Scalable Synthesis of Boron-Dioxygen Neutral Radicals for Advanced Electronic Applications
Scalable Synthesis of Boron-Dioxygen Neutral Radicals for Advanced Electronic Applications
The landscape of functional material synthesis is undergoing a significant transformation with the disclosure of patent CN112961176A, which introduces a robust and scalable preparation method for boron-dioxygen ring neutral radical compounds. These unique chemical species have garnered intense interest for their potential applications in organic synthetic reagents, magnetic system components, and next-generation chemical sensors. Historically, the synthesis of such stable neutral radicals has been plagued by harsh reaction conditions and complex purification protocols, limiting their commercial viability. This new technical disclosure offers a paradigm shift by utilizing a direct alkali metal reduction strategy that operates under mild conditions, providing a reliable pathway for the industrial production of high-purity specialty chemicals. For R&D directors and procurement managers alike, this represents a critical opportunity to secure supply chains for advanced electronic materials while simultaneously driving down manufacturing costs through process intensification.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this innovation, the synthesis of boron-containing radicals relied heavily on strategies that were inherently dangerous and operationally complex. For instance, the work by Professor Douglas W. Stephan's group in 2016 utilized a hindered Lewis acid-base pair to activate hydrogen, a process that necessitated high-temperature conditions and the handling of hydrogen gas, introducing significant safety risks in a plant environment. Furthermore, these traditional routes often generated substantial reaction by-products that necessitated tedious column chromatographic separation, a bottleneck that severely hampers scalability and increases solvent consumption. Similarly, the approach by Professor Gregory H. Robinson involving dithiolithium reagents required the pre-synthesis of unstable precursors, adding extra steps and reducing the overall atom economy. These legacy methods simply cannot meet the rigorous demands of modern large-scale production where safety, efficiency, and environmental compliance are paramount.
The Novel Approach
In stark contrast, the method described in patent CN112961176A streamlines the synthesis into a single-pot reaction that directly constructs the boron-dioxygen ring neutral radical. By employing readily available alkali metals such as potassium or potassium graphite as reducing agents, the process activates the precursor molecules under remarkably mild conditions, typically ranging from -20°C to 50°C, with room temperature being preferred. This eliminates the need for high-energy inputs and dangerous gaseous reagents. The reaction proceeds with excellent chemical selectivity, minimizing the formation of side products and allowing for straightforward isolation of the target compound. 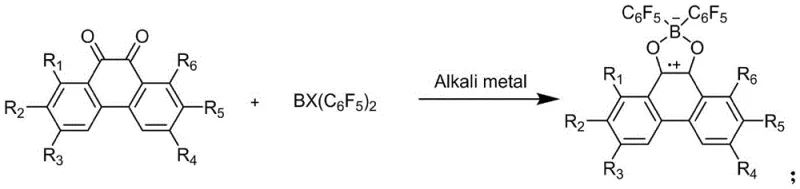 This simplified workflow not only enhances operator safety but also drastically reduces the downtime associated with complex purification sequences, making it an ideal candidate for cost reduction in specialty chemical manufacturing.
This simplified workflow not only enhances operator safety but also drastically reduces the downtime associated with complex purification sequences, making it an ideal candidate for cost reduction in specialty chemical manufacturing.
Mechanistic Insights into Alkali Metal Reduction Strategy
The core of this technological breakthrough lies in the efficient single-electron transfer mechanism facilitated by the alkali metal. When the halogenated bis(pentafluorophenyl)borane reacts with the phenanthrenequinone derivative (Formula I) in the presence of an alkali metal, the metal acts as a potent electron donor. This triggers a reductive coupling that closes the boron-dioxygen ring while simultaneously generating the stable neutral radical character on the boron center. The choice of solvent plays a crucial role in stabilizing the intermediate species; anhydrous tetrahydrofuran is particularly effective due to its ability to coordinate with the metal cations, thereby enhancing the solubility and reactivity of the reducing agent. The structural integrity of the resulting radical is maintained by the steric protection provided by the pentafluorophenyl groups on the boron atom, preventing dimerization or decomposition.
From an impurity control perspective, this mechanism is exceptionally clean. Unlike oxidative methods that often produce varied oxygenated by-products, this reductive pathway is highly specific. The absence of aggressive oxidants or unstable intermediates means that the impurity profile is significantly simplified. 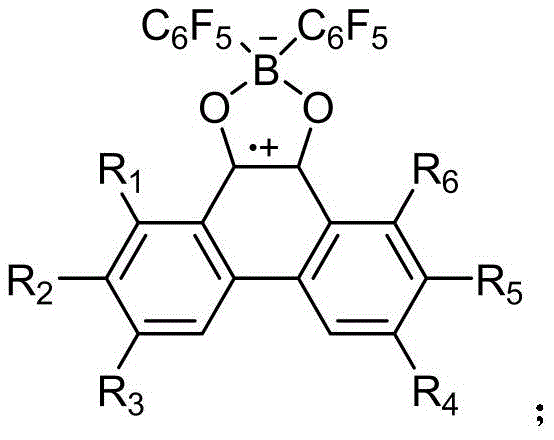 Furthermore, the by-products of the reaction are primarily inorganic salts (such as potassium chloride), which are easily removed by simple filtration through diatomite. This mechanistic elegance ensures that the final product meets stringent purity specifications required for electronic applications without the need for resource-intensive chromatographic purification, thereby preserving the chemical integrity of the sensitive radical species.
