Advanced Synthesis of 1-Hydroxy-pyrrolo[2,3-c]piperidine for Moxifloxacin Side Chains
Introduction to Next-Generation Moxifloxacin Intermediate Synthesis
The global demand for fourth-generation quinolone antibiotics, particularly Moxifloxacin, continues to surge due to its broad-spectrum efficacy against respiratory pathogens. At the heart of manufacturing this critical active pharmaceutical ingredient lies the complex synthesis of its seven-position side chain, specifically the (S,S)-2,8-diazabicyclo[4.3.0]nonane moiety. A pivotal breakthrough in this domain is detailed in patent CN111171022B, which discloses a highly efficient and safe method for synthesizing 1-hydroxy-pyrrolo[2,3-c]piperidine, a key precursor to the final side chain. This innovation addresses long-standing industrial pain points by replacing hazardous reagents with a streamlined, base-mediated cyclization strategy. For R&D directors and procurement leaders, understanding this shift is vital, as it represents a move towards greener, more cost-effective, and scalable chemical manufacturing processes that align with modern regulatory and economic standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of the moxifloxacin side chain has relied heavily on routes starting from dipicolinic acid. While chemically feasible, this traditional pathway is fraught with significant operational and safety challenges that burden supply chains and inflate costs. The process necessitates the use of substantial quantities of sodium borohydride as a reducing agent, a reagent known for its instability and reactivity. During the reaction, the decomposition of sodium borohydride generates large volumes of borane gas, creating a severe fire hazard and requiring specialized, explosion-proof infrastructure. Furthermore, the handling of such reactive hydrides demands rigorous safety protocols, increasing operational overhead and limiting the scalability of production facilities. These factors collectively result in a high-cost manufacturing environment with elevated risk profiles, making the conventional route increasingly unattractive for modern, safety-conscious pharmaceutical supply chains.
The Novel Approach
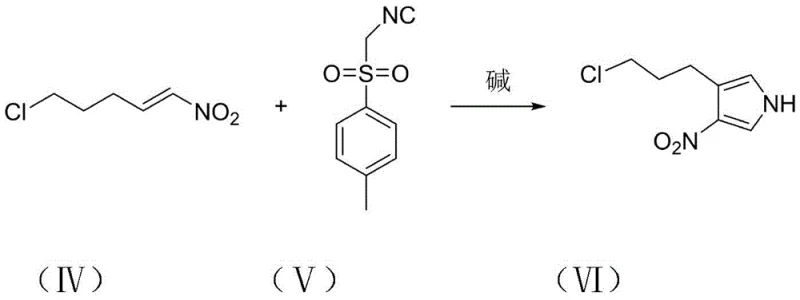
In stark contrast, the methodology outlined in patent CN111171022B introduces a paradigm shift by utilizing 1-nitro-5-chloro-1-pentene and TosMIC (tosylmethyl isocyanide) as primary building blocks. This novel route bypasses the need for dangerous hydride reductions entirely, opting instead for a sophisticated cyclization strategy that operates under much milder conditions. The initial step involves a base-catalyzed reaction between the nitroalkene and TosMIC to construct the pyrrole core, a transformation that is both atom-economical and operationally simple. By eliminating the generation of flammable gases and utilizing readily available, stable starting materials, this approach fundamentally de-risks the production process. The transition from hazardous batch processes to this controlled, step-wise synthesis offers a clear pathway for cost reduction in pharmaceutical intermediates manufacturing, enabling producers to achieve higher throughput with significantly lower safety compliance burdens.
Mechanistic Insights into TosMIC-Mediated Cyclization and Hydrogenation
The core of this synthetic innovation lies in the precise orchestration of three distinct chemical transformations, each optimized for yield and purity. The first stage utilizes a strong base, such as potassium tert-butoxide, to deprotonate TosMIC, generating a nucleophilic carbanion that attacks the electron-deficient double bond of the nitroalkene. This Michael addition is followed by an intramolecular cyclization and elimination of the tosyl group, effectively constructing the 3-(3'-chloropropyl)-4-nitro-1H-pyrrole scaffold (Compound VI). This step is critical as it establishes the heterocyclic core with high regioselectivity, minimizing the formation of structural isomers that are difficult to separate later. The reaction is typically conducted in tetrahydrofuran at low temperatures (-20°C to 25°C), which suppresses side reactions and ensures the stability of the reactive intermediates, resulting in yields exceeding 80% in optimized examples.
