Revolutionizing Bis-indolyl Alkyl Synthesis: A Green Solid Superacid Catalytic Route for Commercial Scale-up
Revolutionizing Bis-indolyl Alkyl Synthesis: A Green Solid Superacid Catalytic Route for Commercial Scale-up
The pharmaceutical and fine chemical industries are currently undergoing a paradigm shift towards greener, more sustainable manufacturing processes, driven by both regulatory pressure and the economic necessity of reducing waste. Patent CN1332948C introduces a groundbreaking methodology for the synthesis of bis-indolyl alkyl derivatives, a class of compounds renowned for their significant biological activities including antitumor, antiviral, and anti-inflammatory properties. This innovation replaces traditional, hazardous liquid acid catalysts with a robust solid superacid system, specifically sulfated titanium dioxide (SO4 2-/TiO2), operating under solvent-free conditions. By eliminating the reliance on volatile organic solvents and corrosive mineral acids, this technology not only mitigates environmental impact but also streamlines the downstream purification process, offering a compelling value proposition for manufacturers of high-value pharmaceutical intermediates. The technical elegance of this approach lies in its simplicity: a mechanical grinding process that achieves high conversion rates within a short timeframe, demonstrating that efficiency and sustainability can coexist in modern organic synthesis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of bis-indolyl alkyl compounds has relied heavily on the use of traditional protonic acids such as sulfuric acid, hydrofluoric acid, or phosphoric acid, as well as various Lewis acids like iron(III) chloride, indium trichloride, or ceric ammonium nitrate. While these catalysts can drive the condensation reaction between indoles and aldehydes, they are fraught with significant operational and environmental drawbacks that hinder their suitability for modern large-scale production. The use of liquid acids often necessitates the presence of large volumes of organic solvents to manage heat dissipation and mixing, leading to substantial generation of hazardous waste streams that require costly treatment and disposal protocols. Furthermore, liquid acids are notoriously corrosive, imposing severe demands on reactor materials and maintenance schedules, while their homogeneous nature makes separation from the final product difficult, often requiring complex neutralization and extraction steps that erode overall yield. The inability to easily recover and recycle these homogeneous catalysts results in a linear consumption of expensive reagents, driving up the raw material costs and creating a discontinuous, batch-heavy workflow that struggles to meet the demands of continuous manufacturing.
The Novel Approach
In stark contrast to these legacy methods, the novel approach detailed in the patent utilizes a heterogeneous solid superacid catalyst, SO4 2-/TiO2, which operates effectively under solvent-free conditions through a mechanochemical grinding process. This method fundamentally alters the reaction landscape by confining the interaction between the aldehyde and indole substrates to the surface of the solid acid, thereby maximizing atomic efficiency and minimizing the E-factor of the process. The absence of bulk solvent not only removes the cost and safety risks associated with solvent storage and recovery but also intensifies the reaction rate through close contact of reactants in the solid state. As illustrated in the reaction schemes below, this versatile catalytic system accommodates a wide range of substrates, from simple aromatic aldehydes to complex heterocyclic and aliphatic variants, delivering high-purity products with minimal byproduct formation. The simplicity of the workup procedure, involving merely a solvent wash and filtration to recover the catalyst, represents a drastic simplification of the operational workflow compared to traditional aqueous quenching and extraction methods.
