Scalable Synthesis of Ethyl 24-Cholenoate for High-Purity Vitamin D3 Metabolite Manufacturing
Introduction to Advanced Steroid Intermediate Synthesis
The pharmaceutical industry's demand for high-purity Vitamin D3 metabolites, specifically 25-hydroxycholesterol, necessitates efficient access to key precursors like ethyl 24-cholenoate. Patent CN111320664B discloses a groundbreaking preparation method that addresses longstanding inefficiencies in steroid side-chain modification. This technology represents a significant leap forward for any reliable pharmaceutical intermediate supplier aiming to optimize their portfolio of hormonal metabolites. By shifting away from multi-step degradations and complex olefinations, this novel route offers a streamlined pathway that enhances both economic viability and operational safety. The strategic implementation of nickel-catalyzed reductive coupling allows for the direct construction of the C24 side chain with exceptional fidelity. This report analyzes the technical merits and commercial implications of adopting this methodology for large-scale API intermediate production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of ethyl 24-cholenoate has been plagued by prohibitive complexity and poor atom economy. One prominent prior art route utilizes lithocholic acid as the starting material, necessitating a tedious 11-step sequence to reach the target molecule. As illustrated in the reaction scheme below, this pathway suffers from a dismal total yield of merely 11.13%, rendering it economically unfeasible for modern manufacturing standards. Furthermore, the reliance on expensive experimental reagents and the accumulation of impurities over such a long synthetic linear sequence create significant bottlenecks for supply chain continuity. Another conventional approach employs dehydroepiandrosterone (DHEA) derivatives but relies heavily on Wittig reactions.
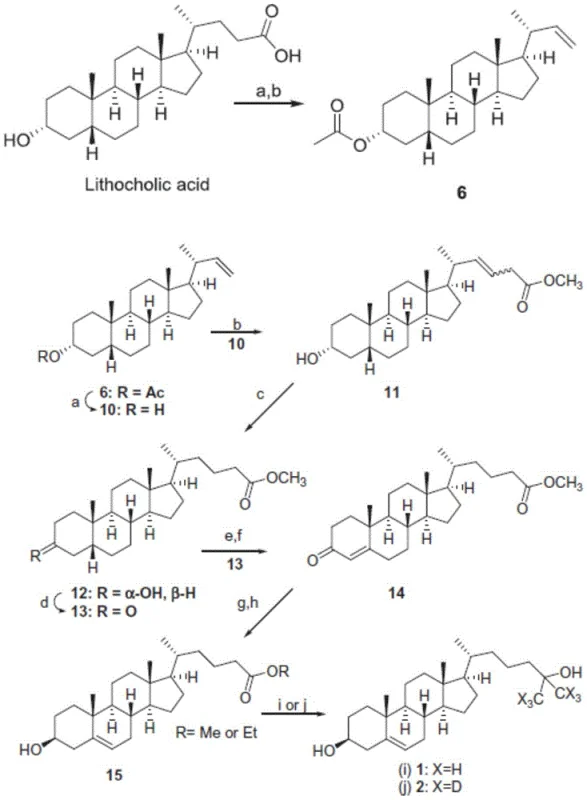
These Wittig protocols demand stringent anhydrous and anaerobic conditions, often requiring cryogenic temperatures as low as -78°C. Such harsh operating parameters drastically increase energy consumption and require specialized reactor infrastructure, which limits the ability for commercial scale-up of complex steroid intermediates. The combination of low overall throughput and high operational expenditure makes these legacy methods unsuitable for meeting the growing global demand for Vitamin D3 analogs.
The Novel Approach
In stark contrast, the methodology described in CN111320664B introduces a concise two-step strategy that fundamentally reshapes the production landscape. The core innovation lies in the transformation of an iodo-steroid precursor (Compound 02) directly into the esterified side-chain intermediate (Compound 03) via a zinc-mediated alkylation. This is followed by a mild acid-catalyzed elimination to yield the final ethyl 24-cholenoate (Compound 04). The reaction scheme below highlights this elegant simplification, bypassing the need for protecting group manipulations and cryogenic olefinations.
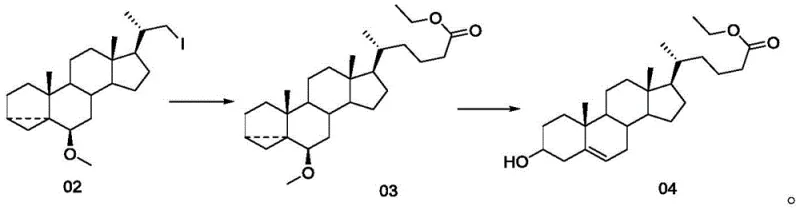
This novel approach achieves a remarkable two-step total yield ranging from 80% to 91%, representing a nearly eight-fold improvement over the lithocholic acid route. By utilizing inexpensive reagents such as zinc powder, nickel chloride hexahydrate, and ethyl acrylate, the process significantly lowers the raw material cost basis. Moreover, the reaction conditions are exceptionally mild, proceeding effectively between 0°C and 80°C, which eliminates the need for energy-intensive cooling systems. This robustness makes the technology ideally suited for reliable agrochemical intermediate supplier networks and pharma manufacturers alike who prioritize process stability.
