Scalable Asymmetric Synthesis of (+)-γ-Lycorane for Pharmaceutical Applications
The pharmaceutical industry continues to seek efficient pathways for synthesizing complex alkaloids, particularly those within the Amaryllidaceae family, due to their potent biological activities ranging from anti-tumor to anti-viral properties. Patent CN111087405B discloses a groundbreaking method for the asymmetric synthesis of (+)-γ-lycorane, a key scaffold in this class of bioactive molecules. This innovation addresses long-standing challenges in the field by providing a concise, six-step route that begins with a readily available chiral lactone compound. Unlike traditional methods that often rely on tedious racemic synthesis followed by difficult resolution, this approach leverages the inherent chirality of the starting material to ensure high optical purity throughout the sequence. The strategic design of this pathway not only streamlines the production of (+)-γ-lycorane but also establishes a versatile platform for synthesizing related lycorine-type alkaloids, offering significant value for drug discovery and development programs targeting oncology and infectious diseases.
For R&D directors evaluating new supply chains, the structural integrity and purity of the intermediate are paramount. This patented methodology ensures that the critical stereocenters are preserved through careful selection of reaction conditions, such as the mild deprotection in the initial step and the controlled radical processes in the subsequent elimination. The ability to access high-purity (+)-γ-lycorane without the baggage of racemic impurities simplifies downstream processing and regulatory filing. Furthermore, the route demonstrates excellent functional group tolerance, allowing for potential derivatization at later stages if structure-activity relationship (SAR) studies require analogues. This technical robustness makes the process highly attractive for integration into existing medicinal chemistry pipelines where reliability and reproducibility are non-negotiable requirements for advancing candidates into preclinical and clinical phases.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Amaryllidaceae alkaloids has been plagued by inefficiencies inherent in racemic strategies. Conventional approaches typically involve constructing the core skeleton without stereocontrol, resulting in a 50:50 mixture of enantiomers that must then be separated. This resolution step is notoriously wasteful, theoretically discarding half of the synthesized material, and often requires expensive chiral chromatography or multiple recrystallizations to achieve acceptable enantiomeric excess. Additionally, many existing asymmetric methods rely on precious metal catalysts with specialized chiral ligands, which drive up the cost of goods and introduce risks of heavy metal contamination that must be meticulously managed to meet strict pharmaceutical standards. The cumulative effect of these factors is a prolonged development timeline and inflated production costs, creating a bottleneck for the commercial viability of lycorine-based therapeutics.
The Novel Approach
The novel approach detailed in the patent circumvents these hurdles by employing a chiral pool strategy that fundamentally alters the economic and technical landscape of production. By initiating the synthesis with a chiral lactone, the method bypasses the need for external asymmetric induction or post-synthetic resolution, effectively locking in the desired stereochemistry from the outset. The six-step sequence is remarkably direct, minimizing unit operations and solvent consumption while maximizing atom economy. Key transformations, such as the microwave-assisted elimination and the palladium-catalyzed cyclization, are optimized for high yield and selectivity, ensuring that the process remains efficient even as scale increases. This streamlined workflow not only accelerates the time-to-market for research quantities but also lays a solid foundation for industrial-scale manufacturing, offering a clear competitive advantage over legacy synthetic routes.
