Revolutionizing Antiarrhythmic Production: A Technical Deep Dive into High-Purity Mexiletine Hydrochloride Synthesis
Revolutionizing Antiarrhythmic Production: A Technical Deep Dive into High-Purity Mexiletine Hydrochloride Synthesis
The pharmaceutical landscape for cardiovascular therapeutics is constantly evolving, driven by the need for safer, more efficient, and environmentally sustainable manufacturing processes. A pivotal advancement in this domain is detailed in patent CN102603543B, which outlines a robust preparation method for Mexiletine Hydrochloride, a critical Class IB antiarrhythmic agent widely used for managing ventricular arrhythmias. This technical insight report analyzes the transformative potential of this synthesis route, moving away from hazardous traditional oxidation methods toward a streamlined sulfonylation-ammonolysis strategy. By leveraging catalytic triethylamine and avoiding toxic heavy metals, this approach not only enhances the safety profile of the manufacturing facility but also ensures the delivery of high-purity active pharmaceutical ingredients (APIs) essential for patient safety. For R&D directors and procurement specialists, understanding the nuances of this pathway is vital for securing a reliable supply chain of this life-saving medication.
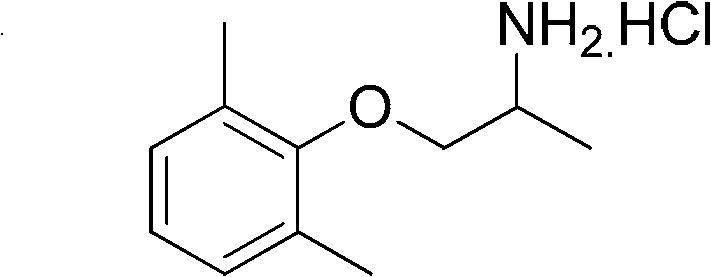
Mexiletine Hydrochloride, chemically known as 1-(2,6-dimethylphenoxy)-2-propylamine hydrochloride (CAS No: 5370-01-4), serves as a sodium channel blocker that stabilizes cardiac membrane excitability. The structural integrity and purity of this molecule are paramount, as impurities can lead to severe adverse effects in patients suffering from acute or chronic heart conditions. The patent provides a comprehensive blueprint for synthesizing this compound with exceptional fidelity, utilizing a four-step sequence that begins with the hydroxypropylation of 2,6-dimethylphenol. This initial step sets the foundation for the entire synthetic architecture, ensuring that the stereochemical and regiochemical outcomes are controlled effectively. As we delve deeper into the technical specifics, it becomes evident that this methodology represents a significant leap forward in process chemistry, offering a viable solution for manufacturers seeking to optimize their production lines while adhering to increasingly strict global environmental regulations.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of Mexiletine Hydrochloride has been plagued by significant safety and environmental challenges inherent to older synthetic methodologies. Traditional routes typically rely on the oxidation of intermediate alcohols using sodium dichromate, a reagent containing hexavalent chromium which is classified as a potent carcinogen and highly toxic substance. The handling of such hazardous materials necessitates elaborate safety protocols, specialized waste treatment facilities, and rigorous worker protection measures, all of which inflate operational expenditures and introduce substantial liability risks. Furthermore, conventional processes often employ Raney-Nickel catalyzed hydrogenation under high pressure, specifically around 10 Kg/cm², to reduce oxime intermediates. Raney-Nickel is pyrophoric and poses a severe fire hazard, while high-pressure hydrogenation requires expensive autoclaves and meticulous monitoring to prevent catastrophic failures. These factors combined result in a cumbersome production workflow with a relatively low total yield of approximately 35.3%, making it economically inefficient and environmentally unsustainable for modern green chemistry standards.
The Novel Approach
In stark contrast, the methodology disclosed in patent CN102603543B introduces a paradigm shift by eliminating the need for toxic oxidants and high-pressure reduction steps entirely. This innovative route utilizes a direct nucleophilic substitution strategy where the hydroxyl group of the intermediate alcohol is activated via sulfonylation using methanesulfonyl chloride or p-toluenesulfonyl chloride. This activation converts a poor leaving group into an excellent sulfonate ester, facilitating a smooth subsequent displacement by ammonia. The process operates under much milder conditions, utilizing catalytic amounts of triethylamine as a base and acid scavenger, which simplifies the reaction workup and minimizes the generation of saline waste. By bypassing the oxidation-reduction cycle completely, the new method achieves a significantly improved overall yield exceeding 45% and delivers a final product with purity greater than 99.5%. This transition not only mitigates the environmental footprint by removing chromium waste streams but also drastically simplifies the equipment requirements, allowing for more flexible and scalable manufacturing operations.
