Advanced Manufacturing of 2,4-Diamino-6-Chloropyrimidine: A Breakthrough in Minoxidil Precursor Production
The pharmaceutical industry constantly seeks robust, scalable, and environmentally compliant routes for critical active pharmaceutical ingredient (API) precursors. A pivotal development in this domain is detailed in patent CN113754592A, which discloses a highly optimized preparation method for 2,4-diamino-6-chloropyrimidine (DACP). This compound serves as a fundamental building block for the synthesis of Minoxidil, a widely used vasodilator and hair growth agent, as well as finding applications in the electrolysis industry. The traditional manufacturing landscape for DACP has long been plagued by inefficiencies, particularly regarding the quenching of phosphorus oxychloride (POCl3) and the subsequent isolation of the product. The disclosed innovation introduces a paradigm shift by replacing hazardous water quenching with a controlled alcoholysis protocol, thereby enhancing safety profiles and enabling the recovery of valuable phosphate byproducts. For global procurement teams and R&D directors, understanding this technological leap is essential for securing a reliable pharmaceutical intermediate supplier capable of delivering high-quality materials with reduced environmental impact.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of 2,4-diamino-6-chloropyrimidine has relied heavily on the chlorination of 2,4-diamino-6-hydroxypyrimidine using phosphorus oxychloride, followed by a quenching step involving water. This conventional approach presents severe operational and economic bottlenecks that hinder efficient cost reduction in API manufacturing. The primary defect lies in the quenching phase; adding water to a reaction mixture containing excess POCl3 is highly exothermic and dangerous, posing significant safety risks in large-scale reactors. Furthermore, water quenching converts all phosphorus species into phosphoric acid, which has a high boiling point and is thermally unstable, making it extremely difficult to separate and recover. This results in the generation of massive volumes of phosphorus-containing wastewater, creating a heavy burden on waste treatment facilities and inflating disposal costs. Additionally, literature reports indicate that extracting the product from this aqueous acidic slurry requires excessive amounts of organic solvents, such as ethyl acetate, often needing 30 to 40 times the volume of the reaction mass, which drastically lowers equipment utilization rates and overall yield.
The Novel Approach
In stark contrast, the methodology outlined in patent CN113754592A offers a sophisticated solution by employing alcohol, specifically ethanol or other lower alcohols, for the quenching step. This strategic modification transforms the quenching byproduct from non-volatile phosphoric acid into volatile phosphate esters. These esters possess significantly lower boiling points, allowing them to be easily separated and recovered via distillation alongside the solvent, thus closing the material loop and minimizing waste. The process also incorporates the use of a dispersing agent with lower polarity, such as ethyl acetate, which facilitates the precipitation of the intermediate hydrochloride salt directly from the reaction mixture. This eliminates the need for the cumbersome and solvent-intensive extraction steps required in the water-quench method. By streamlining the isolation procedure and improving the thermal stability of the byproducts, this novel approach not only boosts the yield to over 70% but also aligns perfectly with modern green chemistry principles, making it an ideal candidate for the commercial scale-up of complex heterocycles.
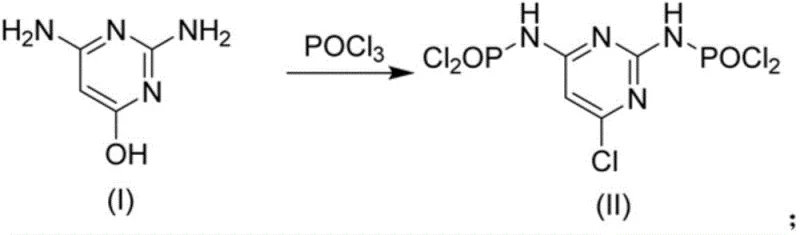
Mechanistic Insights into POCl3-Mediated Chlorination and Alcoholysis
The core of this synthesis lies in the precise control of the chlorination and subsequent alcoholysis mechanisms. The reaction initiates with the nucleophilic attack of the hydroxyl group and the amino groups of 2,4-diamino-6-hydroxypyrimidine on the phosphorus atom of POCl3. Under elevated temperatures ranging from 90°C to 110°C, the hydroxyl group is successfully converted into a chloro group, while the amino groups are transiently protected as dichlorophosphoryl derivatives. This protection is crucial as it prevents side reactions and ensures regioselectivity. The formation of the intermediate, 2,4-di(dichlorophosphoryl)amino-6-chloropyrimidine, is a key determinant of the final product quality. Maintaining the reaction temperature at an optimal 105°C for approximately 6 hours ensures complete conversion while minimizing the formation of poly-chlorinated impurities or degradation products that could compromise the purity profile required for pharmaceutical applications.