Mechanistic Insights into Nickel-Catalyzed Reductive Alkylation
The heart of this synthetic breakthrough is the nickel-catalyzed reductive coupling between the steroidal iodide and ethyl acrylate. Mechanistically, this transformation likely proceeds through the generation of an organozinc species in situ, which subsequently undergoes transmetallation with the nickel catalyst. The resulting organonickel intermediate facilitates the insertion of the acrylate double bond, effectively extending the steroid side chain by three carbons in a single operation. This catalytic cycle is highly tolerant of the complex steric environment presented by the steroid backbone, ensuring high regioselectivity. The use of pyridine as both solvent and base plays a crucial role in stabilizing the reactive intermediates and solubilizing the zinc salts. Understanding this mechanism is vital for R&D directors focused on purity and杂质谱 (impurity profile) control, as it minimizes side reactions common in radical-based alkylations.
Following the alkylation, the conversion to the final alkene involves an acid-catalyzed elimination reaction. In the presence of p-toluenesulfonic acid or methanesulfonic acid within a water-dioxane system, the methoxy group at the C3 position (or adjacent functionality depending on the specific precursor protection) is removed, and the double bond at the C24 position is established. This step is critical for generating the correct unsaturation pattern required for downstream metabolic activity. The aqueous workup conditions are particularly advantageous as they facilitate the removal of metal residues and polar byproducts, leading to a crude product with high HPLC purity (often exceeding 96.5%). This inherent cleanliness reduces the burden on downstream purification units, directly impacting the cost reduction in pharmaceutical intermediate manufacturing.
How to Synthesize Ethyl 24-Cholenoate Efficiently
Implementing this synthesis requires precise control over stoichiometry and temperature to maximize the benefits of the nickel-zinc catalytic system. The process begins with the activation of zinc powder in the presence of the nickel catalyst and ethyl acrylate within a pyridine medium. Once the catalytic species are generated, the iodo-steroid precursor is introduced dropwise to maintain thermal control and prevent exothermic runaway. Detailed standard operating procedures regarding reagent grades, addition rates, and quenching protocols are essential for reproducibility. For the complete standardized synthesis steps and specific parameter optimization, please refer to the guide below.
- Perform nickel-catalyzed reductive alkylation of the iodo-steroid precursor with ethyl acrylate using zinc powder in pyridine.
- Isolate the alkylated intermediate (Compound 03) via filtration and column chromatography purification.
- Execute acid-catalyzed elimination in aqueous dioxane to generate the final ethyl 24-cholenoate product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthesis route offers transformative benefits that extend beyond simple yield improvements. The elimination of cryogenic operations and sensitive anhydrous requirements drastically simplifies the facility requirements, allowing production to occur in standard glass-lined or stainless steel reactors without specialized low-temperature capabilities. This flexibility enhances supply chain reliability by reducing dependency on niche processing units that often become bottlenecks during peak demand periods. Furthermore, the shift from expensive phosphorus-based reagents to commodity chemicals like zinc and acrylic esters creates a more resilient raw material supply base that is less susceptible to market volatility.
- Cost Reduction in Manufacturing: The economic impact of this process is driven primarily by the drastic simplification of the synthetic route. By reducing the step count from 11 down to 2, the cumulative loss of material is minimized, and labor costs are significantly reduced. The replacement of costly Wittig reagents with inexpensive zinc and nickel salts removes a major cost driver from the bill of materials. Additionally, the high yield means less raw material is required per kilogram of finished product, leading to substantial cost savings in the overall production budget without compromising quality standards.
- Enhanced Supply Chain Reliability: Operational simplicity translates directly to supply security. The mild reaction conditions (0°C to 80°C) and the use of stable, commercially available solvents like pyridine and dioxane ensure that production can be maintained consistently throughout the year. There is no risk of campaign delays due to the failure of cryogenic cooling systems or the unavailability of specialized anhydrous solvents. This robustness ensures reducing lead time for high-purity pharmaceutical intermediates, allowing manufacturers to respond more agilely to market fluctuations and urgent customer orders.
- Scalability and Environmental Compliance: From an environmental and scaling perspective, this route is superior. The avoidance of stoichiometric phosphorus waste, which is typical of Wittig reactions, simplifies wastewater treatment and reduces the environmental footprint of the manufacturing process. The use of zinc, while requiring proper disposal, is well-understood in industrial waste streams. The high atom economy and the ability to run reactions at higher concentrations contribute to better space-time yields, facilitating the commercial scale-up of complex steroid intermediates from pilot plant to multi-ton production with minimal engineering hurdles.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation. They are designed to assist technical teams in evaluating the feasibility of integrating this route into their existing manufacturing frameworks. Understanding these nuances is key to leveraging the full potential of this innovative synthesis method.
Q: What are the key advantages of this synthesis over traditional Wittig routes?
A: This method eliminates the need for cryogenic conditions (-78°C) and expensive phosphorus ylides required in Wittig reactions, utilizing mild temperatures (0-80°C) and abundant zinc/nickel catalysts instead.
Q: What is the expected yield for the two-step process?
A: The patent data indicates a robust two-step total yield ranging from 80% to 91%, significantly outperforming the 11.13% yield of older lithocholic acid-based routes.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the process uses commercially available reagents, avoids sensitive anhydrous operations, and employs standard equipment, making it highly scalable for industrial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ethyl 24-Cholenoate Supplier
As the global demand for Vitamin D3 metabolites continues to rise, securing a stable source of high-quality intermediates is paramount. NINGBO INNO PHARMCHEM stands ready to support your development and production needs with our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our commitment to stringent purity specifications and rigorous QC labs ensures that every batch of ethyl 24-cholenoate meets the highest international standards. We understand the critical nature of API intermediates in the drug development timeline and are dedicated to providing consistent, high-performance materials.
We invite you to collaborate with us to explore how this advanced synthesis route can optimize your supply chain. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our capabilities align with your strategic goals for cost-effective and sustainable manufacturing.
