Advanced Synthetic Route for 5-Trifluoromethyl Pyrrole-2-Formic Acid and Commercial Scalability
Advanced Synthetic Route for 5-Trifluoromethyl Pyrrole-2-Formic Acid and Commercial Scalability
The strategic incorporation of fluorine atoms into heterocyclic scaffolds remains a cornerstone of modern medicinal chemistry, driven by the unique ability of fluorine to enhance metabolic stability, lipophilicity, and membrane permeability. In this context, Patent CN102827060B discloses a robust and novel synthetic methodology for producing 5-trifluoromethyl pyrrole-2-formic acid, a high-value building block for next-generation pharmaceutical agents. This technical insight report analyzes the patented process, highlighting its superiority over traditional halogenation-based approaches which often suffer from poor regioselectivity and arduous purification challenges. By leveraging sodium triflinate as a safe and efficient trifluoromethylating agent, this route offers a streamlined pathway that addresses critical pain points in the supply chain of fluorinated intermediates. For R&D directors and procurement specialists, understanding this chemistry is vital for securing a reliable pharma intermediate supplier capable of delivering complex fluorinated motifs with consistent quality.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
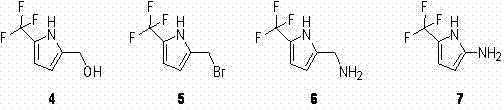
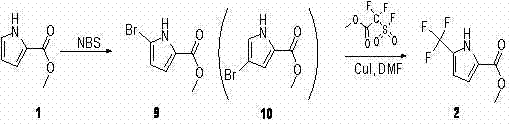
Historically, the functionalization of the pyrrole ring at the 5-position has been fraught with chemoselectivity issues, particularly when attempting to introduce bulky trifluoromethyl groups via indirect halogenation strategies. As illustrated in the prior art attempts referenced within the patent documentation, researchers initially explored a pathway involving N-bromosuccinimide (NBS) to generate a brominated intermediate, intending to subsequently substitute the bromine with a trifluoromethyl group. However, this conventional approach proved fundamentally flawed due to the generation of complex reaction mixtures. Specifically, the reaction of pyrrole-2-methyl formate with NBS yielded a problematic mixture containing only 10% of the desired monobrominated species alongside 80% of a dibrominated byproduct. This lack of selectivity not only devastated the theoretical yield but also created a purification nightmare, as separating these structurally similar brominated congeners is notoriously difficult and cost-prohibitive on a commercial scale. Such inefficiencies render the traditional NBS route unsuitable for the cost reduction in fluorine-containing intermediate manufacturing required by modern high-volume production.
The Novel Approach
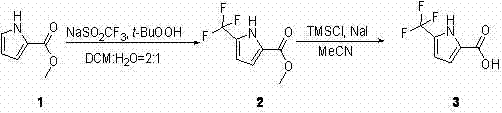
In stark contrast to the失败的 halogenation strategy, the patented method introduces a direct radical trifluoromethylation protocol that bypasses the need for unstable halogenated intermediates entirely. This innovative approach utilizes pyrrole-2-methyl formate as the starting material, reacting it directly with sodium triflinate (NaSO2CF3) in the presence of tert-butyl hydroperoxide (t-BuOOH). The reaction proceeds in a biphasic solvent system of dichloromethane and water, which facilitates the interaction between the organic substrate and the inorganic trifluoromethyl source. This direct installation of the CF3 group eliminates the formation of poly-halogenated impurities, thereby drastically simplifying the downstream workup. Furthermore, the subsequent conversion of the methyl ester to the carboxylic acid is achieved using a mild system comprising trimethylchlorosilane (TMSCl) and sodium iodide (NaI) in acetonitrile. This two-step sequence represents a paradigm shift in how fluorinated pyrroles are accessed, offering a viable solution for the commercial scale-up of complex polymer additives and pharmaceutical precursors that demand high structural fidelity.
Mechanistic Insights into Radical Trifluoromethylation and Mild Hydrolysis
The core of this synthetic breakthrough lies in the generation of trifluoromethyl radicals under mild oxidative conditions. In the first step, tert-butyl hydroperoxide acts as a single-electron oxidant, initiating the decomposition of sodium triflinate to release the highly reactive •CF3 radical. This radical species attacks the electron-rich pyrrole ring preferentially at the 5-position, driven by the electronic activation provided by the nitrogen atom and the ester group at the 2-position. The use of a DCM:Water (2:1) solvent system is critical here, as it solubilizes the organic pyrrole while maintaining the sodium triflinate in the aqueous phase or at the interface, controlling the reaction kinetics to prevent over-trifluoromethylation. This controlled radical mechanism ensures that the reaction stops primarily at the mono-substituted stage, avoiding the statistical distribution of products seen in electrophilic substitutions. For R&D teams, this mechanistic clarity provides confidence in the reproducibility of the process, as the radical flux can be modulated by the rate of oxidant addition, ensuring consistent batch-to-batch quality.
Following the successful construction of the carbon-fluorine bond, the second step involves the cleavage of the methyl ester to reveal the free carboxylic acid. Traditional saponification using strong bases like NaOH or KOH can sometimes lead to ring opening or degradation of sensitive heterocycles. To circumvent this, the patent employs a Lewis acid-mediated hydrolysis using TMSCl and NaI. In this mechanism, sodium iodide reacts with trimethylchlorosilane to generate trimethylsilyl iodide (TMSI) in situ, a potent silylating agent. TMSI effectively cleaves the ester bond to form the trimethylsilyl ester and methyl iodide, which is subsequently hydrolyzed upon aqueous workup to yield the target 5-trifluoromethyl pyrrole-2-formic acid. This mild condition preserves the integrity of the trifluoromethyl group and the pyrrole ring, ensuring high purity specifications are met without the need for aggressive pH adjustments that could compromise product stability.
