Advanced Synthesis of 2-Amino-5-Chlorobenzoic Acid Derivatives for Scalable Agrochemical Production
The chemical industry is constantly evolving to meet the rigorous demands of agrochemical production, particularly for high-value intermediates used in next-generation insecticides. Patent CN115181031A introduces a transformative preparation method for 2-amino-5-chloro-3-substituted benzoic acid derivatives, which serve as critical building blocks for bisamide insecticides such as chlorantraniliprole. This novel synthetic route addresses long-standing challenges in traditional manufacturing by replacing expensive and potentially hazardous chlorinating agents with sulfuryl chloride, enabling reactions to proceed under remarkably mild conditions. The significance of this technological breakthrough lies not only in its economic efficiency but also in its ability to suppress side reactions that have historically plagued the synthesis of these sensitive aromatic acids. By optimizing the reaction parameters to operate between 20°C and 60°C, the process effectively mitigates the risk of thermal decarboxylation, ensuring a cleaner impurity profile and superior yield compared to conventional heating methods. For global supply chains seeking reliable agrochemical intermediate suppliers, this patent represents a pivotal shift towards more sustainable and cost-effective manufacturing paradigms.
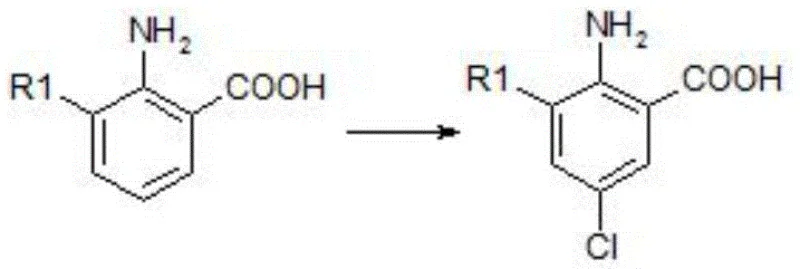
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-amino-5-chloro-3-substituted benzoic acid has relied heavily on the use of N-Chlorosuccinimide (NCS) as the primary chlorinating source, often necessitating the use of polar aprotic solvents like N,N-dimethylformamide under elevated heating conditions. While operationally straightforward, this legacy approach suffers from significant economic and technical drawbacks that hinder large-scale efficiency. The primary constraint is the prohibitively high cost of NCS, which drastically inflates the raw material expenditure for bulk production, making the final intermediate less competitive in price-sensitive agricultural markets. Furthermore, the requirement for heating during the chlorination step introduces a severe risk of thermal instability; specifically, the carboxyl group on the benzoic acid scaffold is prone to decarboxylation under thermal stress. This thermal degradation leads to the formation of undesirable dichloro-substituted by-products, which are difficult to separate and significantly reduce the overall yield of the target molecule. These inefficiencies create a bottleneck for procurement managers aiming for cost reduction in agrochemical intermediate manufacturing, as the purification burden increases and the atom economy decreases.
The Novel Approach
In stark contrast to the thermal limitations of prior art, the methodology disclosed in CN115181031A utilizes sulfuryl chloride ($SO_2Cl_2$) as a highly efficient and economical chlorinating agent, facilitating the reaction in common organic solvents such as chloroform or dichloromethane. This strategic substitution allows the chlorination to occur at near-ambient temperatures, typically ranging from 20°C to 60°C, with a preferred operating point around 30°C. By eliminating the need for aggressive heating, the new process effectively preserves the integrity of the carboxyl functionality, preventing the decarboxylation side reactions that plague older methods. The result is a dramatic improvement in selectivity, where the chlorine atom is introduced precisely at the 5-position of the aromatic ring without compromising the rest of the molecular structure. Additionally, the subsequent saponification step is conducted under similarly mild alkaline conditions, further ensuring that the final acid product is obtained with high purity and minimal degradation. This holistic optimization of reaction conditions translates directly into a robust, high-yield process that is ideally suited for the commercial scale-up of complex agrochemical intermediates.