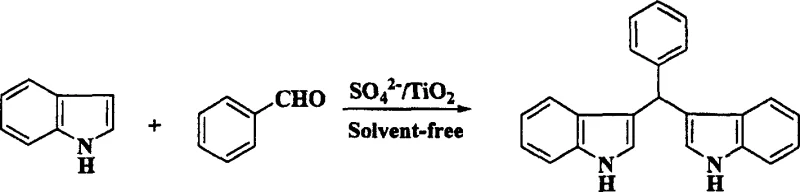
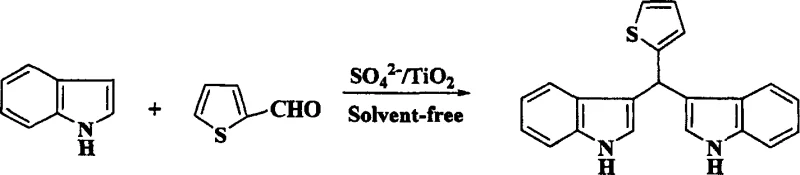
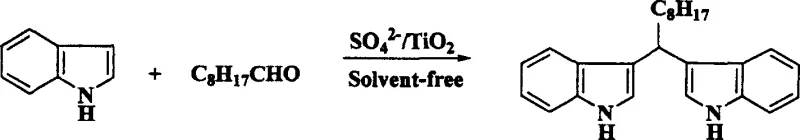
Mechanistic Insights into SO4 2-/TiO2 Catalyzed Condensation
The efficacy of the SO4 2-/TiO2 catalyst stems from its unique electronic structure, where the coordination of sulfate ions to the titanium dioxide surface generates strong Brønsted and Lewis acid sites that are significantly more acidic than the parent oxide. In the context of bis-indolyl synthesis, these superacidic sites play a critical role in activating the carbonyl group of the aldehyde substrate, increasing its electrophilicity and facilitating the nucleophilic attack by the electron-rich C3 position of the indole ring. This activation lowers the energy barrier for the formation of the initial carbinolamine intermediate, which subsequently undergoes dehydration to form the reactive indolyl-methyl cation species. The solid surface acts as a template that stabilizes this cationic intermediate, preventing polymerization side reactions and directing the second molecule of indole to attack, thereby completing the formation of the bis-indolyl methane scaffold. The heterogeneity of the catalyst ensures that the active sites are accessible yet distinct from the bulk product, allowing the reaction to proceed with high selectivity even in the absence of a solvating medium to stabilize charged species.
From an impurity control perspective, the solid-state nature of this reaction offers inherent advantages over solution-phase chemistry by limiting the mobility of reactive intermediates and suppressing bimolecular side reactions that typically lead to oligomerization or tar formation. The mild thermal conditions, typically ranging from 25°C to 60°C, further contribute to product integrity by preventing the thermal decomposition of sensitive aldehyde substrates or the indole nucleus, which can occur under the harsh reflux conditions often required by liquid acid catalysts. Moreover, the ease of catalyst separation via simple filtration means that there is no risk of metal leaching or acid contamination in the final product, a critical quality attribute for pharmaceutical intermediates destined for downstream API synthesis. The ability to regenerate the catalyst through simple drying and calcination ensures that the acidic strength remains consistent over multiple cycles, providing a stable and predictable impurity profile that simplifies the validation process for GMP manufacturing environments.
How to Synthesize Bis-indolyl Alkyl Derivatives Efficiently
The operational protocol for this synthesis is remarkably straightforward, designed to minimize technical barriers for adoption in both laboratory and pilot plant settings. The process begins with the precise stoichiometric mixing of the aldehyde and indole substrates, typically in a 1:2 molar ratio to ensure complete consumption of the aldehyde and maximize yield. The solid superacid catalyst is then introduced, and the mixture is subjected to mechanical grinding, which can be performed using standard mortar and pestle setups for small batches or industrial ball mills for larger scales. Detailed standardized synthetic steps, including specific catalyst loading amounts and reaction times for various substrates, are outlined in the guide below to ensure reproducibility and optimal performance.
- Mix aldehyde (C1-C10 aliphatic or aromatic) and indole in a 1: 2 molar ratio with 30-80 mg of SO4 2-/TiO2 catalyst per mmol of aldehyde.
- Perform grinding reaction at temperatures between 25°C to 60°C for 0.5 to 5 hours until TLC indicates completion.
- Wash the reaction mixture with ethyl acetate, filter to recover the solid catalyst, and concentrate the filtrate to obtain the pure product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this solid superacid-catalyzed process represents a strategic opportunity to de-risk the supply of critical pharmaceutical intermediates while simultaneously driving down the total cost of ownership. By eliminating the need for volatile organic solvents and corrosive liquid acids, the process drastically reduces the logistical burden associated with the storage, handling, and disposal of hazardous chemicals, leading to significant operational expenditure savings. The simplified workup procedure, which avoids complex aqueous extractions and neutralization steps, shortens the overall production cycle time, allowing for faster throughput and improved responsiveness to market demand fluctuations. Furthermore, the recyclability of the solid catalyst introduces a circular economy element to the manufacturing process, reducing the dependency on fresh catalyst purchases and insulating the supply chain from volatility in raw material pricing.
