Advanced U-Type Template Strategy for Meta-Selective C-H Activation in Pharmaceutical Intermediates
The pharmaceutical industry continuously seeks robust methodologies to access structurally complex intermediates with high regioselectivity, particularly for non-steroidal anti-inflammatory drugs (NSAIDs) and related analgesics. Patent CN109665999B introduces a groundbreaking approach to synthesizing meta-acyloxy substituted phenylacetic acid compounds, addressing a longstanding challenge in organic synthesis regarding meta-selective carbon-hydrogen bond activation. This technology leverages a sophisticated U-type template strategy, employing a pyrimidine moiety as a novel directing group to facilitate direct acetoxylation at the meta-position of phenylacetic acid derivatives. By bypassing the need for pre-functionalized starting materials, this method significantly streamlines the synthetic workflow, offering a viable pathway for the efficient production of high-value pharmaceutical intermediates. The innovation lies not only in the chemical transformation itself but in the strategic design of the template that bridges the spatial gap between the catalyst and the remote meta-carbon, thereby overcoming entropic penalties that typically hinder such reactions.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional methods for functionalizing aromatic rings predominantly rely on electrophilic aromatic substitution or ortho-directed metalation, which inherently lack the ability to selectively target the meta-position due to electronic and steric constraints. In conventional transition metal-catalyzed C-H activation, the formation of a five- or six-membered metallacycle is energetically favorable, directing reactivity to the ortho-position, whereas accessing the meta-position requires the formation of a larger, thermodynamically unstable macrocyclic transition state. This instability often results in poor yields, lack of selectivity, and the necessity for harsh reaction conditions that are incompatible with sensitive functional groups commonly found in drug molecules. Furthermore, existing strategies to achieve meta-substitution often involve multi-step sequences requiring the installation and subsequent removal of bulky blocking groups or the use of stoichiometric amounts of toxic reagents, which drastically increases waste generation and manufacturing costs. The inability to directly functionalize the meta-position of phenylacetic acid scaffolds has historically forced process chemists to adopt circuitous synthetic routes, leading to reduced overall atom economy and prolonged development timelines for new therapeutic candidates.
The Novel Approach
The methodology disclosed in the patent revolutionizes this landscape by utilizing a pyrimidine-based U-type template that effectively acts as a molecular tether, bringing the palladium catalyst into close proximity with the meta-carbon atom. 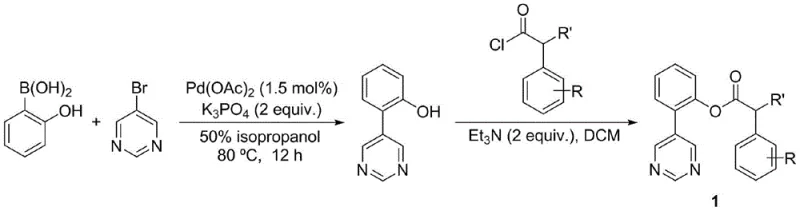 This design allows for the formation of a stable macrocyclic palladacycle intermediate, lowering the activation energy required for meta-C-H bond cleavage and ensuring high regioselectivity. The process utilizes readily available reagents such as iodobenzene diacetate (PhI(OAc)2) as a mild oxidant and operates under relatively moderate thermal conditions, typically around 100 °C, which preserves the integrity of sensitive substrates. By integrating the directing group directly into the substrate architecture via a simple ester linkage, the method achieves a level of precision that eliminates the need for protecting groups or complex purification steps associated with ortho-isomer separation. This novel approach not only fills a critical technical gap in C-H activation chemistry but also provides a versatile platform that can be adapted for the late-stage functionalization of complex drug molecules like ibuprofen, thereby accelerating the discovery of new analogs with improved pharmacological profiles.
This design allows for the formation of a stable macrocyclic palladacycle intermediate, lowering the activation energy required for meta-C-H bond cleavage and ensuring high regioselectivity. The process utilizes readily available reagents such as iodobenzene diacetate (PhI(OAc)2) as a mild oxidant and operates under relatively moderate thermal conditions, typically around 100 °C, which preserves the integrity of sensitive substrates. By integrating the directing group directly into the substrate architecture via a simple ester linkage, the method achieves a level of precision that eliminates the need for protecting groups or complex purification steps associated with ortho-isomer separation. This novel approach not only fills a critical technical gap in C-H activation chemistry but also provides a versatile platform that can be adapted for the late-stage functionalization of complex drug molecules like ibuprofen, thereby accelerating the discovery of new analogs with improved pharmacological profiles.
