Revolutionizing Trenbolone Acetate Production with Advanced Cytochrome P450 BM3 Mutants
The pharmaceutical and veterinary industries are constantly seeking more efficient pathways for the synthesis of high-value anabolic steroids, particularly Trenbolone Acetate, a critical compound known for its potent muscle-building properties. A significant breakthrough in this domain is documented in Chinese Patent CN113528472B, which discloses a novel method utilizing engineered Cytochrome P450 BM3 mutants. This technology represents a paradigm shift from traditional multi-step chemical synthesis to a streamlined bio-chemical hybrid approach. By leveraging specific amino acid substitutions in the P450 BM3 enzyme structure, this innovation achieves unprecedented regioselectivity in the 11α-hydroxylation of steroid precursors. For global procurement leaders and R&D directors, understanding the implications of this patent is vital, as it offers a potential solution to long-standing challenges regarding yield, purity, and environmental impact in steroid manufacturing. The ability to directly functionalize the steroid nucleus at the 11α-position with high fidelity opens new doors for cost-effective production scales.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of Trenbolone Acetate has been plagued by cumbersome synthetic routes that rely heavily on harsh chemical reagents and extensive protection-deprotection strategies. Traditional methods often start with 4,9(11)-diene-androsta-3,17-dione, requiring the protection of the 3-position carbonyl group using corrosive catalysts like p-toluenesulfonic acid or acetyl chloride. These processes are not only operationally complex, involving multiple solvent swaps and aqueous washes, but they also generate significant amounts of hazardous waste that is difficult to treat. Furthermore, the overall yield of these conventional chemical pathways is often suboptimal, frequently hovering around 63%, due to the formation of numerous by-products during reduction, dehydrogenation, and hydrolysis steps. The reliance on stoichiometric oxidants and the difficulty in separating closely related steroid impurities further exacerbate the cost and environmental burden, making the supply chain vulnerable to regulatory scrutiny and raw material price volatility.
The Novel Approach
In stark contrast, the methodology outlined in the patent introduces a sophisticated biocatalytic strategy that fundamentally simplifies the synthetic architecture. By employing engineered E. coli cells co-expressing specific P450 BM3 mutants and 17β-hydroxysteroid dehydrogenase (17β-HSD), the process converts inexpensive and readily available estro-4,9-diene-3,17-dione directly into 11α-hydroxymethyldienolone in a single enzymatic cascade. This approach effectively bypasses the need for tedious protection groups and harsh chemical oxidation steps. The integration of a glucose dehydrogenase (GDH) system for in-situ cofactor regeneration ensures that the reaction remains economically viable by recycling NADPH continuously. Subsequent chemical steps involve a mild dehydration and a final esterification, resulting in a significantly shorter process flow. This hybrid model not only enhances the overall mass balance but also aligns with modern green chemistry principles, offering a robust alternative for the commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into P450 BM3-Mediated 11α-Hydroxylation
The core of this technological advancement lies in the precise protein engineering of the Cytochrome P450 BM3 enzyme. The patent highlights specific mutants, namely LG-23/T438S, LG-23/T438A, and LG-23/T438G, where the threonine at position 438 is substituted with serine, alanine, or glycine, respectively. These mutations are not arbitrary; they are strategically designed to reshape the enzyme's active site pocket to accommodate the bulky steroid substrate in a specific orientation that favors 11α-hydroxylation over other potential oxidation sites. The steric and electronic changes introduced by these amino acid swaps enhance the binding affinity and catalytic turnover for substrates like 4,9-diene-3,17-dione and methyl dienolone. Experimental data indicates that the LG-23/T438S mutant, in particular, achieves a remarkable 11α-hydroxylation selectivity of up to 92% for methyl dienolone, a significant improvement over the parent LG-23 strain. This high selectivity is crucial for minimizing downstream purification costs and ensuring the final API meets stringent pharmacopeial standards.
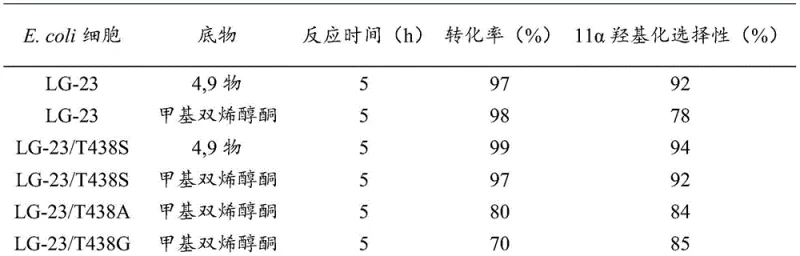
Beyond the primary hydroxylation event, the mechanism incorporates a synergistic dual-enzyme system. The co-expression of 17β-HSD allows for the simultaneous reduction of the 17-ketone group, effectively performing two distinct chemical transformations in one bioreactor vessel. This tandem catalysis is supported by a robust cofactor regeneration loop driven by GDH, which oxidizes glucose to gluconolactone while reducing NADP+ back to NADPH. This closed-loop system eliminates the need for expensive external addition of cofactors, which is typically a major cost driver in biocatalysis. The result is a highly efficient, self-sustaining catalytic cycle that operates under mild physiological conditions (20-30°C, pH 8.0), preserving the integrity of the sensitive steroid skeleton while introducing the critical oxygen functionality required for biological activity. Such mechanistic elegance translates directly into process reliability and reproducibility at an industrial scale.