Mechanistic Insights into Triethylamine-Catalyzed Hydroxypropylation and Sulfonylation
The core of this synthetic success lies in the precise control of reaction kinetics and thermodynamics during the initial functionalization of the phenolic ring. In the first step, 2,6-dimethylphenol undergoes hydroxypropylation with propylene oxide. The presence of catalytic triethylamine is crucial here; it acts as a nucleophilic catalyst that activates the epoxide ring, facilitating the attack by the phenolic oxygen. This reaction is conducted in alcoholic solvents such as methanol or ethanol at temperatures ranging from 60°C to 100°C. The choice of solvent and temperature is optimized to balance reaction rate with selectivity, ensuring that the formation of the desired 1-(2,6-dimethylphenoxy)isopropanol is maximized while minimizing polymerization of the epoxide or over-alkylation of the phenol. The resulting alcohol is a stable intermediate that can be isolated or telescoped directly into the next stage, demonstrating the robustness of the chemical transformation.
Following the formation of the ether linkage, the mechanism proceeds through a classic activation-substitution sequence. The secondary hydroxyl group of the isopropanol derivative is inherently unreactive towards direct nucleophilic displacement by ammonia due to the poor leaving group ability of the hydroxide ion. To overcome this kinetic barrier, the patent employs a sulfonylation step where the alcohol is treated with a sulfonyl chloride in the presence of triethylamine at low temperatures (0-35°C). Triethylamine serves a dual purpose: it neutralizes the hydrochloric acid byproduct generated during sulfonylation, driving the equilibrium forward, and it prevents the acid-catalyzed decomposition of sensitive intermediates. The resulting mesylate or tosylate ester possesses a sulfonate leaving group that is highly susceptible to nucleophilic attack. In the final amination step, liquid ammonia or concentrated aqueous ammonia acts as a strong nucleophile, displacing the sulfonate group in an SN2-type mechanism to install the primary amine functionality. This mechanistic elegance ensures high regioselectivity and minimizes the formation of elimination byproducts, which is critical for maintaining the high purity profile required for pharmaceutical applications.
How to Synthesize Mexiletine Hydrochloride Efficiently
Implementing this synthesis route requires a disciplined approach to process parameters to ensure reproducibility and quality at scale. The patent outlines a clear sequence of operations that balances reaction efficiency with ease of purification. The process begins with the careful mixing of stoichiometric ratios of phenol, epoxide, and catalyst, followed by controlled heating to drive the etherification to completion. Subsequent steps involve precise temperature management during the exothermic sulfonylation to prevent runaway reactions, followed by a high-temperature ammonolysis in a sealed vessel to ensure sufficient pressure for the gas-liquid reaction. The final crystallization step is particularly critical, utilizing specific solvent systems like ethanol and diethyl ether to precipitate the hydrochloride salt in a highly pure crystalline form. For detailed operational guidelines and specific stoichiometric ratios derived from the experimental examples, please refer to the standardized synthesis protocol below.
- Hydroxypropylation: React 2,6-dimethylphenol with propylene oxide using catalytic triethylamine in an alcohol solvent at 60-100°C to form 1-(2,6-dimethylphenoxy)isopropanol.
- Sulfonylation: Activate the intermediate alcohol by reacting with methanesulfonyl chloride or p-toluenesulfonyl chloride in the presence of triethylamine at 0-35°C.
- Ammonolysis: Substitute the sulfonate group with ammonia in a sealed reactor at 80-120°C to generate the free amine base.