Mechanistic Insights into Pd-Catalyzed Meta-Selective C-H Activation
The catalytic cycle initiates with the coordination of the palladium(II) species to the nitrogen atoms of the pyrimidine template, which serves as a bidentate ligand to stabilize the metal center and orient it towards the meta-position of the aromatic ring. Upon heating, the palladium center undergoes a concerted metalation-deprotonation (CMD) process, facilitated by the acetate ligands derived from the oxidant or additives, to cleave the strong meta-C-H bond and form a key macrocyclic organopalladium intermediate. This step is the turnover-limiting phase of the reaction, where the unique geometry of the U-type template plays a crucial role in reducing the strain of the large metallacycle, a feat that conventional monodentate directing groups cannot achieve. Following C-H activation, the palladium(IV) species is generated through oxidation by PhI(OAc)2, which subsequently undergoes reductive elimination to forge the new carbon-oxygen bond, installing the acetoxy group with high fidelity. The catalytic cycle is completed by the regeneration of the active palladium(II) species, ready to engage another substrate molecule, while the acetic acid byproduct is managed by the buffer capacity of the reaction medium.
Impurity control in this system is inherently robust due to the high specificity of the template-directed mechanism, which minimizes the formation of ortho- or para-substituted byproducts that typically plague non-directed C-H functionalization reactions. The use of hexafluoroisopropanol (HFIP) as the solvent further enhances selectivity by stabilizing cationic intermediates and promoting hydrogen bonding interactions that favor the desired transition state geometry. Additionally, the choice of N-acetyl amino acids, such as N-acetylglycine, as ligands fine-tunes the electronic properties of the palladium center, preventing catalyst deactivation via aggregation or precipitation. 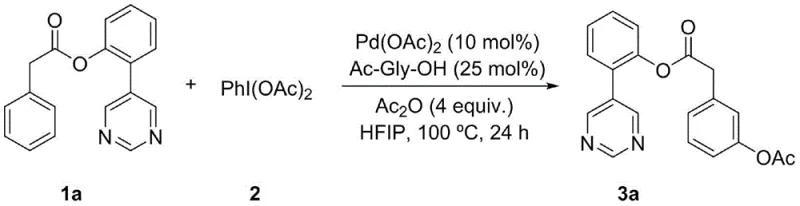 The reaction conditions are optimized to suppress competing pathways, such as homocoupling of the oxidant or decomposition of the ester linkage, ensuring that the crude reaction profile is clean and amenable to straightforward purification via silica gel chromatography. This mechanistic precision translates directly to higher purity specifications for the final isolated product, reducing the burden on downstream processing units and ensuring compliance with stringent regulatory standards for pharmaceutical intermediates.
The reaction conditions are optimized to suppress competing pathways, such as homocoupling of the oxidant or decomposition of the ester linkage, ensuring that the crude reaction profile is clean and amenable to straightforward purification via silica gel chromatography. This mechanistic precision translates directly to higher purity specifications for the final isolated product, reducing the burden on downstream processing units and ensuring compliance with stringent regulatory standards for pharmaceutical intermediates.
How to Synthesize Meta-Acyloxy Phenylacetic Acid Efficiently
The synthesis protocol outlined in the patent provides a reproducible framework for generating these valuable intermediates, starting with the preparation of the pyrimidine-tagged substrate followed by the key acetoxylation step. The process is designed to be operationally simple, requiring standard laboratory equipment such as Schlenk tubes or pressure vessels capable of maintaining an inert nitrogen atmosphere, making it highly transferable from bench-scale discovery to pilot-plant operations. Detailed standardized synthesis steps are provided below to guide process development teams in implementing this technology.
- Prepare the phenylacetic acid derivative substrate (Formula I) containing the pyrimidine directing group via Suzuki coupling and esterification.
- Combine the substrate with PhI(OAc)2 oxidant, Pd(OAc)2 catalyst, Ac-Gly-OH ligand, and Ac2O additive in HFIP solvent under nitrogen atmosphere.
- Heat the reaction mixture to 100 °C for 24 hours, then purify the crude product via column chromatography to isolate the meta-acetoxylated derivative.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this technology offers substantial advantages by simplifying the raw material portfolio and reducing the dependency on exotic or highly specialized reagents that often suffer from supply volatility. The core reactants, including palladium acetate, iodobenzene diacetate, and various N-acetyl amino acids, are commodity chemicals available from multiple global suppliers, ensuring a resilient supply chain that is not vulnerable to single-source bottlenecks. Furthermore, the elimination of pre-functionalization steps means that manufacturers can procure simpler, lower-cost starting materials, such as basic phenylacetic acid derivatives, rather than expensive halogenated or boronated precursors required for cross-coupling alternatives. This consolidation of the synthetic route directly correlates to a reduction in the total number of unit operations, which lowers capital expenditure requirements for reactor volume and decreases the overall consumption of solvents and energy per kilogram of product produced.