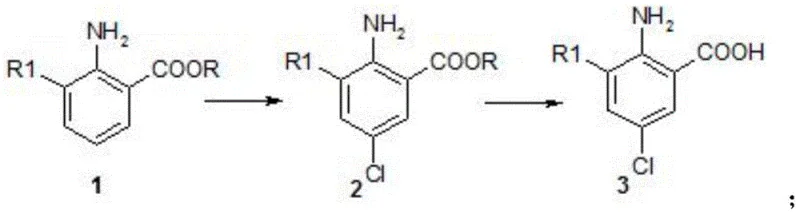
Mechanistic Insights into Sulfuryl Chloride-Mediated Electrophilic Substitution
The core of this innovative synthesis lies in the mechanism of electrophilic aromatic substitution facilitated by sulfuryl chloride, which acts as a source of electrophilic chlorine species under mild conditions. In the presence of the electron-donating amino group at the 2-position, the aromatic ring becomes activated towards electrophilic attack, directing the incoming chlorine atom primarily to the para-position relative to the amino group, which corresponds to the 5-position on the benzoic acid scaffold. The use of sulfuryl chloride is particularly advantageous because it generates the necessary electrophile without the need for strong Lewis acid catalysts or high thermal energy, which could otherwise promote non-selective radical chlorination or decomposition. The reaction proceeds through a sigma-complex intermediate that is stabilized by the resonance effects of the adjacent substituents, ensuring high regioselectivity for the 5-chloro isomer. This mechanistic precision is critical for R&D directors focused on purity and impurity spectra, as it minimizes the formation of ortho-chlorinated or poly-chlorinated impurities that are common in less controlled radical processes. Furthermore, the choice of solvent plays a pivotal role in stabilizing the transition state; solvents like chloroform provide an ideal medium that dissolves both the organic substrate and the inorganic chlorinating agent while maintaining a low dielectric constant that favors the desired ionic mechanism over radical pathways.
Following the chlorination, the conversion of the ester intermediate to the final carboxylic acid via saponification is a classic nucleophilic acyl substitution, yet it is executed here with specific attention to preserving the newly installed chlorine atom and the sensitive amino group. The process employs aqueous alkali solutions, such as sodium hydroxide or potassium hydroxide, in alcohol solvents like methanol or ethanol, creating a homogeneous phase that accelerates the hydrolysis rate without requiring excessive heat. The mild temperature range of 20°C to 70°C ensures that the amino group remains intact and does not undergo unwanted oxidation or displacement, which can occur under harsher basic conditions at elevated temperatures. Upon completion of the saponification, the reaction mixture is carefully acidified to a pH of 3-4, causing the target 2-amino-5-chloro-3-substituted benzoic acid to precipitate out of the solution in high purity. This controlled precipitation serves as an inherent purification step, effectively excluding soluble inorganic salts and residual organic impurities, thereby delivering a product that meets stringent quality specifications required for downstream pesticide synthesis without the need for extensive chromatographic purification.
How to Synthesize 2-Amino-5-Chloro-3-Substituted Benzoic Acid Efficiently
The practical implementation of this synthesis involves a streamlined two-stage protocol that begins with the precise chlorination of the starting benzoate ester, followed by a straightforward hydrolysis to yield the final acid. The process is designed to be operationally simple, utilizing standard laboratory or plant equipment without the need for specialized high-pressure reactors or cryogenic cooling systems. Detailed standardized synthetic steps, including specific molar ratios, solvent volumes, and workup procedures derived from the patent examples, are outlined below to guide process chemists in replicating these high-yielding results.
- Perform chlorination of 2-amino-3-substituted benzoate with sulfuryl chloride in an organic solvent at 20-60°C.
- Isolate the intermediate 2-amino-5-chloro-3-substituted benzoate via extraction and solvent removal.
