Revolutionizing Acetylhydroxyproline Production via Advanced Biocatalysis for Commercial Scale
Revolutionizing Acetylhydroxyproline Production via Advanced Biocatalysis for Commercial Scale
The landscape of fine chemical manufacturing is undergoing a paradigm shift towards greener, more efficient biocatalytic processes, as exemplified by the groundbreaking technology disclosed in patent CN113403350A. This patent introduces a highly sophisticated method for synthesizing N-acetyl-L-4-hydroxyproline, a critical active ingredient widely utilized in the cosmetic and pharmaceutical industries for its potent anti-aging and moisturizing properties. By leveraging the specificity of biological enzymes, specifically acyltransferases, this innovation bypasses the traditional limitations of chemical synthesis, offering a route that is not only environmentally benign but also economically superior for large-scale operations. The core of this technology lies in the direct transesterification of hydroxyproline with various acetates in an aqueous phase, achieving remarkable conversion rates without the need for toxic solvents or extreme reaction conditions. For industry leaders seeking a reliable cosmetic ingredient supplier, understanding the nuances of this enzymatic pathway is essential for securing a competitive edge in the market.
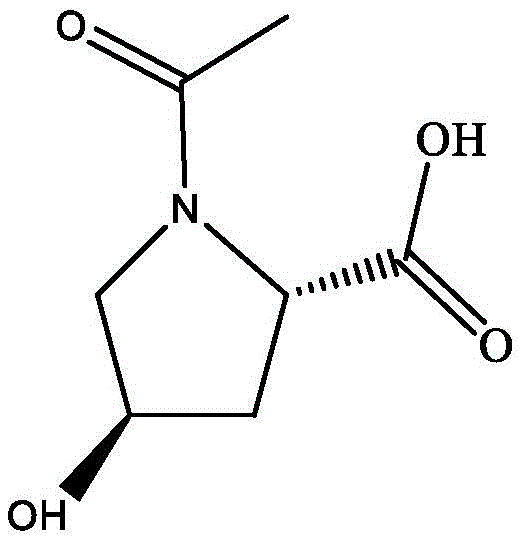
N-acetyl-L-4-hydroxyproline serves as a vital building block in the formulation of advanced skincare products, known for its ability to promote collagen synthesis and enhance skin barrier function. The molecular architecture of this compound, characterized by the acetylation of the secondary amine group on the proline ring, is crucial for its bioactivity and stability in final formulations. Traditional manufacturing approaches have often struggled with regioselectivity issues, where unwanted O-acetylation occurs alongside the desired N-acetylation, leading to complex purification challenges and reduced overall yields. However, the methodology presented in CN113403350A elegantly solves this problem by utilizing the inherent selectivity of the MsACT enzyme, ensuring that the acetyl group is transferred exclusively to the nitrogen atom. This precision not only simplifies the downstream processing but also guarantees a final product of exceptional purity, meeting the stringent quality standards required by global regulatory bodies for both cosmetic and quasi-drug applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of acetylated amino acids like N-acetyl-L-4-hydroxyproline has relied heavily on classical organic synthesis techniques that are fraught with significant operational and environmental drawbacks. Conventional chemical acetylation typically involves the use of aggressive acetylating agents such as acetic anhydride or acetyl chloride, which necessitate strictly anhydrous conditions and the use of hazardous organic solvents like dichloromethane or tetrahydrofuran. These reactions are often highly exothermic, requiring precise temperature control to prevent runaway reactions and the formation of undesirable by-products, which complicates the safety profile of the manufacturing plant. Furthermore, chemical methods frequently lack the necessary chemoselectivity, leading to the simultaneous acetylation of the hydroxyl group on the proline ring, thereby generating difficult-to-separate impurities that degrade the quality of the final active ingredient. The subsequent workup procedures usually involve multiple neutralization steps, extensive washing, and energy-intensive distillation processes to remove residual solvents and salts, all of which contribute to a substantial increase in production costs and a heavy environmental footprint due to the generation of large volumes of chemical waste.