- Cost Reduction in Manufacturing: The primary economic driver for adopting this methodology is the drastic simplification of the synthetic sequence, which removes the need for multiple isolation and purification stages associated with traditional multi-step syntheses. By achieving direct meta-functionalization in a single catalytic step, the process significantly reduces labor costs, solvent usage, and waste disposal fees, leading to a leaner and more cost-effective manufacturing model. Although palladium is a precious metal, the catalyst loading is optimized to 10 mol%, and the potential for catalyst recovery and recycling in continuous flow setups could further diminish the effective cost of the metal input over time. Moreover, the high selectivity of the reaction minimizes the loss of valuable starting materials to isomeric byproducts, maximizing the yield of the desired intermediate and improving the overall mass balance of the production campaign.
- Enhanced Supply Chain Reliability: Utilizing a robust catalytic system with widely available reagents mitigates the risk of production delays caused by the shortage of niche custom synthons. The reaction tolerates a broad range of substituents on the aromatic ring, including alkyl, alkoxy, and halogen groups, which allows for the flexible sourcing of diverse phenylacetic acid feedstocks without requiring custom synthesis for each variant. This flexibility empowers supply chain managers to negotiate better pricing with vendors by leveraging the interchangeability of raw materials and to maintain safety stock levels more effectively. Additionally, the mild reaction conditions reduce the wear and tear on processing equipment, extending asset life and minimizing unplanned maintenance downtime that could disrupt delivery schedules to downstream API manufacturers.
- Scalability and Environmental Compliance: The process demonstrates excellent scalability potential, as evidenced by the successful execution of reactions on millimole scales with consistent yields, suggesting a smooth path to kilogram and tonne-scale production with appropriate engineering controls. The use of HFIP, while a specialized solvent, is employed in manageable quantities, and the overall waste profile is improved compared to stoichiometric heavy metal oxidation methods, aligning with modern green chemistry principles. The ability to remove the pyrimidine template under mild conditions post-reaction ensures that the final waste stream does not contain persistent organic pollutants derived from the directing group, facilitating easier wastewater treatment and regulatory compliance. This environmental compatibility is increasingly critical for maintaining operating licenses in regions with strict emission standards, positioning this technology as a sustainable choice for long-term commercial manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical and operational inquiries regarding the implementation of this meta-selective acetoxylation technology, based on the detailed experimental data and scope provided in the patent documentation. These insights are intended to assist R&D and process engineering teams in evaluating the feasibility of integrating this route into their existing manufacturing pipelines.
Q: What is the primary advantage of the U-type template strategy in this synthesis?
A: The U-type template strategy utilizes a pyrimidine directing group to overcome the high energy barrier associated with forming macrocyclic transition states, enabling selective functionalization at the sterically challenging meta-position which is typically inaccessible via conventional ortho-directed methods.
Q: Can the pyrimidine template be removed after the reaction?
A: Yes, the patent specifies that the pyrimidine-based template can be removed under mild conditions after the functionalization step, ensuring that the final drug molecule retains its biological activity without interference from the directing group.
Q: What represents the most significant cost driver in this catalytic system?
A: While palladium catalysts are used, the process employs relatively low loading (10 mol%) and utilizes commercially available oxidants like PhI(OAc)2; the primary cost benefit arises from eliminating multi-step pre-functionalization sequences required in traditional synthetic routes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Meta-Acyloxy Phenylacetic Acid Supplier
The technological breakthroughs described in patent CN109665999B represent a significant leap forward in the synthesis of complex pharmaceutical intermediates, offering a clear pathway to more efficient and sustainable production of anti-inflammatory agents. NINGBO INNO PHARMCHEM stands at the forefront of translating such innovative academic and patent literature into commercial reality, leveraging our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle the specific requirements of palladium-catalyzed reactions, including rigorous containment protocols for metal handling and advanced purification capabilities to meet stringent purity specifications. With our dedicated rigorous QC labs, we ensure that every batch of meta-acyloxy phenylacetic acid derivatives delivered meets the highest standards of quality and consistency required by global regulatory bodies.
We invite pharmaceutical partners and contract research organizations to collaborate with us to explore the full potential of this U-type template strategy for their specific drug development programs. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your target molecule, comparing this direct acetoxylation route against your current synthetic methodology. We encourage you to contact us today to obtain specific COA data for our reference standards and to discuss route feasibility assessments that could accelerate your project timelines and reduce your overall cost of goods sold.
