Advanced Synthesis of 5-Chloro-4-Formyl Oxazole for Commercial Pharmaceutical Manufacturing
The pharmaceutical and fine chemical industries are constantly seeking more sustainable and efficient pathways for synthesizing critical heterocyclic intermediates. A pivotal advancement in this domain is detailed in patent CN102060800B, which discloses a novel method for synthesizing 5-chloro-4-formyl oxazole. This compound serves as a vital building block for various bioactive molecules, yet its historical production has been plagued by environmental and efficiency challenges. The patented technology introduces a groundbreaking approach utilizing bis(trichloromethyl)carbonate (BTC) instead of traditional phosphorus-based reagents. By shifting the chemical paradigm from phosphorus oxychloride (POCl3) to BTC, the process not only enhances reaction selectivity and yield but also fundamentally alters the waste profile of the manufacturing operation. For R&D directors and procurement specialists, understanding this transition is crucial for optimizing supply chains and reducing the total cost of ownership for complex pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 5-chloro-4-formyl oxazole and its derivatives relied heavily on the Vilsmeier-Haack reaction, which typically employs phosphorus oxychloride (POCl3) in conjunction with dimethylformamide (DMF). While chemically effective, this conventional route presents severe drawbacks for modern industrial applications. The primary concern is the generation of substantial phosphorus-containing wastewater, which necessitates complex and costly treatment protocols to meet stringent environmental regulations. Furthermore, POCl3 is a highly corrosive and hazardous material, requiring specialized storage and handling infrastructure that increases operational overhead. From a yield perspective, literature prior to this invention, such as reports in J. Chem. Soc. Perkin Trans. 1, indicated total yields hovering around only 56%. This low efficiency, combined with difficult product separation and purification steps, resulted in a process that was neither economically nor environmentally sustainable for large-scale commercial production.
The Novel Approach
The innovative methodology described in the patent data offers a robust alternative by replacing the hazardous phosphorus reagent with bis(trichloromethyl)carbonate (BTC), also known as triphosgene. This substitution is transformative, as BTC acts as a safe, solid substitute for phosgene and effectively facilitates the chlorination and cyclization required to form the oxazole ring without introducing phosphorus atoms into the waste stream. The reaction proceeds by mixing glycine derivatives, BTC, and formamide compounds in common organic solvents like toluene or xylene. As illustrated in the reaction scheme below, this pathway allows for precise control over the substitution patterns on the oxazole ring.
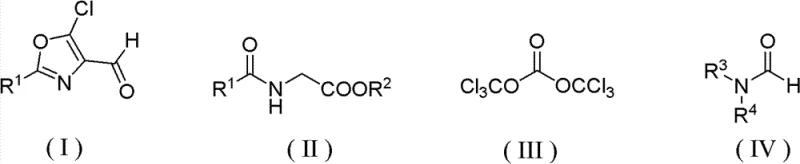
This new route significantly simplifies the post-reaction workup. Instead of dealing with phosphoric acid sludge, the process involves a straightforward aqueous hydrolysis step followed by phase separation. The elimination of phosphorus waste drastically reduces the burden on effluent treatment plants, directly translating to lower operational costs. Moreover, the reaction demonstrates superior selectivity, minimizing the formation of by-products and enabling easier purification through standard recrystallization techniques, thereby ensuring a high-purity final product suitable for sensitive pharmaceutical applications.
Mechanistic Insights into BTC-Mediated Cyclization and Formylation
The core of this synthetic breakthrough lies in the unique reactivity of bis(trichloromethyl)carbonate (BTC) when activated by formamide derivatives. Mechanistically, BTC decomposes in situ to generate phosgene equivalents, which react with the formamide to create a reactive chloroiminium species similar to the Vilsmeier reagent but without the phosphorus backbone. This electrophilic species then attacks the enolizable position of the acyl glycine derivative. The subsequent intramolecular cyclization is driven by the nucleophilic attack of the amide oxygen onto the activated carbonyl carbon, facilitated by the chloride ions released during the decomposition of BTC. This cascade results in the formation of the stable oxazole ring with the chlorine atom strategically positioned at the 5-position and the formyl group at the 4-position. The use of BTC ensures a steady, controlled release of the chlorinating agent, which prevents the runaway exotherms often associated with liquid phosgene or aggressive POCl3 additions, thus enhancing process safety.
Impurity control is another critical aspect where this mechanism excels. In traditional POCl3 methods, over-chlorination or phosphorylation of the substrate can occur, leading to complex impurity profiles that are difficult to remove. The BTC-mediated pathway, operating under milder thermal conditions (typically 50°C to 100°C), exhibits high chemoselectivity. The hydrolysis step post-reaction is particularly effective; by adding water and heating to 40°C to 100°C, any remaining reactive intermediates or unreacted anhydrides are quenched and converted into water-soluble species that partition into the aqueous phase. This leaves the desired 5-chloro-4-formyl oxazole in the organic layer with high fidelity. The ability to tune the R-groups on the glycine derivative and the formamide allows for the synthesis of a diverse library of analogs while maintaining this clean impurity profile, which is essential for meeting the rigorous quality standards of global pharmaceutical regulators.