Mechanistic Insights into the Multi-Step Cascade Synthesis
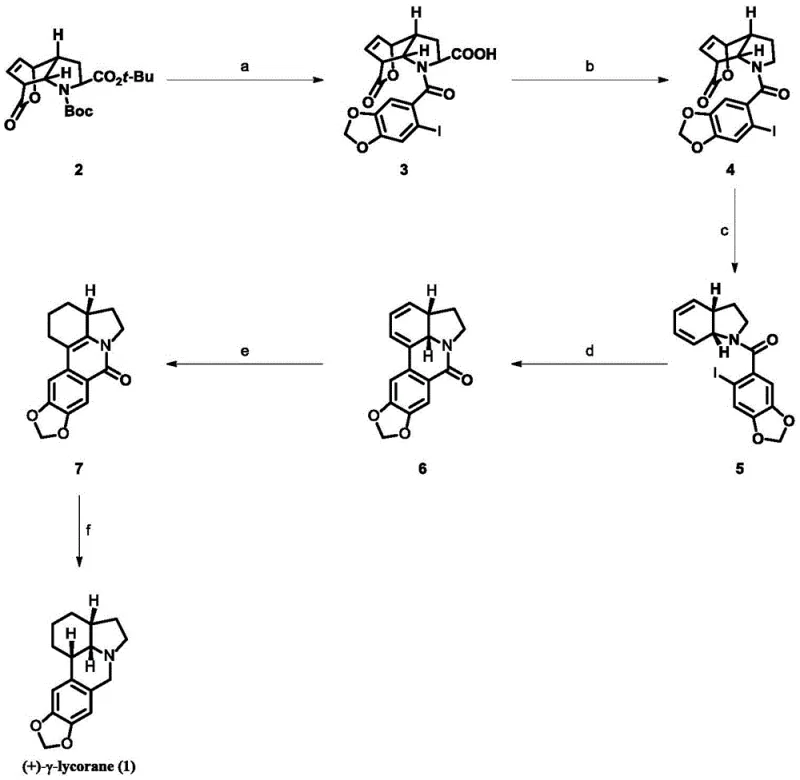
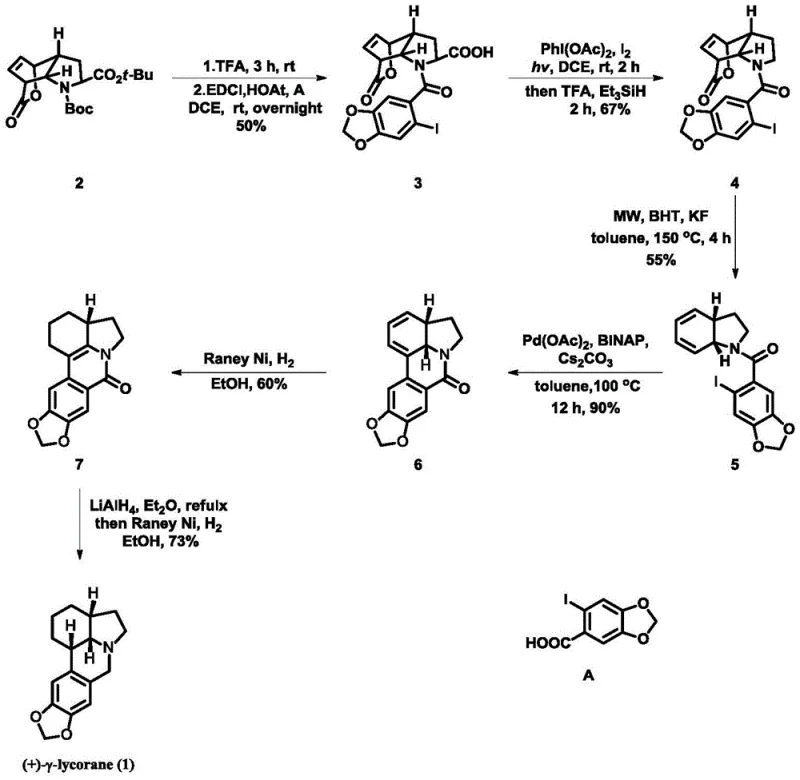
The chemical elegance of this synthesis lies in its orchestrated sequence of bond-forming and bond-breaking events that construct the complex tetracyclic core of (+)-γ-lycorane. The process begins with a condensation reaction where the chiral lactone is coupled with 2-iodopiperonyl benzoic acid, establishing the crucial amide linkage that serves as the tether for subsequent cyclizations. Following this, a photo-induced decarboxylative elimination generates a reactive iminium intermediate, which is immediately trapped by a hydride source to set the stage for ring closure. This radical-mediated step is particularly noteworthy for its ability to forge carbon-carbon bonds under mild conditions, avoiding the harsh reagents that often degrade sensitive alkaloid scaffolds. The precision required in controlling these reactive intermediates underscores the sophistication of the protocol, ensuring that side reactions are minimized and the desired pathway is favored thermodynamically and kinetically.
Further mechanistic refinement is observed in the microwave-assisted elimination step, which facilitates the formation of the dihydrophenanthridine core with high efficiency. The use of potassium fluoride and an antioxidant like BHT in this stage is critical for suppressing polymerization and oxidative degradation, common pitfalls in high-temperature organic synthesis. Subsequent palladium-catalyzed intramolecular coupling closes the final ring system, leveraging the power of transition metal catalysis to create the rigid architecture characteristic of lycorane alkaloids. The final reduction stages utilize Raney Nickel, a cost-effective heterogeneous catalyst, to saturate the olefinic bonds and reduce the amide to the amine, completing the transformation to the target alkaloid. This comprehensive understanding of the reaction mechanism allows for precise troubleshooting and optimization, ensuring consistent quality across batches.
How to Synthesize (+)-γ-Lycorane Efficiently
Executing this synthesis requires strict adherence to the optimized conditions outlined in the patent to ensure maximum yield and purity. The process is divided into distinct operational phases, beginning with the preparation of the amide precursor and culminating in the final reductive amination. Each step has been fine-tuned regarding temperature, solvent choice, and stoichiometry to mitigate risks associated with scale-up. For instance, the photochemical step requires specific wavelength irradiation to proceed efficiently, while the microwave step demands precise temperature control to prevent decomposition. Operators must be trained to handle reagents like lithium aluminum hydride and Raney Nickel with appropriate safety protocols, as these materials pose specific hazards if mishandled. The following guide summarizes the critical operational milestones necessary to replicate this high-performance synthesis in a GMP-compliant environment.
- Deprotect the starting lactone with TFA and couple with 2-iodopiperonyl benzoic acid using EDCI/HOAt to form the amide intermediate.
