Scalable Production of Moxifloxacin Key Intermediate Using Optimized Decarboxylation Technology
Scalable Production of Moxifloxacin Key Intermediate Using Optimized Decarboxylation Technology
The pharmaceutical industry continuously seeks robust synthetic pathways for critical antibiotic intermediates, particularly for third-generation quinolones like Moxifloxacin. Patent CN110746334A introduces a transformative methodology for preparing 3-(3-chloropropyl)-4-oxopyrrolidine-1-carboxylate, a pivotal building block in the synthesis of the bicyclic amine core found in Moxifloxacin. This innovation addresses long-standing challenges in step economy and waste generation associated with traditional manufacturing protocols. By shifting from complex protection-deprotection sequences to a streamlined four-step process, this technology offers a compelling value proposition for reliable pharmaceutical intermediate suppliers aiming to enhance production efficiency. The strategic reordering of synthetic steps, specifically the early introduction of the carbamate protecting group followed by cyclization, fundamentally alters the impurity profile and operational complexity of the manufacture.
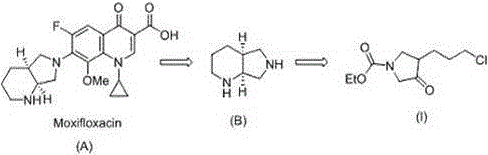
Moxifloxacin remains a cornerstone in the treatment of respiratory tract infections due to its broad spectrum and favorable pharmacokinetic profile. However, the economic viability of its production hinges on the cost-effectiveness of its precursors. The intermediate designated as (I) in the patent literature serves as the direct precursor to the octahydro-1H-pyrrolo[3,4-b]pyridine core. Traditional approaches to accessing this scaffold often relied on harsh conditions or expensive reagents that complicated scale-up. The disclosed method leverages common industrial chemicals such as glycine esters and acrylates, positioning it as a highly attractive route for cost reduction in API manufacturing. For procurement managers and supply chain heads, understanding the nuances of this pathway is essential for securing long-term supply continuity and mitigating risks associated with raw material volatility.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of the pyrrolidine core for Moxifloxacin involved arduous multi-step sequences that were fraught with inefficiencies. Early methodologies, such as those utilizing 3,4-pyridinedicarboxylic acid, necessitated high-pressure hydrogenation and expensive reducing agents, creating significant barriers to entry for many manufacturers due to equipment costs and safety concerns. Furthermore, subsequent patents like CN103044418B introduced routes based on benzyl protection strategies. While an improvement, these methods typically required six distinct synthetic steps to reach the target intermediate. The reliance on benzyl groups introduced additional purification burdens, as the removal of the benzyl moiety often required specific hydrogenation or cleavage conditions that could compromise the integrity of sensitive functional groups. Moreover, the intermediates generated in these longer sequences were frequently unstable, leading to difficult purification processes and inconsistent batch-to-batch quality.
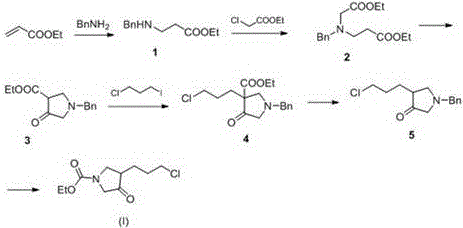
The instability of intermediates in conventional routes posed a severe challenge for industrial control. Long reaction sequences inherently accumulate yield losses at each stage, often resulting in overall yields hovering around 50%. Additionally, the generation of hazardous waste was significant, particularly when strong bases were used for final decarboxylation steps, which frequently triggered unwanted self-reaction of the product. These factors combined to create a high-cost, high-waste manufacturing environment that struggled to meet the growing global demand for fluoroquinolone antibiotics. For supply chain leaders, these legacy processes represented a vulnerability, as any disruption in the supply of specialized reagents or catalysts could halt production entirely.
