Scalable Synthesis of 2-Halo-1-(2-(2,4-dimethylphenoxy)phenyl)ethanone for Antifungal API Production
The pharmaceutical industry's relentless pursuit of efficient antifungal therapies has placed significant spotlight on the synthesis of Abafungin, a broad-spectrum agent developed by York Pharma. Central to the commercial viability of this active pharmaceutical ingredient (API) is the robust production of its key precursor, 2-halo-1-(2-(2,4-dimethylphenoxy)phenyl)ethanone. Patent CN101823946A introduces a transformative methodology for preparing this critical intermediate, addressing long-standing challenges in regioselectivity and environmental impact. By utilizing N-haloimidazolidine-2,4-diones (hydantoins) in conjunction with silica gel catalysis, this innovation offers a pathway that bypasses the harsh conditions and toxic by-products associated with traditional halogenation techniques. For R&D directors and process chemists, this represents a pivotal shift towards greener chemistry without compromising yield or scalability.
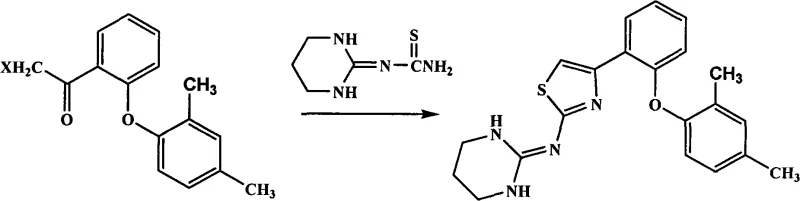
The strategic importance of this intermediate cannot be overstated, as it serves as the electrophilic partner in the final cyclization step to form the thiazole ring of Abafungin. As illustrated in the reaction scheme above, the efficiency of this coupling dictates the overall throughput of the API manufacturing line. Historically, the supply chain for such specialized heterocyclic intermediates has been bottlenecked by purification complexities. However, the technical breakthrough detailed in CN101823946A suggests that high-purity material can be generated with minimal downstream processing. This capability is essential for reliable pharmaceutical intermediates supplier networks aiming to support large-scale clinical and commercial demands for antifungal medications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art, specifically US Patent 4956370, established the baseline for synthesizing this ketone derivative using sulfuryl chloride as the chlorinating agent. While conceptually straightforward, this legacy approach suffers from severe practical deficiencies that hinder industrial adoption. Experimental data indicates that the sulfuryl chloride method yields a conversion rate of merely 70%, leaving a substantial quantity of unreacted starting material that complicates isolation. More critically, the reaction lacks selectivity, generating significant amounts of dichlorinated by-products that possess physical properties nearly identical to the desired mono-chlorinated target. Separating these impurities typically requires resource-intensive column chromatography, a technique that is economically unfeasible for multi-ton production. Furthermore, the stoichiometric generation of sulfonic acid waste creates a heavy environmental burden, necessitating costly neutralization and disposal protocols that inflate the total cost of ownership.
The Novel Approach
In stark contrast, the methodology disclosed in CN101823946A leverages dichlorohydantoin or dibromohydantoin as the halogen source, mediated by silica gel. This novel approach fundamentally alters the reaction landscape by providing a controlled release of the halogen species, thereby enhancing selectivity for the alpha-position of the ketone. The process operates under mild thermal conditions, typically between 30-50°C for chlorination, which significantly reduces energy consumption compared to refluxing methods. Most importantly, the reaction achieves a conversion rate of 95%, drastically minimizing raw material loss. The resulting crude product exhibits a purity of 85-90%, which is sufficiently high to proceed directly to the next synthetic step without intermediate purification. This elimination of the purification bottleneck is a game-changer for cost reduction in API manufacturing.
Mechanistic Insights into Silica Gel Catalyzed Halogenation
The success of this synthetic route lies in the unique interplay between the solid acid catalyst and the N-halo reagent. Silica gel, traditionally viewed merely as a stationary phase, functions here as a heterogeneous Lewis acid that activates the carbonyl group of the substrate, facilitating enolization. This activation is crucial because the substrate, 1-(2-(2,4-dimethylphenoxy)phenyl)ethanone, contains two potential sites for electrophilic attack: the alpha-methyl group adjacent to the carbonyl and the benzylic methyl group on the phenoxy ring. Conventional strong acids or radical conditions often lead to indiscriminate halogenation at the benzylic position, creating inseparable impurities. The silica-hydantoin system, however, creates a microenvironment that favors thermodynamic control, directing the halogen exclusively to the more acidic alpha-protons of the acetyl group. This regioselectivity is the cornerstone of the method's high purity profile.
Furthermore, the mechanism avoids the formation of free molecular halogen (Cl2 or Br2) in the bulk solution, which is a primary driver of side reactions in traditional methods. Instead, the halogen is transferred directly from the hydantoin ring to the enolized substrate via a concerted or semi-concerted transition state stabilized by the silanol groups on the catalyst surface. This "on-water" or "solid-supported" character minimizes the exposure of the sensitive ether linkage to harsh acidic conditions, preserving the integrity of the biaryl ether scaffold. For process chemists, understanding this mechanism allows for fine-tuning of catalyst loading (0.01 to 1.0 mass ratio) to optimize reaction kinetics without triggering over-halogenation, ensuring a consistent impurity profile suitable for GMP manufacturing.
