Advanced Resolution Technology for Optically Active Oxathiolane Nucleosides and Commercial Scalability
Advanced Resolution Technology for Optically Active Oxathiolane Nucleosides and Commercial Scalability
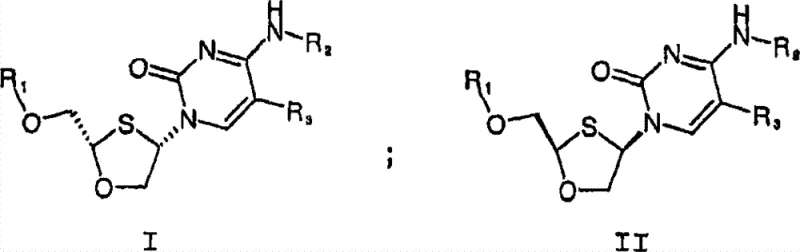
The pharmaceutical industry continuously seeks robust and scalable methodologies for producing high-purity chiral intermediates, particularly for antiviral therapies. Patent CN101142211B introduces a sophisticated process for preparing optically active cis-2-hydroxymethyl-4-(cytosine-1'-yl)-1,3-oxathiolane and its pharmaceutically acceptable salts. This technology addresses the critical need for efficient enantiomeric separation in the synthesis of nucleoside analogs used in treating HIV and Hepatitis B. Unlike traditional methods that rely on expensive chiral HPLC or complex enzymatic resolutions, this invention leverages the formation and fractional crystallization of diastereomeric salts. By utilizing specific chiral acids, such as camphorsulfonic acid, the process achieves high enantiomeric excess through physical separation techniques that are inherently more amenable to large-scale manufacturing. This approach not only enhances the purity profile of the final active pharmaceutical ingredient but also significantly streamlines the production workflow, making it a vital asset for reliable pharmaceutical intermediate supplier networks aiming to optimize their antiviral portfolios.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the isolation of single enantiomers for nucleoside analogs has presented substantial challenges in both technical feasibility and economic efficiency. Conventional strategies often involve chiral high-performance liquid chromatography (HPLC), which, while effective for analytical purposes, suffers from severe limitations when applied to industrial production due to low throughput and high operational costs. Alternatively, stereospecific synthesis from pure starting materials can be prohibitively expensive due to the cost of chiral pool precursors. Enzymatic resolution, another common technique, requires highly specific conditions and often struggles with substrate tolerance, leading to inconsistent yields and extended reaction times. Furthermore, these traditional methods frequently generate significant waste streams and require complex downstream processing to remove biological catalysts or organic solvents, complicating the regulatory approval process for cost reduction in API manufacturing. The reliance on these less efficient techniques often results in supply chain bottlenecks and inflated production costs that are ultimately passed down to the healthcare system.
The Novel Approach
The methodology described in the patent offers a transformative alternative by employing classical resolution through diastereomeric salt formation, optimized for modern manufacturing standards. This novel approach involves reacting the racemic cis-configured oxathiolane compound with a chiral acid to generate two distinct diastereomeric salts with different physical properties, specifically solubility. By carefully controlling parameters such as solvent composition, temperature, and stoichiometry, one diastereomer can be selectively crystallized out of the solution with high purity. This physical separation bypasses the need for expensive chromatographic columns or sensitive enzymes, allowing for the use of standard crystallization and filtration equipment found in any multipurpose chemical plant. The process is versatile, accommodating various chiral acids like (1R)-(-)-10-camphorsulfonic acid, and allows for the recovery and recycling of the mother liquor to maximize overall yield. This strategic shift from separation-based to crystallization-based purification represents a significant leap forward in process chemistry, enabling the commercial scale-up of complex nucleoside analogs with greater economic predictability.