The Novel Approach
In stark contrast, the methodology outlined in CN110746334A presents a streamlined four-step synthesis that elegantly bypasses the pitfalls of its predecessors. By initiating the sequence with the reaction of glycine ester hydrochloride and chloroformate, the process immediately establishes the necessary nitrogen protection without the need for benzyl groups. This strategic shift eliminates the requirement for subsequent de-benzylation steps, effectively shortening the critical path of the synthesis. The core of this innovation lies in the tandem cyclization and Dieckmann condensation, which constructs the pyrrolidone ring in a single operational step from the linear precursor. This convergence not only saves time but also drastically reduces solvent consumption and labor costs associated with isolating multiple intermediates.
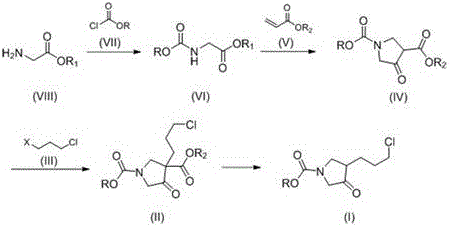
Perhaps the most significant advancement in this novel approach is the implementation of acidic decarboxylation for the final transformation. Unlike previous methods that relied on basic conditions which promoted side reactions, this protocol utilizes sulfuric acid in an alcohol-water mixture. This change in reaction milieu stabilizes the final product, preventing the degradation and oligomerization observed in alkaline environments. Furthermore, the patent highlights the potential for recycling the aqueous acid phase, a feature that aligns perfectly with modern green chemistry principles. For a reliable pharmaceutical intermediate supplier, adopting this route translates directly into enhanced process robustness and a substantially lower environmental footprint, making it an ideal candidate for commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Acidic Decarboxylation and Cyclization
The mechanistic elegance of this synthesis is rooted in the precise control of reactivity at each stage. The initial formation of the carbamate (VI) from glycine ester (VIII) serves a dual purpose: it protects the amine from over-alkylation in subsequent steps and activates the alpha-proton for the ensuing cyclization. When reacted with acrylate (V) under basic conditions, the system undergoes a Michael addition followed by an intramolecular Dieckmann condensation. This cascade reaction efficiently closes the five-membered ring to form the 4-oxopyrrolidine scaffold (IV). The choice of base, such as sodium methoxide or sodium ethoxide, is critical here to ensure complete conversion while minimizing hydrolysis of the ester groups. This one-pot transformation is a masterclass in atom economy, merging two bond-forming events into a single unit operation.
The final step, acidic decarboxylation, represents a paradigm shift in handling beta-keto ester derivatives. In traditional basic hydrolysis, the resulting enolate is highly nucleophilic and prone to attacking other molecules of the substrate, leading to polymeric by-products. By switching to an acidic medium, specifically using sulfuric acid, the decarboxylation proceeds via a protonated transition state that facilitates the loss of carbon dioxide without generating a reactive nucleophile. This mechanism ensures that the 3-(3-chloropropyl) side chain remains intact and the pyrrolidone ring is preserved. The ability to tune the acidity and temperature (40-60°C) allows for precise control over the reaction rate, ensuring high purity of the final ester (I). This mechanistic understanding is vital for R&D directors focused on impurity control and regulatory compliance.
How to Synthesize 3-(3-chloropropyl)-4-oxopyrrolidine-1-carboxylate Efficiently
Implementing this synthesis requires careful attention to reaction parameters to maximize the reported yields of over 70%. The process begins with the neutralization of glycine ester hydrochloride, followed by acylation. The subsequent cyclization step demands strict temperature control to prevent polymerization of the acrylate. Finally, the alkylation with 1,3-halochloropropane must be managed to avoid di-alkylation, although the carbamate protection largely mitigates this risk. The detailed standardized synthetic steps below outline the specific conditions, reagents, and workup procedures necessary to replicate the high efficiency described in the patent documentation.
- React glycine ester hydrochloride with chloroformate in the presence of a base like sodium carbonate to form the ethoxycarbonyl glycine ester intermediate.
- Perform a one-pot cyclization and Dieckmann condensation by reacting the glycine ester intermediate with acrylate under basic conditions to form the pyrrolidone ring.
- Alkylate the pyrrolidone intermediate using 1,3-halochloropropane (such as 1-bromo-3-chloropropane) to introduce the chloropropyl side chain.