Furthermore, the by-products of the reaction are primarily inorganic salts (such as potassium chloride), which are easily removed by simple filtration through diatomite. This mechanistic elegance ensures that the final product meets stringent purity specifications required for electronic applications without the need for resource-intensive chromatographic purification, thereby preserving the chemical integrity of the sensitive radical species.
How to Synthesize Boron-Dioxygen Ring Neutral Radical Efficiently
Implementing this synthesis route requires careful attention to inert atmosphere techniques and solvent quality to ensure optimal yields and reproducibility. The process is designed to be adaptable, allowing for variations in the substituents on the phenanthrenequinone core (R1-R6) to tune the electronic properties of the final radical. The following guide outlines the standardized operational procedure derived from the patent examples, focusing on the critical control points for successful scale-up. For detailed step-by-step instructions including exact stoichiometry and workup parameters, please refer to the technical guide below.
- Under an inert atmosphere, combine halogenated bis(pentafluorophenyl)borane, the phenanthrenequinone derivative (Formula I), and an alkali metal reducing agent in a dried reaction vessel.
- Add anhydrous organic solvent such as tetrahydrofuran or toluene and maintain the reaction mixture at a temperature between -20°C and 50°C for 12 to 24 hours.
- Remove the solvent under vacuum, extract the residue with toluene or ether, filter off inorganic salts through diatomite, and purify the final radical product via recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthesis method translates into tangible strategic advantages that go beyond mere technical feasibility. The elimination of hazardous reagents like hydrogen gas and the removal of column chromatography from the workflow fundamentally alter the cost structure of production. By simplifying the unit operations, manufacturers can achieve a significant reduction in capital expenditure related to safety infrastructure and a drastic decrease in operating expenses associated with solvent recovery and waste disposal. This process intensification allows for faster batch turnover times, directly addressing the industry-wide challenge of reducing lead time for high-purity specialty chemicals.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the simplification of the purification train. Traditional methods relying on column chromatography consume vast quantities of silica gel and solvents, creating a heavy burden on waste management budgets. By shifting to a recrystallization-based purification, the process eliminates these variable costs entirely. Additionally, the use of commodity alkali metals instead of specialized organometallic precursors lowers the raw material bill of materials. The mild reaction conditions also reduce energy consumption for heating and cooling, contributing to a leaner and more cost-effective manufacturing profile that enhances overall margin potential.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of widely available starting materials. The precursors, such as phenanthrenequinone derivatives and chloroboranes, are commercially accessible and do not rely on fragile supply chains for exotic catalysts. The robustness of the reaction conditions—tolerating a range of temperatures and solvents—means that production is less susceptible to minor fluctuations in utility availability. This reliability ensures consistent output volumes, allowing downstream customers in the electronics and polymer sectors to plan their inventory with greater confidence and security against market volatility.
- Scalability and Environmental Compliance: Scaling this chemistry from gram to kilogram levels is straightforward due to the absence of exothermic hazards associated with hydrogen activation. The reaction generates benign inorganic salts as the primary waste stream, which are easy to handle and dispose of in compliance with environmental regulations. The ability to synthesize these compounds in large amounts via simple recrystallization demonstrates clear industrial-grade potential. This aligns perfectly with green chemistry principles by minimizing solvent usage and avoiding toxic heavy metal catalysts, thereby future-proofing the supply chain against tightening environmental legislation.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating this technology for their specific applications, we have compiled answers to common questions regarding the synthesis and properties of these boron-dioxygen radicals. These insights are derived directly from the experimental data and technical disclosures within the patent documentation, ensuring accuracy and relevance for process development discussions. Understanding these nuances is critical for assessing the feasibility of integrating this material into your existing product lines.
Q: How does this new method improve upon previous synthesis strategies like those by Professor Stephan?
A: Unlike previous methods requiring high-temperature hydrogen activation or complex dithiolithium precursors, this patented process utilizes mild alkali metal reduction at room temperature, eliminating dangerous hydrogen gas handling and simplifying the operational workflow significantly.
Q: Is column chromatography required for purifying these boron-containing radicals?
A: No, one of the key commercial advantages of this invention is that the reaction produces no obvious by-products, allowing for purification solely through simple recrystallization from solvents like toluene, which drastically reduces processing time and solvent waste compared to column chromatography.
Q: What yields can be expected for the phenanthrenequinone boron ring neutral radicals?
A: Experimental data within the patent indicates isolated yields ranging from approximately 14% to 52%, depending on the specific substituents on the phenanthrenequinone core, with methoxy-substituted variants generally showing higher efficiency.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Boron-Dioxygen Ring Neutral Radical Supplier
As the demand for advanced functional materials continues to surge, having a manufacturing partner with deep expertise in radical chemistry is essential. NINGBO INNO PHARMCHEM stands at the forefront of this field, leveraging the latest innovations like patent CN112961176A to deliver superior quality compounds. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements without compromising on quality. We operate stringent purity specifications and utilize rigorous QC labs to guarantee that every batch of boron-dioxygen neutral radical meets the exacting standards required for high-performance electronic and magnetic applications.
We invite you to collaborate with us to explore how this cost-effective synthesis route can benefit your projects. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume needs. Please contact us today to request specific COA data and route feasibility assessments, and let us help you secure a competitive advantage in the rapidly evolving market for specialty electronic chemicals.