Mechanistic Insights into Chiral Resolution via Diastereomeric Salt Formation
The core mechanism driving this innovation lies in the thermodynamic differences between the diastereomeric salts formed during the reaction. When the racemic mixture of the cis-oxathiolane cytosine derivative interacts with an optically pure acid, such as (1R)-(-)-10-camphorsulfonic acid, it forms a pair of diastereomeric salts: one comprising the (-)-enantiomer of the base and the other comprising the (+)-enantiomer. Although these salts share similar chemical connectivity, their three-dimensional spatial arrangements differ, leading to distinct lattice energies and solubility profiles in specific solvent systems like isopropanol-water mixtures. The process exploits these solubility differences; under controlled cooling conditions, the less soluble diastereomer precipitates as a crystalline solid, while the more soluble counterpart remains in the supernatant. This selective crystallization is the key to achieving high optical purity, often exceeding 98% enantiomeric excess after recrystallization. The ability to tune the solvent ratio and temperature allows process chemists to manipulate the supersaturation point, ensuring that only the desired isomer nucleates and grows, effectively filtering out the unwanted enantiomer at the molecular level.
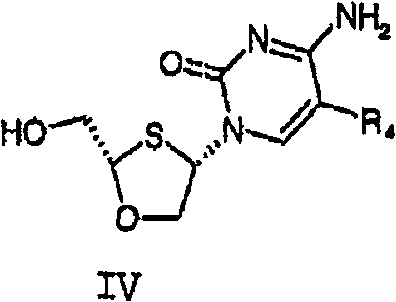
Furthermore, the mechanism includes a robust purification strategy for the final conversion step, ensuring that no trace of the chiral resolving agent contaminates the final product. After isolating the desired diastereomeric salt, it must be converted back to the free base form. The patent details the use of strong base anion exchange resins, such as Dowex Marathon A-OH, to facilitate this transformation. As the salt solution passes through the resin column, the chiral sulfonate anions are captured by the resin, releasing the free amine base into the eluent. This ion exchange mechanism is highly efficient and avoids the generation of inorganic salt byproducts that would typically result from neutralization with inorganic bases like sodium hydroxide. Consequently, the final workup is simplified to solvent removal and crystallization, drastically reducing the impurity burden. This mechanistic elegance ensures that the final high-purity antiviral intermediates meet stringent regulatory specifications for residual solvents and heavy metals, which is critical for downstream drug substance manufacturing.
How to Synthesize Optically Active Cis-Oxathiolane Cytosine Derivatives Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for implementing this resolution technology in a production environment. The process begins with the dissolution of the racemic cis-2-hydroxymethyl-4-(cytosine-1'-yl)-1,3-oxathiolane in a heated solvent mixture, typically isopropanol and water, to ensure complete solubility. A chiral acid, preferably (1R)-(-)-10-camphorsulfonic acid, is then added, either in stoichiometric or sub-stoichiometric amounts depending on the specific embodiment desired. The solution is subsequently cooled to induce crystallization of the target diastereomeric salt, which is then collected via filtration. Detailed standardized synthesis steps see the guide below.
- React the racemic cis-configured oxathiolane compound with a stoichiometric or sub-stoichiometric amount of a chiral acid, such as (1R)-(-)-10-camphorsulfonic acid, in a suitable solvent system like isopropanol and water.
- Cool the reaction mixture to induce crystallization of the less soluble diastereomeric salt, specifically targeting the desired enantiomer salt, and separate the solid via filtration.