- Perform decarboxylative elimination using PhI(OAc)2 and I2 under light irradiation, followed by reduction with Et3SiH.
- Execute microwave-assisted elimination in toluene with KF and BHT to construct the core ring system.
- Conduct Pd-catalyzed intramolecular cyclization using BINAP and Cs2CO3 to close the phenanthridine ring.
- Reduce the double bond using Raney Nickel and hydrogen, followed by final reduction with LiAlH4 and hydrogenation to yield (+)-γ-lycorane.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, this synthetic route offers compelling advantages that directly translate to improved margins and supply security. The reliance on cheap and easily obtained raw materials, such as the starting lactone and commodity reagents like trifluoroacetic acid and iodine, insulates the supply chain from the volatility associated with exotic or scarce chemicals. By eliminating the need for expensive chiral catalysts and complex resolution columns, the process significantly reduces the direct material costs per kilogram of product. Furthermore, the shortened six-step sequence minimizes the accumulation of work-in-progress inventory and reduces the overall cycle time from raw material intake to finished goods. This efficiency allows for more responsive production scheduling, enabling suppliers to react quickly to fluctuations in demand from pharmaceutical partners without maintaining excessive safety stock levels.
- Cost Reduction in Manufacturing: The economic benefits of this route are driven by the strategic avoidance of costly chiral resolution steps and precious metal catalysts typically required for asymmetric induction. By utilizing a chiral pool starting material, the process eliminates the theoretical 50% yield loss associated with racemic synthesis, effectively doubling the material efficiency of the upstream feedstock. Additionally, the substitution of expensive homogeneous catalysts with heterogeneous Raney Nickel in the final reduction steps lowers catalyst procurement costs and simplifies the removal of metal residues, reducing downstream purification expenses. These cumulative savings result in a substantially lower cost of goods sold, making the final API intermediate more price-competitive in the global market.
- Enhanced Supply Chain Reliability: Supply continuity is bolstered by the use of robust, commercially available reagents that are not subject to the same geopolitical or logistical constraints as specialized fine chemicals. The synthetic pathway is designed to be operationally simple, reducing the risk of batch failures due to procedural complexity or sensitivity to minor environmental variations. This reliability ensures a steady flow of high-purity intermediates to downstream manufacturers, mitigating the risk of production stoppages that can derail clinical trial timelines. The scalability of key steps, such as the microwave-assisted reaction which can be adapted to continuous flow processing, further enhances the ability to ramp up production volumes rapidly when commercial demand surges.
- Scalability and Environmental Compliance: The process aligns well with modern green chemistry principles by minimizing waste generation and solvent usage through its concise step count. The high yields observed in critical coupling and cyclization steps mean that less raw material is wasted as byproducts, reducing the burden on waste treatment facilities. Moreover, the avoidance of toxic heavy metals in favor of nickel-based systems simplifies the environmental compliance profile of the manufacturing site. The ability to scale this process from gram to multi-kilogram quantities without significant re-engineering demonstrates its maturity and readiness for industrial adoption, providing a sustainable long-term solution for the production of Amaryllidaceae alkaloids.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. Understanding these details is crucial for stakeholders evaluating the feasibility of integrating this intermediate into their supply chain. The answers are derived directly from the technical specifications and experimental data provided in the patent documentation, ensuring accuracy and relevance for decision-makers.
Q: What are the advantages of this asymmetric synthesis method over traditional racemic routes?
A: This method avoids the need for complex chiral resolution steps required in racemic synthesis. By starting from a chiral lactone pool, it maintains stereochemistry throughout the six-step sequence, significantly simplifying the process and improving overall optical purity without expensive chiral catalysts.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the process utilizes readily available raw materials and avoids extremely sensitive reagents. Key steps like the microwave-assisted elimination and Raney Nickel hydrogenation are amenable to scale-up, providing a robust pathway for manufacturing multi-kilogram quantities of high-purity intermediates.
Q: How does this route impact the cost of goods for Amaryllidaceae alkaloids?
A: The route reduces costs by shortening the synthetic sequence to only six steps and eliminating the need for precious metal asymmetric catalysts. The use of cost-effective reagents like Raney Nickel for hydrogenation further drives down manufacturing expenses compared to traditional methods.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (+)-γ-Lycorane Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of securing a stable supply of high-quality alkaloid intermediates for your drug development programs. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements whether you are in early-stage research or late-stage clinical trials. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of (+)-γ-lycorane meets the highest international standards. Our commitment to technical excellence means we can navigate the complexities of this six-step synthesis with precision, delivering material that is ready for immediate use in your downstream processes.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can optimize your project economics. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the potential financial benefits of switching to this more efficient supply source. We are prepared to provide specific COA data and route feasibility assessments tailored to your unique project needs, ensuring full transparency and confidence in our partnership. Contact us today to secure a reliable supply of this critical pharmaceutical intermediate and accelerate your path to market.