- Cost Reduction in Manufacturing: The elimination of organic solvents removes a major cost center associated with solvent purchase, recovery distillation, and waste incineration, directly improving the gross margin of the final product. Additionally, the replacement of expensive and corrosive liquid acids with a reusable solid superacid catalyst reduces the frequency of reactor maintenance and replacement, extending the lifespan of capital equipment. The high atom economy of the solvent-free grinding process ensures that raw materials are converted efficiently into product, minimizing waste generation and the associated costs of disposal and environmental compliance. These cumulative efficiencies result in a leaner manufacturing model that is less susceptible to cost inflation in the chemical supply market.
- Enhanced Supply Chain Reliability: The robustness of the solid catalyst and the simplicity of the reaction conditions contribute to a highly reliable production process with minimal risk of batch failure due to catalyst deactivation or side reactions. Since the catalyst can be recovered and reused multiple times without significant loss of activity, the supply chain is less vulnerable to disruptions in the availability of fresh catalytic reagents. The mild reaction temperatures and lack of exothermic solvent hazards allow for safer scaling of production, ensuring that delivery schedules can be met consistently even during periods of high demand. This stability is crucial for maintaining continuity in the supply of key intermediates for downstream drug manufacturing.
- Scalability and Environmental Compliance: The solvent-free nature of this technology aligns perfectly with increasingly stringent global environmental regulations, reducing the facility's carbon footprint and regulatory reporting burden. Scaling this process is technically straightforward, as it relies on mechanical grinding equipment like ball mills which are readily available and easy to operate at multi-ton scales without the need for complex solvent containment systems. The reduction in hazardous waste generation simplifies the permitting process for new production lines and enhances the company's sustainability profile, which is becoming a key differentiator in B2B procurement decisions. This future-proofing of the manufacturing process ensures long-term viability and compliance with evolving green chemistry standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this solid superacid catalytic technology, based on the specific data and embodiments provided in the patent literature. These insights are intended to clarify the operational parameters and benefits for potential partners evaluating this route for their own supply chains. Comprehensive answers derived from the patent specifications follow below to assist in your decision-making process.
Q: What are the primary advantages of using SO4 2-/TiO2 over traditional liquid acids?
A: The use of solid superacid SO4 2-/TiO2 eliminates the need for corrosive liquid acids like H2SO4 or HF, significantly reducing equipment maintenance costs and hazardous waste generation while allowing for easy catalyst recovery and reuse.
Q: Can this solvent-free method be scaled for industrial production?
A: Yes, the process is highly scalable as it utilizes simple grinding or ball milling techniques under mild temperatures (25°C-60°C), avoiding the complexities of solvent handling and recovery associated with large-scale batch reactors.
Q: What types of aldehydes are compatible with this synthesis route?
A: The method demonstrates broad substrate scope, successfully reacting with C1 to C10 aliphatic aldehydes, aromatic aldehydes like benzaldehyde, and heterocyclic aldehydes such as thiophene-2-carbaldehyde to yield corresponding bis-indolyl derivatives.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Bis-indolyl Alkyl Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the solid superacid catalytic route described in patent CN1332948C for the production of high-quality bis-indolyl alkyl derivatives. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this green chemistry approach are fully realized in a GMP-compliant environment. Our facilities are equipped with state-of-the-art milling and solid-state reaction capabilities, supported by rigorous QC labs that enforce stringent purity specifications to meet the exacting standards of the global pharmaceutical industry. We are committed to leveraging this innovative technology to deliver intermediates that are not only cost-effective but also environmentally responsible.
We invite forward-thinking procurement leaders to engage with our technical team to explore how this advanced synthesis route can optimize your specific supply chain requirements. By requesting a Customized Cost-Saving Analysis, you can gain a detailed understanding of the economic advantages tailored to your volume needs. We encourage you to contact our technical procurement team today to索取 specific COA data and route feasibility assessments, ensuring that your next project benefits from the highest standards of efficiency and quality assurance.
