Advanced Biocatalytic Production of High-Purity (S)-2-Phenylbutyric Acid for Pharmaceutical Applications
The pharmaceutical industry constantly seeks robust, scalable, and environmentally sustainable pathways for synthesizing chiral intermediates, particularly for cardiovascular therapeutics like Indobufen. Patent CN108546720B introduces a groundbreaking method for preparing (S)-2-phenylbutyric acid, a critical chiral building block, through stereoselective enzymatic hydrolysis. This technology leverages the high specificity of lipases combined with the solubilizing power of cyclodextrins to overcome traditional limitations in aqueous biocatalysis. By shifting away from harsh chemical resolutions and toxic organic solvents, this process aligns perfectly with modern green chemistry principles while delivering optical purities exceeding 96% ee. For R&D directors and procurement strategists, this patent represents a significant opportunity to optimize the supply chain for antithrombotic drug precursors, ensuring both regulatory compliance and cost-efficiency in high-volume manufacturing environments.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the resolution of racemic 2-phenylbutyric acid has relied on methods such as fractional crystallization, chromatographic separation, or chemical extraction, each carrying substantial operational burdens. Crystallization techniques, while economically attractive for simple molecules, often suffer from inherently low theoretical yields (maximum 50%) and require multiple recrystallization steps to achieve acceptable optical purity, making them inefficient for large-scale continuous production. Chromatographic methods offer high separation efficiency but are prohibitively expensive due to the cost of chiral stationary phases and the complexity of scaling up column operations for tonnage quantities. Furthermore, traditional chemical resolution often necessitates the use of volatile organic solvents and stoichiometric amounts of resolving agents, generating significant hazardous waste streams that complicate environmental compliance and increase disposal costs for manufacturing facilities.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes a biocatalytic system where lipase selectively hydrolyzes racemic 2-phenylbutyrate esters in a mild aqueous buffer. This method fundamentally transforms the process economics by replacing expensive organic solvents with water-based systems and eliminating the need for stoichiometric chiral auxiliaries. The integration of cyclodextrin derivatives addresses the critical bottleneck of substrate solubility, allowing the hydrophobic ester to interact efficiently with the enzyme in the aqueous phase. As illustrated in the reaction scheme below, this synergy between biocatalysis and supramolecular chemistry enables a continuous, high-yield process that produces the desired (S)-enantiomer with exceptional purity.
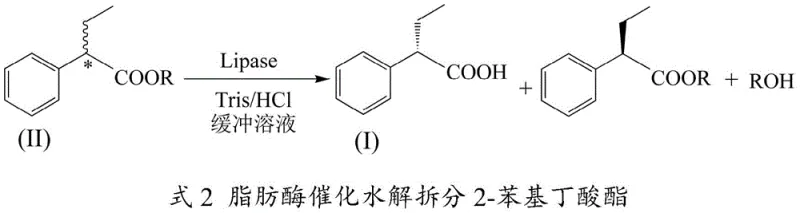
Mechanistic Insights into Lipase-Catalyzed Stereoselective Hydrolysis
The core of this technological advancement lies in the precise molecular recognition capabilities of Candida antarctica lipase A, which possesses an asymmetric active center capable of distinguishing between enantiomers. In this catalytic cycle, the enzyme preferentially binds to and hydrolyzes the ester bond of one specific enantiomer of the 2-phenylbutyrate substrate, releasing the free (S)-2-phenylbutyric acid while leaving the (R)-ester unreacted. This kinetic resolution is driven by the specific spatial arrangement of amino acid residues within the lipase active site, which creates a steric environment favorable only for the transition state of the target enantiomer. The result is a highly enantioselective transformation that minimizes the formation of unwanted by-products and simplifies downstream purification processes significantly.
A critical innovation in this patent is the strategic use of cyclodextrin derivatives to modulate the reaction microenvironment. Since the lipase operates optimally in aqueous buffers while the substrate is a hydrophobic organic ester, mass transfer limitations typically restrict reaction rates. The cyclodextrin molecule, with its hydrophilic exterior and hydrophobic interior cavity, forms an inclusion complex with the 2-phenylbutyrate ester, effectively solubilizing it within the aqueous phase. This host-guest interaction, depicted in the structural diagram below, ensures a high local concentration of the substrate near the enzyme, drastically improving conversion rates without denaturing the biocatalyst or requiring co-solvents that might inhibit enzyme activity.
