large excess of deuterium source, resulting in the formation of Intermediate Compound II, which carries five deuterium atoms on the alkyl chain adjacent to the phenyl ring.
This equilibrium process is driven to completion by the large excess of deuterium source, resulting in the formation of Intermediate Compound II, which carries five deuterium atoms on the alkyl chain adjacent to the phenyl ring.
Following the successful installation of the isotopic label on the ketone fragment, the synthesis proceeds via a reductive amination mechanism to couple the two halves of the ractopamine molecule. Intermediate Compound II reacts with Compound III, an amino-alcohol derivative, to form an imine or iminium ion intermediate in situ.  The addition of a deuterated reducing agent, such as sodium cyanoborodeuteride, selectively reduces this nitrogen-carbon double bond. Crucially, this step introduces the sixth deuterium atom at the benzylic position adjacent to the amine nitrogen. This dual-labeling strategy—combining H-D exchange on the carbon skeleton with deuterated reduction at the amine linkage—ensures that the final molecule possesses the necessary mass shift for distinct MS detection. The final step involves the removal of protecting groups (such as methyl or benzyl ethers) and subsequent salt formation with hydrochloric acid to yield the stable hydrochloride salt, Compound V.
The addition of a deuterated reducing agent, such as sodium cyanoborodeuteride, selectively reduces this nitrogen-carbon double bond. Crucially, this step introduces the sixth deuterium atom at the benzylic position adjacent to the amine nitrogen. This dual-labeling strategy—combining H-D exchange on the carbon skeleton with deuterated reduction at the amine linkage—ensures that the final molecule possesses the necessary mass shift for distinct MS detection. The final step involves the removal of protecting groups (such as methyl or benzyl ethers) and subsequent salt formation with hydrochloric acid to yield the stable hydrochloride salt, Compound V. 
How to Synthesize Ractopamine Hydrochloride-D6 Efficiently
The operational protocol for synthesizing this high-value isotopic standard is designed for robustness and ease of execution in a standard pharmaceutical pilot plant. The process begins with the dissolution of the starting ketone in an anhydrous organic solvent under an inert atmosphere, followed by the careful addition of the deuterium source and base. Reaction monitoring is typically conducted to ensure complete exchange before proceeding to the coupling stage. The subsequent reductive amination requires precise temperature control, initially cooling the mixture to suppress side reactions before allowing it to warm to ambient temperature for completion.
- Perform H-D exchange on Compound I using NaOD and D2O in 1,4-dioxane at 50°C to generate the pentadeuterated ketone intermediate (Compound II).
- Conduct reductive amination by reacting Compound II with Compound III (octopamine derivative) using sodium cyanoborodeuteride in deuterated methanol.
- Execute deprotection using boron tribromide or catalytic hydrogenation, followed by salt formation with hydrochloric acid to yield the final Ractopamine-D6 hydrochloride.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of the synthesis route described in CN113061094A offers transformative advantages in terms of cost structure and supply security. The primary driver of cost reduction is the elimination of dependency on proprietary, high-cost labeled starting materials. By shifting the labeling burden to a chemical transformation step using inexpensive heavy water and commodity bases, the raw material cost profile is significantly flattened. This structural change in the bill of materials means that the final price of the Ractopamine-D6 standard can be optimized substantially, allowing testing laboratories to budget more effectively for their consumables without compromising on the quality of their data.
From a supply chain reliability perspective, this method mitigates the risk of bottlenecks associated with specialized isotope suppliers. Since the key intermediates are generated in-house from widely available precursors, manufacturers are not held hostage by the lead times or allocation quotas of external vendors who produce labeled octopamine or raspberry ketone. The use of common solvents like tetrahydrofuran, dioxane, and methanol further enhances supply resilience, as these chemicals are produced at a massive global scale and are unlikely to face shortages. This autonomy in raw material sourcing ensures a consistent and uninterrupted flow of finished goods to the market, which is critical for maintaining the accreditation and operational continuity of high-throughput testing facilities.
Scalability and environmental compliance are also markedly improved with this streamlined process. The reaction conditions are mild, typically requiring temperatures around 50°C, which reduces energy consumption compared to high-temperature or high-pressure alternatives. Furthermore, the process avoids the use of transition metal catalysts that often require complex and costly removal steps to meet residual metal specifications in pharmaceutical standards. The simplified workup procedures, involving standard extraction and chromatography techniques, facilitate easier waste management and reduce the generation of hazardous byproducts. This alignment with green chemistry principles not only lowers disposal costs but also future-proofs the manufacturing process against increasingly stringent environmental regulations in the fine chemical sector.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of deuterated ractopamine standards. These insights are derived directly from the experimental data and process descriptions within the patent documentation, providing clarity for potential partners and end-users. Understanding these nuances is essential for integrating this material into your quality assurance workflows.
Q: Why is Ractopamine-D6 preferred over mono-deuterated standards for LC-MS analysis?
A: Mono-deuterated analogues often suffer from mass spectral interference due to the small mass difference (1 Da) from the natural abundance analyte. Ractopamine-D6 provides a mass shift of 6 units, ensuring distinct separation and accurate quantification in Isotope Dilution Mass Spectrometry (IDMS), which is critical for meeting WADA and international food safety standards.
Q: What is the achievable isotopic abundance using this H-D exchange method?
A: The patented process utilizes aggressive H-D exchange conditions with sodium deuteroxide and heavy water, followed by deuterated reduction. This dual-labeling strategy consistently achieves isotopic abundances exceeding 96%, with chemical purity reaching up to 99.8% after purification.
Q: Is this synthesis route scalable for commercial production of reference standards?
A: Yes, the process relies on conventional chemical reactions such as base-catalyzed exchange and reductive amination using readily available solvents like THF and dioxane. The mild reaction temperatures (50°C) and absence of exotic catalysts make it highly suitable for scale-up from gram to kilogram quantities without significant process redesign.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ractopamine Hydrochloride-D6 Supplier
At NINGBO INNO PHARMCHEM, we recognize that the integrity of food safety and anti-doping testing hinges on the quality of the reference standards used. Our technical team has extensively analyzed the route disclosed in CN113061094A and validated its potential for industrial application. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the rigorous demands of the global analytical market. Our facility is equipped with stringent purity specifications and rigorous QC labs capable of verifying isotopic abundance and chemical purity to the highest international standards, guaranteeing that every batch of Ractopamine-D6 performs flawlessly in your LC-MS instruments.
We invite you to collaborate with us to secure a sustainable supply of this critical reagent. Our experts are ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how our optimized manufacturing process can reduce your overall procurement spend. Please contact our technical procurement team today to request specific COA data, route feasibility assessments, and samples for your validation protocols. Let us be your partner in advancing precision analytics and safeguarding public health through superior chemical manufacturing.