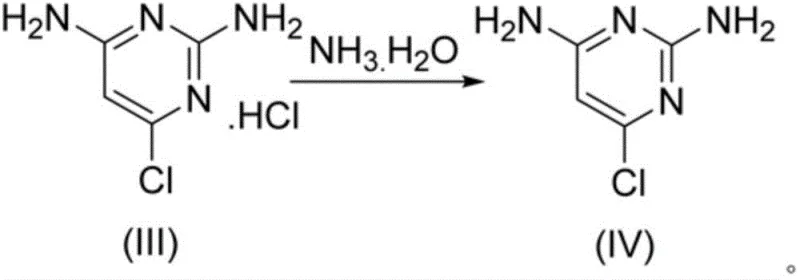
Following the chlorination, the mechanism shifts to alcoholysis, where the addition of alcohol acts as a nucleophile to cleave the P-N bonds of the protecting groups. Unlike water, which hydrolyzes these bonds violently to release ammonia and phosphoric acid, alcohol reacts more gently to form phosphate esters and the hydrochloride salt of the amine. This step is critical for impurity control; by carefully controlling the temperature during alcohol addition (0-40°C) and subsequent reflux, the process ensures that the free amine is protonated immediately, preventing it from acting as a nucleophile in unwanted side reactions. The final neutralization with ammonia water at a controlled pH of 6-7 liberates the free base of 2,4-diamino-6-chloropyrimidine. This specific pH window is vital; it is high enough to deprotonate the amine for extraction into the organic phase but low enough to prevent the hydrolysis of the sensitive chloro-pyrimidine ring, thereby preserving the structural integrity of the high-purity minoxidil precursor.
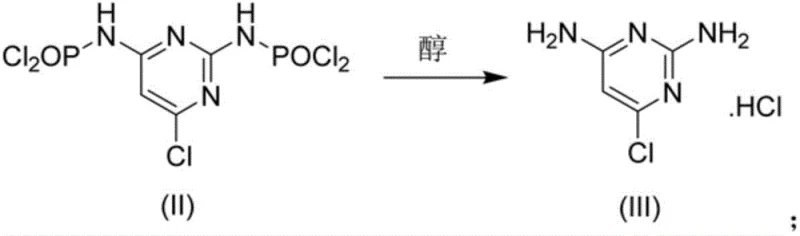
How to Synthesize 2,4-Diamino-6-Chloropyrimidine Efficiently
The implementation of this synthesis route requires strict adherence to the sequential addition of reagents and temperature controls to maximize efficiency and safety. The process begins with the charging of the starting material and POCl3, followed by a controlled heat-up to drive the chlorination to completion. Once the excess POCl3 is distilled off for recycling, the critical quenching step with alcohol is performed under cooling to manage the exotherm. The subsequent addition of a dispersing solvent induces crystallization of the intermediate salt, which is then filtered and neutralized.
- Chlorinate 2,4-diamino-6-hydroxypyrimidine with phosphorus oxychloride at 90-110°C to form the dichlorophosphoryl intermediate.
- Quench the reaction mixture with ethanol at 0-40°C, followed by heating to reflux to precipitate the hydrochloride salt.