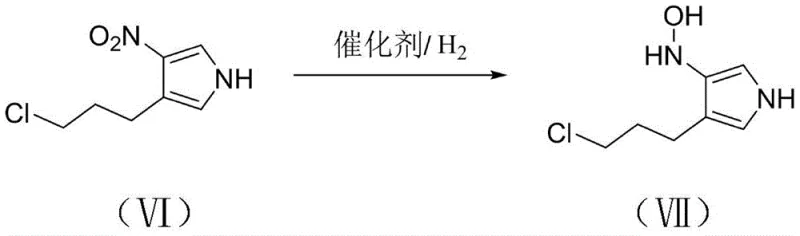
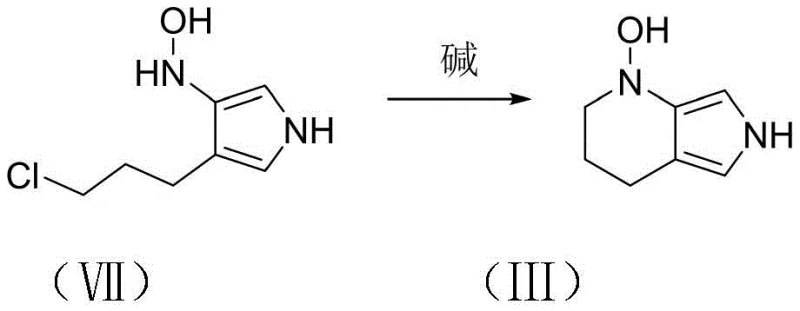
Following the construction of the nitro-pyrrole framework, the process proceeds to a selective catalytic hydrogenation step. Unlike harsh chemical reductions that might over-reduce the system or affect other functional groups, this method employs palladium on carbon (Pd/C) under mild hydrogen pressure (1-4 atm) in methanol. This specific condition set is engineered to reduce the nitro group selectively to a hydroxylamino functionality, yielding Compound VII without compromising the integrity of the chloropropyl side chain or the pyrrole ring. The subsequent final step involves a base-mediated intramolecular nucleophilic substitution. Here, the newly formed hydroxylamino group acts as a nucleophile, attacking the terminal chloride of the propyl chain to close the piperidine ring. This elegant cascade transforms the linear precursor into the bicyclic 1-hydroxy-pyrrolo[2,3-c]piperidine (Compound III), completing the synthesis with exceptional efficiency.
How to Synthesize 1-Hydroxy-pyrrolo[2,3-c]piperidine Efficiently
Implementing this synthesis requires careful attention to reaction parameters, particularly temperature control during the initial TosMIC coupling and catalyst loading during hydrogenation. The protocol dictates a sequential addition of reagents to manage exotherms and ensure complete conversion. For the initial cyclization, maintaining the reaction mixture between -10°C and 25°C is paramount to prevent the decomposition of the isocyanide species. Following isolation of the nitro-intermediate, the hydrogenation step should be monitored closely to avoid over-reduction, although the Pd/C system described shows high selectivity. Finally, the ring-closing step benefits from vigorous stirring to facilitate the interaction between the organic substrate and the inorganic base. For a comprehensive, step-by-step operational guide including exact stoichiometry and workup procedures, please refer to the standardized synthesis protocol below.
- React 1-nitro-5-chloro-1-pentene with TosMIC under basic conditions (e.g., potassium tert-butoxide) in THF at low temperature to form the nitro-pyrrole intermediate.
- Subject the nitro-pyrrole intermediate to catalytic hydrogenation using Pd/C in methanol to reduce the nitro group to a hydroxylamino group.