How to Synthesize Trenbolone Acetate Efficiently
Implementing this novel synthesis route requires a disciplined approach to bioprocess optimization followed by precise chemical finishing. The process begins with the cultivation of the genetically engineered E. coli strain harboring the co-expression plasmids for the P450 mutant and 17β-HSD. Once the biomass reaches the optimal optical density, the biotransformation is initiated by feeding the steroid substrate dissolved in a co-solvent like DMF, along with glucose and the GDH enzyme preparation. The reaction proceeds until the substrate is fully consumed, typically within a few hours, yielding the 11α-hydroxy intermediate which is then extracted. The subsequent chemical steps involve a controlled dehydration using p-toluenesulfonic acid and a final acetylation. For detailed operational parameters and safety guidelines, please refer to the standardized protocol below.
- Biocatalytic Hydroxylation: Utilize engineered E. coli co-expressing P450 BM3 mutant (e.g., LG-23/T438S) and 17β-HSD to convert 4,9-diene-3,17-dione into 11α-hydroxymethyldienolone with cofactor regeneration.
- Chemical Dehydration: React the crude 11α-hydroxymethyldienolone with p-toluenesulfonic acid monohydrate in chloroform to effect dehydration and form the Trenbolone backbone.
- Esterification: Perform final acetylation of Trenbolone using acetic anhydride and DMAP in dichloromethane to yield the final Trenbolone Acetate product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this P450 BM3-mediated pathway offers compelling strategic advantages that extend beyond mere technical novelty. The primary value proposition lies in the drastic simplification of the supply chain and the reduction of operational risks associated with hazardous chemical handling. By replacing multiple chemical steps with a single biocatalytic fermentation, manufacturers can significantly reduce the consumption of organic solvents and aggressive reagents, leading to a smaller environmental footprint and lower waste disposal costs. This alignment with green chemistry standards not only mitigates regulatory compliance risks but also enhances the brand reputation of the final product in increasingly eco-conscious markets. Furthermore, the use of fermentation-based production leverages well-established infrastructure, allowing for rapid scalability from pilot batches to multi-ton commercial production without the need for specialized high-pressure or high-temperature equipment.
- Cost Reduction in Manufacturing: The elimination of expensive protecting group reagents and the reduction in total synthetic steps directly translate to lower raw material costs. Additionally, the high selectivity of the enzyme minimizes the formation of difficult-to-remove impurities, thereby reducing the burden on purification units and increasing the overall recovery of the valuable steroid backbone. This efficiency gain allows for a more competitive pricing structure in the global market for veterinary and pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: Relying on biocatalysis reduces dependency on volatile petrochemical-derived reagents. The key inputs for this process—glucose, simple buffers, and genetically stable bacterial strains—are commoditized and widely available, ensuring a stable and resilient supply chain. This stability is crucial for maintaining continuous production schedules and meeting the growing global demand for anabolic agents without interruption due to raw material shortages.
- Scalability and Environmental Compliance: The mild reaction conditions inherent to this enzymatic process simplify the engineering requirements for scale-up. There is no need for complex cryogenic cooling or high-pressure reactors, which lowers capital expenditure for new production lines. Moreover, the aqueous nature of the biotransformation step significantly reduces the volume of hazardous organic waste generated, facilitating easier wastewater treatment and ensuring compliance with stringent environmental regulations across different jurisdictions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this P450 BM3 mutant technology. These insights are derived from the patent specifications and are intended to clarify the feasibility and benefits of adopting this route for industrial applications. Understanding these nuances is essential for stakeholders evaluating the transition from traditional chemical synthesis to this advanced bio-hybrid methodology.
Q: How does the P450 BM3 mutant improve selectivity compared to wild-type enzymes?
A: The specific mutations at position 438 (Threonine to Serine, Alanine, or Glycine) in the P450 BM3 backbone significantly alter the active site geometry. This modification enhances the 11α-hydroxylation selectivity up to 92% for methyl dienolone substrates, drastically reducing unwanted by-products common in traditional chemical oxidation methods.
Q: What are the supply chain advantages of this bio-chemical hybrid route?
A: This route eliminates the need for hazardous reagents like acetyl chloride used in traditional protection steps and reduces the total number of synthetic steps. By starting from cheap and readily available 4,9-diene-3,17-dione and using a one-pot enzymatic cascade, manufacturers can achieve substantial cost savings and improved environmental compliance.
Q: Is this process scalable for commercial API intermediate production?
A: Yes, the process utilizes whole-cell biocatalysis in E. coli, which is a highly scalable platform. The mild reaction conditions (20-30°C) and the use of glucose dehydrogenase for cofactor regeneration make it economically viable for large-scale manufacturing of complex steroid intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trenbolone Acetate Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of biocatalytic technologies like the P450 BM3 mutant system in reshaping the landscape of steroid synthesis. As a premier CDMO partner, we possess the technical expertise and infrastructure to translate such innovative patent methodologies into robust, commercial-grade manufacturing processes. Our facilities are equipped to handle complex biotransformations alongside traditional organic synthesis, ensuring a seamless integration of these hybrid routes. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, adhering to the highest industry standards for quality and safety. Our commitment to excellence is underpinned by stringent purity specifications and rigorous QC labs that guarantee every batch meets the exacting requirements of the global pharmaceutical market.
We invite forward-thinking partners to collaborate with us to leverage this cutting-edge technology for your supply chain. Whether you are looking to optimize an existing process or develop a new cost-effective route for Trenbolone Acetate, our team is ready to assist. Please contact our technical procurement team today to request a Customized Cost-Saving Analysis. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how we can drive value and efficiency for your organization.