- Salt Formation and Purification: Treat the crude amine with hydrochloric acid, followed by recrystallization from alcohol/ether mixtures to obtain high-purity crystals.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing and supply chain perspective, the adoption of this patented synthesis method offers profound advantages that extend beyond mere technical feasibility. For procurement managers tasked with optimizing the cost of goods sold (COGS) for cardiovascular APIs, this route presents a compelling value proposition by fundamentally altering the input cost structure. The elimination of expensive and heavily regulated reagents like sodium dichromate and Raney-Nickel removes significant line items from the raw material budget. Furthermore, the avoidance of high-pressure hydrogenation equipment reduces capital expenditure (CAPEX) requirements for new production lines and lowers maintenance costs for existing facilities. The simplified workflow, which potentially allows for the telescoping of steps without intermediate purification, translates directly into reduced solvent consumption and shorter cycle times. These efficiencies collectively contribute to a substantial reduction in manufacturing costs, enabling suppliers to offer more competitive pricing structures while maintaining healthy margins in a price-sensitive generic pharmaceutical market.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the removal of hazardous waste disposal costs associated with hexavalent chromium. Treating chromium-containing effluent is notoriously expensive and subject to stringent environmental levies; by designing these toxins out of the process, manufacturers achieve immediate and significant operational savings. Additionally, the use of commodity chemicals like triethylamine and methanesulfonyl chloride, which are produced at massive global scales, ensures stable pricing and protects against the volatility often seen with specialized catalytic metals. The higher overall yield of over 45% compared to the traditional 35.3% means that less raw material is required to produce the same amount of finished API, further amplifying the cost efficiency per kilogram of output.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the reliance on readily available, non-proprietary starting materials. 2,6-Dimethylphenol and propylene oxide are bulk petrochemical derivatives with robust global supply networks, reducing the risk of shortages that can plague supply chains dependent on niche catalysts or complex precursors. The simplified process flow also reduces the number of potential failure points in the manufacturing chain; fewer unit operations mean fewer opportunities for equipment downtime or batch failures. This reliability is crucial for pharmaceutical companies that must guarantee continuous supply to meet regulatory commitments and patient demand, minimizing the risk of costly stockouts or production delays that can disrupt the entire downstream formulation schedule.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces new challenges, but this methodology is inherently designed for scalability due to its reliance on standard batch reactor operations rather than specialized high-pressure or high-temperature units. The mild reaction conditions facilitate easier heat transfer and mixing control, which are critical factors when moving from pilot plant to multi-ton commercial production. Moreover, the environmental compliance aspect cannot be overstated; in an era of increasing ESG (Environmental, Social, and Governance) scrutiny, a chromium-free and pyrophoric-metal-free process significantly eases the regulatory burden. This facilitates faster permitting for new facilities and reduces the long-term liability associated with environmental remediation, making it a future-proof investment for sustainable chemical manufacturing.
Frequently Asked Questions (FAQ)
To address common inquiries regarding the technical and commercial implications of this synthesis technology, we have compiled a set of answers based on the detailed data provided in the patent documentation. These questions reflect the concerns typically raised by technical directors and quality assurance teams when evaluating a new supplier or process change. Understanding these nuances helps stakeholders make informed decisions about integrating this high-purity intermediate into their broader drug development pipelines. The following responses clarify the safety improvements, quality metrics, and scalability factors that define the superiority of this approach over legacy methods.
Q: How does the new synthesis method improve safety compared to traditional routes?
A: The novel process eliminates the use of sodium dichromate, a highly toxic and carcinogenic hexavalent chromium oxidant, and avoids high-pressure hydrogenation with pyrophoric Raney-Ni. This significantly reduces hazardous waste generation and operational safety risks associated with high-pressure reactors.
Q: What purity levels can be achieved with this patented production method?
A: According to the patent data, the optimized crystallization and purification steps allow the final Mexiletine Hydrochloride product to achieve a purity exceeding 99.5%, meeting stringent pharmaceutical quality standards without requiring complex chromatographic separations.
Q: Does this process offer advantages for large-scale commercial manufacturing?
A: Yes, the process utilizes readily available raw materials and avoids intermediate purification steps, which simplifies the workflow. The mild reaction conditions and elimination of heavy metal catalysts make it highly suitable for cost-effective commercial scale-up and regulatory compliance.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Mexiletine Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced synthesis methods like the one described in CN102603543B requires a partner with deep technical expertise and proven execution capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in practical, large-volume manufacturing. We are committed to delivering high-purity pharmaceutical intermediates that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our dedication to quality assurance ensures that every batch of Mexiletine Hydrochloride we produce adheres to the highest international standards, providing you with the confidence needed to advance your clinical or commercial programs without supply chain interruptions.
We invite you to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific project requirements. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your volume needs, demonstrating exactly how the elimination of hazardous reagents translates to bottom-line value. We encourage you to contact us today to request specific COA data and route feasibility assessments, allowing us to demonstrate our commitment to being your trusted partner in the complex landscape of cardiovascular drug manufacturing.