The Novel Approach
In stark contrast to these archaic methods, the novel enzymatic approach detailed in the patent offers a streamlined, one-step synthesis that operates under mild, physiological conditions, fundamentally altering the economics of production. By employing an acyltransferase catalyst in a phosphate-buffered saline (PBS) solution, the reaction proceeds efficiently at temperatures ranging from 25°C to 45°C, eliminating the need for energy-intensive heating or cooling systems. The use of simple alkyl acetates, such as ethyl acetate or vinyl acetate, as acyl donors not only reduces raw material costs but also ensures that the by-products of the reaction are benign alcohols or acetaldehyde, which are easily removed or biodegraded. This aqueous-phase strategy inherently suppresses the formation of side products, as the enzyme's active site provides a highly specific microenvironment that favors N-acylation over O-acylation. Consequently, the downstream purification process is drastically simplified, often requiring only a pH adjustment, filtration, and a single extraction step to isolate the product with purity levels exceeding 98%, representing a quantum leap in cost reduction in cosmetic ingredient manufacturing.
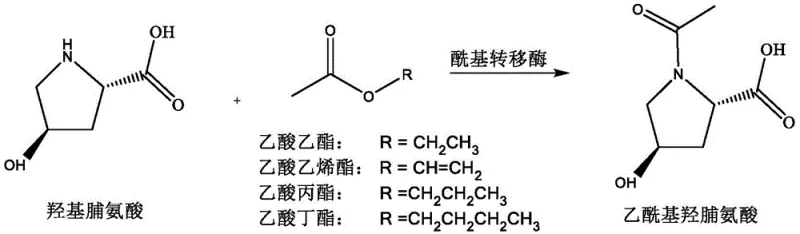
Mechanistic Insights into Acyltransferase-Catalyzed Transesterification
The success of this synthetic route hinges on the sophisticated mechanistic action of the acyltransferase enzyme, specifically the MsACT variant, which facilitates the transfer of an acyl group from an ester donor to the nucleophilic amine of hydroxyproline. Unlike chemical catalysts that rely on brute force thermodynamics, this biocatalyst operates through a precise lock-and-key mechanism where the substrate binds to the enzyme's active site in a specific orientation that exposes the secondary amine group while shielding the hydroxyl group. This spatial arrangement ensures that the nucleophilic attack occurs exclusively at the nitrogen atom, effectively preventing the formation of O-acetyl impurities that plague chemical synthesis. The reaction kinetics are further optimized by the aqueous buffer system, which maintains the enzyme in its active conformation and stabilizes the transition state, allowing for high turnover numbers even at relatively low catalyst loadings. The patent data indicates that conversion rates can consistently reach 95% to 99% within 24 hours, demonstrating the robustness of the catalytic cycle and the stability of the enzyme under the specified reaction conditions of pH 7.0 to 10.0.
From an impurity control perspective, the enzymatic mechanism offers distinct advantages that are critical for producing high-purity acetylhydroxyproline suitable for sensitive dermatological applications. In chemical synthesis, the presence of trace metals or acidic residues from the acetylating agents can catalyze degradation pathways or racemization of the chiral center, compromising the optical purity of the product. However, the biological nature of the MsACT catalyst ensures that the stereochemistry of the L-hydroxyproline starting material is preserved throughout the reaction, as the enzyme is inherently chiral and selective for the L-enantiomer. Moreover, the absence of harsh reagents means that there is no risk of introducing toxic heavy metal contaminants, which is a major concern for cosmetic ingredients intended for long-term topical use. The final product, therefore, exhibits a clean impurity profile with no protein residue, as the enzyme can be easily separated via filtration or denaturation, ensuring that the final active ingredient meets the rigorous safety specifications demanded by international regulatory agencies.
How to Synthesize Acetylhydroxyproline Efficiently
The practical implementation of this enzymatic synthesis is designed to be straightforward and adaptable to various scales of production, making it an ideal candidate for technology transfer and industrial adoption. The process begins with the preparation of a reaction mixture containing a buffered aqueous solution, the hydroxyproline substrate, and a slight excess of the acetate donor, creating a homogeneous or biphasic system depending on the solubility of the ester. Once the acyltransferase is introduced, the reaction is allowed to proceed under gentle stirring and nitrogen purging to prevent oxidative degradation of the enzyme or substrates. Detailed standard operating procedures regarding specific molar ratios, agitation speeds, and monitoring intervals are critical for maximizing yield and ensuring batch-to-batch consistency.
- Prepare an aqueous reaction system by adding PBS buffer solution, hydroxyproline substrate, and acetate donor into a reaction vessel under nitrogen atmosphere.
- Introduce the acyltransferase catalyst (MsACT) to the mixture and maintain stirring at controlled temperatures between 25°C and 45°C.