How to Synthesize 5-Chloro-4-Formyl Oxazole Efficiently
Implementing this synthesis requires careful attention to stoichiometry and thermal management to maximize the benefits of the BTC chemistry. The patent outlines a generalized procedure that is adaptable to various substrates, making it a versatile tool for process chemists. The key to success lies in the initial mixing at lower temperatures to ensure homogeneity before ramping up to the reaction temperature, followed by a controlled hydrolysis workup. This approach minimizes side reactions and ensures consistent batch-to-batch quality. For detailed operational parameters, including specific solvent volumes and stirring rates, the following standardized guide provides the essential framework for laboratory and pilot-scale execution.
- Mix glycine derivatives, bis(trichloromethyl)carbonate (BTC), and formamide compounds in an organic solvent like toluene at -20°C to 30°C.
- Heat the reaction mixture to 50°C to 100°C and maintain for 2 to 4 hours to facilitate cyclization and formylation.
- Add water to hydrolyze the mixture at 50°C, separate the organic layer, dry, evaporate solvent, and recrystallize to obtain the pure product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this BTC-based synthesis represents a strategic opportunity to optimize costs and mitigate risk. The shift away from phosphorus oxychloride is not merely a technical improvement but a significant supply chain enhancement. POCl3 is a regulated substance in many jurisdictions due to its dual-use potential and high toxicity, often leading to volatile pricing and restricted availability. In contrast, BTC is a stable, solid reagent that is easier to transport, store, and handle, ensuring a more reliable supply continuity. Furthermore, the reduction in hazardous waste generation directly impacts the bottom line by lowering waste disposal fees and reducing the regulatory compliance burden associated with phosphorus effluent.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven by both raw material efficiency and waste management savings. By eliminating the need for expensive phosphorus waste treatment infrastructure, manufacturers can achieve substantial cost reductions in pharma manufacturing. The higher yields reported in the patent examples, reaching up to 83% compared to the historical 56%, mean that less raw material is required to produce the same amount of active intermediate. Additionally, the simplified workup procedure reduces solvent consumption and energy usage during the purification stages. These factors combine to lower the overall cost of goods sold (COGS), providing a competitive pricing advantage in the global market for fine chemical intermediates.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of widely available and stable reagents. BTC and common solvents like toluene or xylene are commodity chemicals with robust global supply networks, reducing the risk of shortages that can plague specialized reagents. The process tolerance for a wide range of temperatures (-20°C to 150°C) and solvent systems offers flexibility in sourcing; if one solvent becomes scarce or expensive, the protocol can be adapted to use alternatives like dichloromethane or ethyl acetate without compromising the reaction outcome. This flexibility ensures that production schedules remain uninterrupted even amidst market fluctuations, securing the continuity of supply for downstream API manufacturers.
- Scalability and Environmental Compliance: Scaling this process from kilogram to multi-ton production is straightforward due to the absence of highly corrosive liquids and the simplicity of the unit operations involved. The reaction does not require exotic catalysts or extreme pressure conditions, allowing it to be run in standard glass-lined or stainless steel reactors found in most multipurpose chemical plants. From an environmental standpoint, the elimination of phosphorus discharge aligns perfectly with increasingly strict global environmental, social, and governance (ESG) mandates. This compliance not only avoids potential fines but also enhances the brand reputation of the manufacturer as a sustainable partner, which is a growing requirement for contracts with major multinational pharmaceutical corporations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis method. These insights are derived directly from the experimental data and claims within the patent documentation, providing clarity on how this technology can be integrated into existing manufacturing workflows. Understanding these details is essential for technical teams evaluating the feasibility of adopting this greener and more efficient route for their specific product portfolios.
Q: How does this BTC-based method improve upon traditional Vilsmeier reactions?
A: Traditional methods utilize phosphorus oxychloride (POCl3), generating significant phosphorus-containing waste and posing safety hazards. The patented BTC method eliminates POCl3 entirely, reducing environmental impact and simplifying waste treatment while improving yields from approximately 56% to over 80%.
Q: What are the optimal reaction conditions for maximum yield?
A: According to patent data, optimal results are achieved using toluene or xylene as solvents, with a molar ratio of glycine derivative to formamide to BTC of roughly 1:3-10:1-2. Reaction temperatures between 50°C and 100°C for 2 to 4 hours provide the best balance of conversion and selectivity.
Q: Is this process scalable for industrial production?
A: Yes, the process is highly scalable. It utilizes commercially available reagents like BTC and avoids hazardous phosphorus reagents that require specialized handling infrastructure. The workup involves simple aqueous hydrolysis and recrystallization, which are standard unit operations in large-scale chemical manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Chloro-4-Formyl Oxazole Supplier
The transition to advanced synthetic methods like the BTC-mediated cyclization requires a partner with deep technical expertise and proven scale-up capabilities. NINGBO INNO PHARMCHEM stands at the forefront of this evolution, offering comprehensive CDMO services tailored to the complex needs of the pharmaceutical industry. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising results seen in the laboratory are faithfully reproduced on an industrial scale. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of 5-chloro-4-formyl oxazole meets the highest international standards for impurity profiles and physical properties.
We invite you to collaborate with us to leverage this innovative technology for your next project. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis that evaluates how switching to this phosphorus-free route can impact your specific budget and timeline. We encourage potential partners to contact our technical procurement team to request specific COA data and route feasibility assessments. Let us help you secure a sustainable, cost-effective, and reliable supply of high-quality pharmaceutical intermediates that drive your drug development programs forward.