- Convert the isolated diastereomeric salt into the free base optically active compound using an ion exchange resin or basic treatment, followed by final purification and drying.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this resolution technology translates into tangible strategic benefits that extend beyond mere technical feasibility. By eliminating the dependency on chiral HPLC columns and specialized enzymatic reactors, the process significantly lowers the barrier to entry for manufacturing these critical intermediates. The use of commodity chemicals like isopropanol, water, and camphorsulfonic acid ensures that raw material sourcing is stable and cost-effective, mitigating the risks associated with supply chain disruptions for exotic reagents. Moreover, the scalability of crystallization processes is well-understood in the chemical industry, allowing for seamless transition from pilot plant to full commercial production without the need for extensive re-engineering of equipment. This inherent scalability reduces lead time for high-purity oxathiolane derivatives, enabling faster response to market demands for antiviral medications. The robustness of the method also implies a more consistent quality profile, reducing the frequency of batch failures and the associated costs of reprocessing or disposal.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the replacement of expensive chromatographic separation with crystallization. Chiral HPLC requires costly stationary phases and large volumes of organic solvents, whereas crystallization utilizes inexpensive solvents and standard filtration hardware. Additionally, the ability to recover and recycle the chiral acid from the mother liquor or through ion exchange further diminishes the net consumption of resolving agents. This reduction in consumable costs, combined with higher throughput capabilities, leads to substantial cost savings in the overall production budget. The elimination of transition metal catalysts or complex biocatalysts also removes the need for expensive metal scavenging steps or enzyme stabilization protocols, simplifying the unit operations and reducing utility consumption.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of widely available raw materials and standard processing equipment. Unlike proprietary enzymatic processes that may rely on single-source suppliers for specific biocatalysts, the reagents used here, such as camphorsulfonic acid and common alcohols, are produced by multiple global vendors. This diversification of the supply base reduces the risk of shortages and price volatility. Furthermore, the process operates under mild conditions that do not require extreme pressures or temperatures, enhancing operational safety and reducing the likelihood of unplanned shutdowns due to equipment stress. The demonstrated success on a kilogram scale confirms that the process is not merely theoretical but is ready for immediate integration into existing manufacturing lines, ensuring continuity of supply for critical antiviral drugs.
- Scalability and Environmental Compliance: From an environmental and regulatory perspective, this method offers a greener alternative to traditional resolution techniques. The solvent systems employed, primarily alcohol and water mixtures, are easier to recover and recycle compared to the complex mobile phases used in chromatography. The avoidance of heavy metal catalysts aligns with increasingly strict guidelines regarding elemental impurities in pharmaceutical products, simplifying the regulatory filing process. The high selectivity of the crystallization process minimizes the generation of waste isomers, improving the atom economy of the synthesis. Waste streams are predominantly aqueous or alcoholic, which are more straightforward to treat in standard effluent treatment plants compared to halogenated or aromatic solvent wastes. This alignment with green chemistry principles not only reduces environmental impact but also lowers the costs associated with waste disposal and environmental compliance monitoring.

Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this resolution technology. These insights are derived directly from the experimental data and embodiments provided in the patent documentation, offering clarity on process parameters and expected outcomes. Understanding these details is crucial for technical teams evaluating the feasibility of adopting this route for their specific manufacturing needs. The answers reflect the proven capabilities of the method in achieving high purity and yield under defined conditions.
Q: What chiral resolving agents are most effective for this oxathiolane resolution?
A: The patent identifies (1R)-(-)-10-camphorsulfonic acid as a highly effective chiral selector, along with (-)-2,3-dibenzoyl-L-tartaric acid and (+)-L-tartaric acid, capable of achieving high enantiomeric excess through diastereomeric salt formation.
Q: Can this resolution process be scaled to industrial production volumes?
A: Yes, the methodology has been demonstrated on a kilogram scale (e.g., 193.7 kg batch size) using standard reactor equipment, proving its viability for commercial scale-up of complex pharmaceutical intermediates without requiring specialized chromatography.
Q: How is the chiral acid removed after the resolution step?
A: The process utilizes ion exchange resins, specifically strong base anion exchange resins like Dowex Marathon A-OH, to effectively remove the chiral sulfonic acid from the product solution, ensuring high purity of the final free base.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable cis-2-hydroxymethyl-4-(cytosine-1'-yl)-1,3-oxathiolane Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of securing a stable and high-quality supply of chiral intermediates for the global pharmaceutical market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the volumetric demands of major drug manufacturers. We are committed to delivering products that adhere to stringent purity specifications, utilizing rigorous QC labs to verify enantiomeric excess and impurity profiles for every batch. Our facility is equipped to handle the specific solvent systems and crystallization requirements outlined in advanced patents like CN101142211B, guaranteeing that the optically active oxathiolane nucleosides we supply are of the highest caliber.
We invite potential partners to engage with our technical procurement team to discuss how our capabilities align with your project requirements. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits of switching to our optimized manufacturing routes. We encourage you to contact us to obtain specific COA data and route feasibility assessments tailored to your development timeline. Let us collaborate to accelerate the delivery of life-saving antiviral therapies to patients worldwide through superior process chemistry and supply chain excellence.