- Neutralize the hydrochloride salt with ammonia water at pH 6-7 and extract with ethyl acetate to isolate the pure product.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain leaders and procurement managers, the adoption of this alcohol-quenching technology translates into tangible operational benefits that extend beyond simple yield metrics. The shift away from water quenching fundamentally alters the waste management profile of the facility, turning a costly waste disposal problem into a resource recovery opportunity. The ability to distill and recover phosphate esters and solvents means that raw material consumption per kilogram of product is significantly lowered. Furthermore, the enhanced safety profile of using alcohol instead of water for quenching reduces the risk of runaway reactions, leading to lower insurance premiums and reduced downtime associated with safety incidents. This robustness ensures a more consistent supply flow, which is critical for maintaining production schedules in downstream API manufacturing.
- Cost Reduction in Manufacturing: The elimination of massive wastewater treatment requirements and the recovery of valuable phosphate byproducts lead to substantial cost savings. By avoiding the need for 30-40 volumes of extraction solvent, the process drastically reduces solvent purchase and recovery costs. The simplified work-up procedure also shortens the batch cycle time, increasing the throughput of existing reactor assets without the need for capital expenditure on new equipment.
- Enhanced Supply Chain Reliability: The use of common, commercially available reagents like ethanol and ethyl acetate ensures that the supply chain is not vulnerable to the shortages of exotic catalysts or specialized reagents. The high yield consistency (>70%) guarantees that production targets can be met reliably, reducing lead time for high-purity intermediates and ensuring that downstream customers receive their materials on schedule. The stability of the intermediate salt also allows for flexible storage and transport if necessary.
- Scalability and Environmental Compliance: This process is inherently designed for industrial mass production, with parameters that are easily scalable from pilot plant to multi-ton reactors. The significant reduction in phosphorus-containing effluent simplifies compliance with increasingly stringent environmental regulations. The lower boiling point of the byproducts facilitates energy-efficient distillation, contributing to a lower carbon footprint for the manufacturing process.
Frequently Asked Questions (FAQ)
To further clarify the technical and commercial implications of this patented technology, we have compiled answers to common inquiries regarding the synthesis and application of 2,4-diamino-6-chloropyrimidine. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing a transparent view of the process capabilities. Understanding these details helps stakeholders make informed decisions regarding vendor selection and process integration.
Q: Why is alcohol quenching preferred over water quenching for DACP synthesis?
A: Alcohol quenching generates phosphate esters which have lower boiling points and are easier to recover than phosphoric acid generated by water quenching. Additionally, alcohol quenching is significantly safer and smoother, avoiding the violent exotherms associated with water addition to excess POCl3.
Q: What is the expected yield and purity of this novel process?
A: According to patent CN113754592A, this method achieves a recovery rate of more than 70%, with optimal yields reaching 82%. The final product purity can reach 99.2% as determined by HPLC analysis.
Q: How does this method address environmental concerns regarding phosphorus waste?
A: Traditional water quenching produces massive amounts of phosphorus-containing wastewater that is difficult to treat. This new method allows for the recovery of phosphate esters and solvents through distillation, drastically reducing the environmental burden and waste disposal costs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,4-Diamino-6-Chloropyrimidine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of pharmaceutical development and production. Our technical team has thoroughly analyzed the pathway described in CN113754592A and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring this optimized route to life. We are committed to delivering materials that meet stringent purity specifications, supported by our rigorous QC labs equipped with advanced analytical instrumentation to verify every batch. Our facility is designed to handle reactive chemistries safely, ensuring that the benefits of the alcohol-quenching method are fully realized in a GMP-compliant environment.
We invite potential partners to engage with us for a Customized Cost-Saving Analysis tailored to your specific volume requirements. By leveraging our expertise in process optimization and supply chain management, we can help you secure a stable source of this vital intermediate. Please contact our technical procurement team to request specific COA data and route feasibility assessments, and let us demonstrate how our commitment to innovation can drive value for your organization.