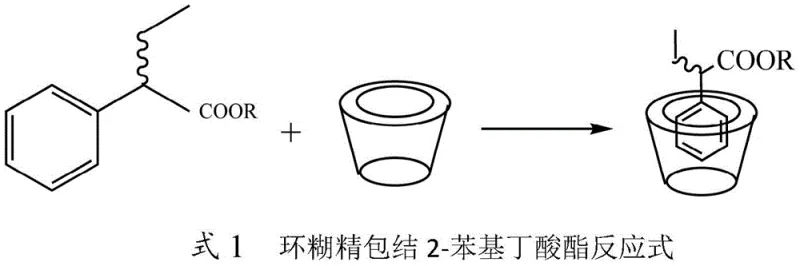
How to Synthesize (S)-2-Phenylbutyric Acid Efficiently
The synthesis protocol outlined in the patent provides a streamlined workflow for producing high-purity chiral acids suitable for pharmaceutical applications. The process begins with the preparation of a buffered reaction medium containing a specific concentration of hydroxyethyl-beta-cyclodextrin, which serves as the phase-transfer solubilizer. Following the addition of the racemic ester substrate and the immobilized or free lipase catalyst, the mixture is subjected to controlled agitation and heating to maintain optimal enzymatic activity. Detailed standard operating procedures regarding specific molar ratios, temperature gradients, and reaction times are essential for replicating the high enantiomeric excess reported in the examples.
- Prepare the reaction medium by dissolving a beta-cyclodextrin derivative, such as hydroxyethyl-beta-cyclodextrin, in a Tris/HCl buffer solution to enhance substrate solubility.
- Introduce the racemic 2-phenylbutyrate ester substrate and Candida antarctica lipase A into the closed reaction system under controlled stirring and heating conditions.
- Monitor the reaction progress via HPLC analysis to ensure high enantiomeric excess (ee) and conversion, followed by product isolation and purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this enzymatic technology offers profound strategic benefits beyond mere technical feasibility. The shift from organic solvent-based chemical resolution to an aqueous enzymatic process fundamentally alters the cost structure of manufacturing by eliminating the procurement, storage, and disposal costs associated with volatile organic compounds. Additionally, the use of readily available, non-toxic cyclodextrins and robust commercial lipases reduces dependency on specialized, high-cost chiral reagents, thereby stabilizing raw material costs and mitigating supply risks associated with fluctuating chemical markets.
- Cost Reduction in Manufacturing: The elimination of expensive organic solvents and stoichiometric resolving agents leads to a drastic simplification of the material bill of goods. By utilizing water as the primary reaction medium, facilities can avoid the capital expenditure and operational costs linked to solvent recovery systems and explosion-proof infrastructure. Furthermore, the high selectivity of the lipase reduces the burden on downstream purification units, lowering energy consumption and waste treatment expenses associated with separating close-boiling impurities or diastereomeric salts.
- Enhanced Supply Chain Reliability: The raw materials required for this process, including buffer salts, cyclodextrins, and commercial lipases, are commodity chemicals with stable, global supply chains. This contrasts sharply with traditional methods that may rely on bespoke chiral catalysts or resolving agents with limited suppliers and long lead times. The robustness of the enzymatic system also allows for flexible production scheduling, as the reaction conditions are mild and do not require extreme temperatures or pressures that could strain utility infrastructure.
- Scalability and Environmental Compliance: The aqueous nature of the reaction system makes it inherently safer and easier to scale from pilot plant to commercial tonnage production without the safety hazards associated with large volumes of flammable solvents. From a regulatory perspective, the reduction in hazardous waste generation and the use of biodegradable catalysts align with increasingly stringent environmental regulations, facilitating smoother permitting processes and enhancing the corporate sustainability profile of the manufacturing entity.
Frequently Asked Questions (FAQ)
The following questions address common technical and operational inquiries regarding the implementation of this stereoselective hydrolysis technology. These insights are derived directly from the experimental data and process descriptions found in the patent literature, aiming to clarify the practical aspects of adopting this green chemistry route for industrial applications. Understanding these nuances is crucial for technical teams evaluating the feasibility of integrating this method into existing production lines.
Q: Why is cyclodextrin added to the enzymatic hydrolysis system?
A: Cyclodextrin acts as a solubilizer due to its unique structure with a hydrophobic cavity and hydrophilic exterior. It encapsulates the hydrophobic 2-phenylbutyrate ester, significantly improving its solubility in the aqueous buffer and thereby increasing the reaction conversion rate without compromising stereoselectivity.
Q: What represents the primary advantage of this lipase method over traditional crystallization?
A: Unlike traditional crystallization which often suffers from low yields and intermittent processing, this enzymatic method operates under mild conditions in an aqueous system. It achieves high optical purity (ee ≥ 96%) continuously, eliminates the need for toxic organic solvents, and offers a more environmentally sustainable pathway suitable for large-scale production.
Q: Which specific lipase demonstrates the highest selectivity for this transformation?
A: Among various lipases tested, Candida antarctica lipase A exhibits superior catalytic activity and stereoselectivity for the hydrolysis of racemic 2-phenylbutyrate. It preferentially recognizes and hydrolyzes the specific enantiomer required to produce (S)-2-phenylbutyric acid with high efficiency.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (S)-2-Phenylbutyric Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-purity chiral intermediates in the development of next-generation cardiovascular therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced chiral HPLC capabilities to guarantee that every batch of (S)-2-phenylbutyric acid meets the exacting standards required for API synthesis.
We invite pharmaceutical partners to collaborate with us to leverage this advanced enzymatic technology for their supply chains. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage potential clients to contact us directly to obtain specific COA data and comprehensive route feasibility assessments, ensuring that your project benefits from the highest levels of quality and economic efficiency available in the market today.