- Execute acidic decarboxylation using sulfuric acid in an alcohol-water mixture to remove the ester group and yield the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology offers tangible strategic benefits beyond mere technical superiority. The reduction in synthetic steps from six to four directly correlates with a significant reduction in manufacturing lead time and operational expenditure. By eliminating the need for high-pressure hydrogenation equipment and expensive chiral resolution agents, the capital expenditure required for production facilities is markedly lower. Furthermore, the use of commodity chemicals like glycine esters and chloroformates ensures a stable and diversified supply base, reducing the risk of bottlenecks associated with specialty reagents. This stability is crucial for maintaining continuous supply lines in the volatile pharmaceutical market.
- Cost Reduction in Manufacturing: The streamlined four-step process inherently lowers the cost of goods sold by reducing labor hours, solvent usage, and energy consumption. The elimination of the benzyl protection group removes the cost associated with benzyl halides and the subsequent hydrogenation or cleavage steps. Additionally, the high overall yield (>70%) means less raw material is wasted per kilogram of finished product. The ability to recycle the sulfuric acid solution in the final step further contributes to cost savings by minimizing waste disposal fees and reagent procurement costs.
- Enhanced Supply Chain Reliability: Relying on widely available starting materials such as glycine ethyl ester hydrochloride and ethyl acrylate insulates the supply chain from the volatility of niche chemical markets. The robustness of the acidic decarboxylation step ensures consistent product quality, reducing the likelihood of batch failures that can disrupt delivery schedules. This reliability allows supply chain planners to optimize inventory levels and respond more agilely to fluctuations in downstream demand for Moxifloxacin.
- Scalability and Environmental Compliance: The process is designed with industrial scalability in mind, avoiding unit operations that are difficult to scale, such as high-pressure hydrogenation. The significant reduction in three wastes (wastewater, waste gas, and solid waste) aligns with increasingly stringent environmental regulations globally. The acidic workup generates less hazardous organic waste compared to basic hydrolysis routes, simplifying effluent treatment and lowering the environmental compliance burden for manufacturing sites.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis of this critical Moxifloxacin intermediate. These insights are derived directly from the comparative data and experimental examples provided in the patent literature, offering a clear picture of the technology's advantages over legacy methods. Understanding these details is essential for technical teams evaluating the feasibility of technology transfer.
Q: Why is acidic decarboxylation preferred over basic conditions for this intermediate?
A: Previous methods utilized basic conditions for decarboxylation, which often led to self-reaction of the product and significant by-product formation. The novel method employs acidic conditions (e.g., sulfuric acid), which stabilizes the product, minimizes side reactions, and allows for the recycling of the acid solution, thereby reducing waste.
Q: What is the overall yield improvement compared to conventional benzyl-protection routes?
A: Conventional routes involving benzyl protection and high-pressure hydrogenation typically achieved yields around 50%. The optimized process described in patent CN110746334A streamlines the synthesis to four steps and achieves a total yield exceeding 70%, representing a substantial increase in material efficiency.
Q: What are the primary starting materials for this synthesis?
A: The process utilizes readily available and cost-effective raw materials, specifically glycine ester hydrochlorides (such as glycine ethyl ester hydrochloride) and chloroformates. This avoids the need for expensive reducing agents or complex chiral resolution steps required in older methodologies.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-(3-chloropropyl)-4-oxopyrrolidine-1-carboxylate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the global supply of life-saving antibiotics. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the efficiencies demonstrated in patent CN110746334A can be fully realized at an industrial level. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 3-(3-chloropropyl)-4-oxopyrrolidine-1-carboxylate meets the exacting standards required for API synthesis. Our commitment to process optimization allows us to deliver cost-effective solutions without compromising on quality or regulatory compliance.
We invite pharmaceutical partners to engage with our technical procurement team to discuss how this advanced synthetic route can benefit your specific supply chain needs. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic impact of switching to this optimized methodology. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your production requirements, ensuring a seamless integration of this high-performance intermediate into your manufacturing portfolio.