- Monitor conversion via HPLC, adjust pH to 3-5 upon completion, filter through celite, extract with dichloromethane, and purify via rotary evaporation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this enzymatic manufacturing process represents a strategic opportunity to optimize cost structures and mitigate supply risks associated with traditional chemical synthesis. The shift from hazardous organic solvents to water as the primary reaction medium significantly reduces the costs associated with solvent procurement, storage, and disposal, while also lowering the insurance premiums related to fire and safety hazards in the production facility. Furthermore, the use of commodity chemicals like ethyl acetate as acyl donors ensures a stable and predictable supply chain, insulating the manufacturer from the volatility of specialized reagent markets. The simplicity of the workup procedure, which avoids complex chromatographic separations, translates directly into shorter production cycles and higher throughput, allowing suppliers to respond more rapidly to fluctuating market demands without compromising on quality.
- Cost Reduction in Manufacturing: The elimination of expensive protecting groups and harsh acetylating agents leads to substantial cost savings in raw material expenditure, while the mild reaction conditions reduce energy consumption for heating and cooling. The high selectivity of the enzyme minimizes the loss of valuable starting materials to side reactions, thereby improving the overall atom economy of the process and reducing the cost per kilogram of the final active ingredient. Additionally, the reduced need for extensive purification steps lowers the operational expenses related to labor, equipment maintenance, and waste treatment, contributing to a leaner and more profitable manufacturing model.
- Enhanced Supply Chain Reliability: By utilizing robust enzymes and stable substrates that can be sourced from multiple global suppliers, manufacturers can build a more resilient supply chain that is less susceptible to disruptions caused by geopolitical tensions or raw material shortages. The aqueous nature of the process also simplifies logistics, as there is no need for specialized transport containers for flammable or corrosive solvents, further streamlining the distribution network. This reliability ensures consistent delivery schedules for downstream customers, fostering stronger long-term partnerships and enhancing the reputation of the supplier as a dependable source of critical cosmetic ingredients.
- Scalability and Environmental Compliance: The inherent safety and simplicity of the enzymatic route make it exceptionally easy to scale from pilot plant trials to full commercial production without the engineering challenges associated with managing exothermic chemical reactions. The process aligns perfectly with green chemistry principles, generating minimal hazardous waste and reducing the carbon footprint of the manufacturing operation, which is increasingly important for meeting corporate sustainability goals. This environmental compliance not only future-proofs the production facility against tightening regulations but also appeals to eco-conscious consumers and brands looking to differentiate their products based on sustainable sourcing practices.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the enzymatic synthesis of acetylhydroxyproline, providing clarity on the feasibility and benefits of adopting this innovative technology. These insights are derived directly from the experimental data and technical specifications outlined in the patent documentation, ensuring accuracy and relevance for industry professionals.
Q: What are the primary advantages of the enzymatic method over chemical synthesis for acetylhydroxyproline?
A: The enzymatic method described in patent CN113403350A operates in mild aqueous conditions, eliminating the need for harsh chemical reagents and complex protection-deprotection steps, resulting in higher purity and reduced environmental impact.
Q: Which acyltransferase is preferred for this transesterification reaction?
A: The patent specifically highlights the use of MsACT acyltransferase, which demonstrates high conversion rates and excellent stability in phosphate-buffered saline solutions.
Q: Is this process suitable for large-scale industrial manufacturing?
A: Yes, the process utilizes cheap and readily available raw materials like ethyl acetate and operates in water, making it highly scalable and cost-effective for commercial production compared to traditional organic solvent-based methods.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Acetylhydroxyproline Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the enzymatic synthesis route described in CN113403350A and are fully equipped to leverage this technology for the benefit of our global clientele. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with unwavering consistency and quality. Our state-of-the-art facilities are designed to handle complex biocatalytic processes with precision, featuring stringent purity specifications and rigorous QC labs that guarantee every batch of acetylhydroxyproline meets the highest industry standards. We understand that in the fast-paced world of cosmetic and pharmaceutical development, time-to-market is critical, and our optimized workflows are tailored to accelerate your project timelines without compromising on safety or efficacy.
We invite you to collaborate with us to explore how this advanced manufacturing method can drive value for your specific applications and product lines. Our technical team is ready to provide a Customized Cost-Saving Analysis that quantifies the economic benefits of switching to this enzymatic route for your specific volume requirements. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, allowing you to make informed decisions that will strengthen your supply chain and enhance your competitive position in the market.