How to Synthesize 5-Trifluoromethyl Pyrrole-2-Formic Acid Efficiently
Implementing this synthesis requires careful attention to stoichiometry and reaction monitoring to maximize the yield of the intermediate ester before proceeding to hydrolysis. The process begins with the dissolution of the pyrrole starting material and sodium triflinate in the biphasic solvent, followed by the controlled dropwise addition of the peroxide oxidant at ambient temperature. Reaction times typically range from 24 to 36 hours to ensure complete conversion, after which the mixture is quenched with sodium bisulfite to destroy excess oxidant. The organic layer is separated, washed, and purified via silica gel chromatography to isolate the 5-trifluoromethyl pyrrole-2-methyl formate. The detailed standardized synthesis steps for this procedure are provided below.
- Dissolve pyrrole-2-methyl formate and sodium triflinate in a dichloromethane/water mixture, then add tert-butyl hydroperoxide to initiate radical trifluoromethylation.
- Purify the resulting 5-trifluoromethyl pyrrole-2-methyl formate intermediate using silica gel chromatography.
- Reflux the intermediate in acetonitrile with trimethylchlorosilane and sodium iodide under nitrogen to hydrolyze the ester to the final acid.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain perspective, the transition to this direct trifluoromethylation route offers substantial logistical and economic benefits compared to legacy multi-step halogenation sequences. By eliminating the need for isolating unstable brominated intermediates, the process reduces the number of unit operations and the associated handling risks, leading to a more streamlined manufacturing workflow. This simplification directly translates to enhanced supply chain reliability, as fewer processing steps mean fewer opportunities for yield loss or contamination. Moreover, the reagents employed—sodium triflinate, tert-butyl hydroperoxide, and TMSCl—are commodity chemicals available from multiple global sources, mitigating the risk of single-source dependency that often plagues specialized fluorination reagents. This raw material accessibility ensures reducing lead time for high-purity fluorinated intermediates, allowing procurement managers to plan inventory with greater confidence and flexibility.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the significant improvement in crude reaction purity. Unlike the NBS route which generated an 80% byproduct burden requiring expensive and time-consuming separation techniques, the radical trifluoromethylation produces a much cleaner profile. This reduction in impurity load minimizes the consumption of silica gel and solvents during purification, which are often the hidden cost drivers in fine chemical production. Additionally, the avoidance of precious metal catalysts or exotic ligands further lowers the bill of materials, making the final acid more cost-competitive for large-scale API synthesis.
- Enhanced Supply Chain Reliability: The robustness of the biphasic reaction system contributes to superior process safety and scalability. Operating at moderate temperatures (20-40°C for step 1 and 80°C for step 2) reduces the energy footprint and eliminates the need for cryogenic cooling or high-pressure reactors. This operational simplicity allows for flexible manufacturing across different facility types, ensuring continuity of supply even during periods of high market demand. The stability of the intermediate ester also allows for potential telescoping of steps or stockpiling, providing a buffer against upstream supply fluctuations.
- Scalability and Environmental Compliance: Environmental compliance is increasingly critical for sustainable sourcing, and this route aligns well with green chemistry principles. The use of water as a co-solvent reduces the overall volume of organic waste generated per kilogram of product. Furthermore, the byproducts of the hydrolysis step are relatively benign compared to the heavy metal waste streams associated with cross-coupling alternatives. The process generates minimal hazardous waste, simplifying disposal protocols and reducing the environmental compliance burden on manufacturing sites, which is a key consideration for long-term vendor qualification.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this synthetic pathway. These insights are derived directly from the experimental data and comparative analysis presented in the patent literature, aiming to clarify the operational parameters for potential licensees or contract manufacturing partners. Understanding these nuances is essential for technology transfer teams evaluating the feasibility of adopting this route for their specific portfolio needs.
Q: What are the key advantages of this new synthetic route over conventional NBS methods?
A: The new route avoids the formation of difficult-to-separate isomeric mixtures common in NBS bromination pathways, significantly simplifying purification and improving overall process efficiency.
Q: What reagents are critical for the trifluoromethylation step?
A: The process relies on sodium triflinate (NaSO2CF3) as the trifluoromethyl source and tert-butyl hydroperoxide (t-BuOOH) as the oxidant in a biphasic solvent system.
Q: How is the final hydrolysis step performed to ensure product integrity?
A: Instead of harsh acidic or basic conditions, the method uses trimethylchlorosilane and sodium iodide in acetonitrile, providing a mild environment that preserves the sensitive pyrrole ring.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Trifluoromethyl Pyrrole-2-Formic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality fluorinated intermediates play in accelerating drug discovery and development pipelines. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We adhere to stringent purity specifications and utilize rigorous QC labs to verify the identity and potency of every batch, guaranteeing that our 5-trifluoromethyl pyrrole-2-formic acid meets the exacting standards required for GMP manufacturing. Our commitment to quality assurance ensures that your downstream synthesis remains uninterrupted by variability in raw material performance.
We invite you to collaborate with us to leverage this advanced synthetic technology for your upcoming projects. Our experts are ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this optimized route can improve your overall project economics. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us support your journey from molecule to medicine with reliable supply and technical excellence.