How to Synthesize 2-Halo-1-(2-(2,4-dimethylphenoxy)phenyl)ethanone Efficiently
Implementing this protocol in a pilot or production plant requires precise control over stoichiometry and addition rates to maintain the delicate balance between conversion and selectivity. The patent outlines a robust procedure where the halogenating agent is added portion-wise or dropwise to a suspension of the ketone and silica gel in an alcoholic solvent. This controlled addition prevents local exotherms that could degrade the selectivity of the reaction. The detailed standardized synthesis steps for replicating this high-efficiency route are provided in the guide below.
- Dissolve 1-(2-(2,4-dimethylphenoxy)phenyl)ethanone in an alcoholic solvent such as methanol or ethanol, optionally mixed with acetonitrile.
- Add commercially available silica gel (200-300 mesh) as a catalyst, typically 0.01 to 1.0 times the mass of the raw material.
- Introduce the halogenating agent (dichlorohydantoin or dibromohydantoin) at 0.8 to 1.0 molar equivalents while maintaining temperature between 30-50°C for chlorination or 25-35°C for bromination.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition from sulfuryl chloride-based methods to the hydantoin-silica gel protocol offers profound logistical and financial benefits. The primary advantage stems from the drastic simplification of the workup procedure. By eliminating the need for column chromatography or complex recrystallization sequences to remove dichloro-impurities, manufacturers can significantly reduce solvent consumption and processing time. This streamlining translates directly into lower operational expenditures and a smaller environmental footprint, aligning with modern sustainability mandates. Additionally, the reagents involved—dichlorohydantoin and silica gel—are commodity chemicals with stable global supply chains, reducing the risk of raw material shortages that often plague specialized halogenating agents.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the high atom economy and the removal of purification steps. Since the crude product meets the quality specifications for the subsequent coupling reaction, the costs associated with additional solvents, filtration media, and labor for purification are effectively eliminated. This creates a leaner manufacturing process where the yield of the final API is improved due to reduced material handling losses. The avoidance of sulfonic acid waste also lowers waste treatment costs, contributing to substantial cost savings in the overall production budget.
- Enhanced Supply Chain Reliability: Reliance on liquid bromine or sulfuryl chloride introduces significant safety and regulatory hurdles, as these are hazardous materials requiring special transport and storage permits. By switching to solid N-halo reagents, the facility reduces its regulatory burden and improves onsite safety profiles. Furthermore, the mild reaction conditions (30-50°C) allow the use of standard glass-lined or stainless steel reactors without the need for exotic corrosion-resistant alloys, increasing the number of eligible contract manufacturing organizations (CMOs) capable of producing this intermediate. This flexibility ensures reducing lead time for high-purity pharmaceutical intermediates and guarantees continuity of supply.
- Scalability and Environmental Compliance: The heterogeneous nature of the catalyst simplifies scale-up, as heat transfer and mixing are more manageable compared to homogeneous acid catalysis. The process generates minimal hazardous waste, primarily consisting of spent hydantoin by-products which are easier to handle than corrosive acid streams. This aligns with green chemistry principles, making the process more attractive for facilities operating under strict environmental regulations. The ability to run the reaction in common solvents like methanol or ethanol further enhances the scalability, allowing for seamless transition from kilogram to multi-ton batches without re-optimizing the solvent system.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this halogenation technology. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation, providing clarity on process robustness and product quality.
Q: Why is silica gel used instead of traditional Lewis acids in this halogenation?
A: Silica gel acts as a mild, heterogeneous catalyst that promotes regioselective alpha-halogenation while minimizing benzylic substitution side reactions, which are common with stronger acidic conditions or liquid bromine.
Q: Does the crude product require purification before the next synthetic step?
A: No, the patent explicitly states that the product obtained (85-90% purity) can be directly used in the subsequent condensation reaction to form Abafungin without further purification like column chromatography.
Q: What are the advantages over the sulfuryl chloride method described in US4956370?
A: The hydantoin method avoids the generation of sulfonic acid waste, achieves higher conversion rates (95% vs 70%), and prevents the formation of difficult-to-separate dichlorinated by-products.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Halo-1-(2-(2,4-dimethylphenoxy)phenyl)ethanone Supplier
At NINGBO INNO PHARMCHEM, we recognize that the efficiency of your antifungal API synthesis hinges on the quality and consistency of your starting materials. Our technical team has thoroughly analyzed the pathway described in CN101823946A and integrated these best practices into our own manufacturing protocols. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements without compromising on stringent purity specifications. Our rigorous QC labs employ advanced HPLC and GC-MS methods to verify that every batch of 2-halo-ketone intermediate meets the critical 85-90% purity threshold required for direct downstream usage, guaranteeing a smooth workflow for your synthesis teams.
We invite you to collaborate with us to optimize your supply chain for Abafungin and related antifungal agents. By leveraging our expertise in this specific halogenation chemistry, we can offer a Customized Cost-Saving Analysis tailored to your current production bottlenecks. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how our optimized intermediates can enhance your overall process efficiency.