- Conduct saponification of the ester intermediate with alkali solution, followed by acidification to obtain the final acid product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this patented methodology offers substantial strategic advantages that extend far beyond simple chemical yield improvements. The shift from expensive reagents like NCS to commodity chemicals like sulfuryl chloride fundamentally alters the cost structure of the intermediate, driving down the bill of materials significantly. Moreover, the elimination of high-temperature processing steps reduces energy consumption and lowers the operational burden on manufacturing facilities, allowing for faster batch turnover times. These factors combine to create a more resilient supply chain capable of meeting fluctuating market demands for agrochemical active ingredients with greater agility and cost efficiency.
- Cost Reduction in Manufacturing: The replacement of N-Chlorosuccinimide with sulfuryl chloride represents a massive reduction in raw material costs, as sulfuryl chloride is a widely available bulk chemical with a fraction of the price point of specialized chlorinating agents. Additionally, the ability to run the reaction at near-ambient temperatures eliminates the energy costs associated with prolonged heating and the capital expenditure required for high-temperature reactor maintenance. The high selectivity of the reaction also means that less material is lost to by-products, effectively increasing the mass balance and reducing the cost per kilogram of the final purified product. This comprehensive approach to cost optimization ensures that manufacturers can maintain healthy margins even in competitive pricing environments.
- Enhanced Supply Chain Reliability: By relying on readily available solvents like chloroform, methanol, and ethyl acetate, the process minimizes the risk of supply disruptions caused by niche reagent shortages. The robustness of the reaction conditions means that production can be scaled up rapidly across different manufacturing sites without the need for highly specialized equipment or extensive operator training. This flexibility is crucial for supply chain heads who need to ensure continuous availability of key intermediates like 2-amino-5-chloro-3-methylbenzoic acid to support the uninterrupted production of finished insecticides. The simplified workup procedure, which involves standard extraction and crystallization, further reduces the turnaround time between batches, enhancing overall throughput.
- Scalability and Environmental Compliance: The mild nature of the chemistry inherently reduces the generation of hazardous waste, as there is less thermal degradation and fewer side reactions to manage during wastewater treatment. The process avoids the use of highly toxic oxidants and strong acids in the initial steps, aligning with modern green chemistry principles and simplifying regulatory compliance for environmental discharge. From a scalability perspective, the exothermic nature of the chlorination is easily managed at 30°C, preventing thermal runaways that can be dangerous on a multi-ton scale. This safety profile, combined with the high yields demonstrated in the patent examples, makes the technology an ideal candidate for transfer from pilot plant to full commercial production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route, drawing directly from the experimental data and beneficial effects described in the patent documentation. These insights are intended to clarify the operational benefits and chemical rationale for stakeholders evaluating this technology for potential adoption or sourcing.
Q: Why is sulfuryl chloride preferred over NCS for this synthesis?
A: Sulfuryl chloride is significantly more cost-effective than N-Chlorosuccinimide (NCS) and allows the reaction to proceed at much lower temperatures (20-60°C), minimizing thermal degradation and decarboxylation side reactions.
Q: How does this method improve impurity profiles?
A: By avoiding high-temperature heating required in prior art methods, this process prevents the decarboxylation of the carboxyl group, thereby drastically reducing the formation of dichloro-substituted by-products and improving overall yield.
Q: Is this process suitable for large-scale industrial manufacturing?
A: Yes, the use of common organic solvents like chloroform and mild reaction conditions (near room temperature) makes the process highly scalable, safe, and compatible with standard industrial reactor setups without requiring specialized high-pressure or high-temperature equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Amino-5-Chlorobenzoic Acid Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the global agrochemical supply chain and have positioned ourselves as a leader in the commercialization of advanced synthetic routes like the one described in CN115181031A. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive consistent quality regardless of order volume. We operate stringent purity specifications and maintain rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify every batch against the highest industry standards. Our commitment to technical excellence allows us to deliver 2-amino-5-chlorobenzoic acid derivatives that are free from the decarboxylation by-products often found in conventionally sourced materials.
We invite international partners to engage with our technical procurement team to discuss how this optimized synthesis can benefit your specific production requirements. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic impact of switching to this superior manufacturing method. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your project timelines, ensuring a seamless integration of our high-performance intermediates into your value chain.