- Treat the hydroxylamino intermediate with a base such as potassium carbonate in an organic solvent to induce intramolecular cyclization, yielding the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers tangible strategic benefits beyond mere technical feasibility. The shift away from dipicolinic acid and sodium borohydride eliminates a major bottleneck associated with the sourcing and handling of hazardous materials. By relying on commodity chemicals like nitroalkenes and TosMIC, manufacturers can secure a more resilient supply chain that is less susceptible to regulatory crackdowns on dangerous goods transportation. This stability translates directly into improved delivery reliability and reduced lead times for high-purity pharmaceutical intermediates. Furthermore, the simplified waste profile—devoid of boron-containing byproducts—streamlines environmental compliance and waste disposal processes, which are often significant cost centers in fine chemical production.
- Cost Reduction in Manufacturing: The elimination of expensive and hazardous reducing agents like sodium borohydride results in direct material cost savings. Additionally, the mild reaction conditions reduce energy consumption for heating and cooling, while the high yields observed in each step minimize raw material waste. The avoidance of specialized explosion-proof equipment further lowers capital expenditure requirements for production facilities, contributing to a substantially lower cost of goods sold (COGS) for the final intermediate.
- Enhanced Supply Chain Reliability: The starting materials for this route, specifically 1-nitro-5-chloro-1-pentene and TosMIC, are commercially available from multiple global suppliers, reducing dependency on single-source vendors. The robustness of the reaction conditions means that production is less likely to be interrupted by minor fluctuations in utility supplies or environmental conditions. This reliability ensures a consistent flow of reliable pharmaceutical intermediates supplier products to downstream API manufacturers, safeguarding their production schedules against upstream volatility.
- Scalability and Environmental Compliance: The process is inherently scalable, moving seamlessly from kilogram-scale R&D batches to multi-ton commercial production without significant re-engineering. The absence of toxic borane gas emissions simplifies the permitting process for new manufacturing lines and reduces the burden on scrubber systems. This environmental friendliness aligns with the increasing corporate sustainability goals of major pharmaceutical companies, making this route a preferred choice for long-term partnerships focused on green chemistry initiatives.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent literature, providing a clear picture of what partners can expect when adopting this methodology. Understanding these nuances is crucial for technical teams evaluating the feasibility of technology transfer and for commercial teams negotiating supply agreements based on quality and safety metrics.
Q: What are the safety advantages of this new synthesis route compared to traditional methods?
A: Traditional routes often utilize dipicolinic acid and large quantities of sodium borohydride, which generate hazardous borane gas and pose significant fire risks. This novel method avoids these dangerous reagents, utilizing milder conditions and safer starting materials like TosMIC and nitroalkenes, thereby drastically reducing production hazards.
Q: How does this process impact the cost of manufacturing moxifloxacin intermediates?
A: The process significantly lowers costs by eliminating the need for expensive and hazardous reducing agents like sodium borohydride. Furthermore, the starting materials are readily available and inexpensive, and the reaction conditions are mild, reducing energy consumption and equipment corrosion, leading to substantial overall cost savings.
Q: What is the expected purity and yield profile of the final intermediate?
A: The patented method demonstrates high efficiency with yields reaching up to 84-91% in individual steps. The mild reaction temperatures and specific catalytic hydrogenation steps help minimize side reactions and impurity formation, ensuring a high-purity profile suitable for downstream pharmaceutical applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1-Hydroxy-pyrrolo[2,3-c]piperidine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the global pharmaceutical supply chain. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless. We are committed to delivering stringent purity specifications for complex heterocycles like 1-hydroxy-pyrrolo[2,3-c]piperidine, supported by our rigorous QC labs and state-of-the-art analytical capabilities. Our facility is equipped to handle the specific safety and processing requirements of this novel route, guaranteeing a steady supply of material that meets the exacting standards of international drug regulators.
We invite potential partners to engage with our technical procurement team to discuss how this advanced synthesis route can optimize your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a detailed understanding of the economic benefits specific to your volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that drive efficiency and profitability in your moxifloxacin production programs.
