Revolutionizing Triazoloquinoline Production: A Safe, Scalable, and Metal-Free Synthetic Strategy for Global Supply Chains
The pharmaceutical and fine chemical industries are constantly seeking robust synthetic pathways that balance high purity with operational safety, a challenge vividly addressed in the recent technological disclosure found in patent CN111057058A. This specific intellectual property outlines a groundbreaking method for preparing 1H-[1,2,3]-triazolo[4,5-c]quinoline compounds, a class of fused heterocycles with immense potential in drug discovery and material science. Unlike traditional methodologies that rely heavily on toxic reagents and harsh thermal conditions, this novel approach utilizes a metal-free catalytic system driven by fluorine-containing reagents. For R&D directors and procurement specialists alike, understanding the nuances of this patent is critical, as it represents a shift towards greener, more cost-effective manufacturing protocols. The core innovation lies in the tandem Click/dehydration cyclization strategy, which not only streamlines the synthetic route but also drastically reduces the environmental footprint associated with heavy metal waste disposal. By leveraging this technology, manufacturers can achieve superior impurity profiles essential for high-purity pharmaceutical intermediates, ensuring that the final active ingredients meet the rigorous regulatory standards demanded by global health authorities.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of triazoloquinoline derivatives has been plagued by significant safety hazards and inefficiencies that hinder large-scale commercial adoption. As illustrated in prior art, many conventional routes depend on the use of sodium azide (NaN3) in conjunction with stoichiometric amounts of copper salts, often requiring temperatures exceeding 100°C to drive the reaction to completion. 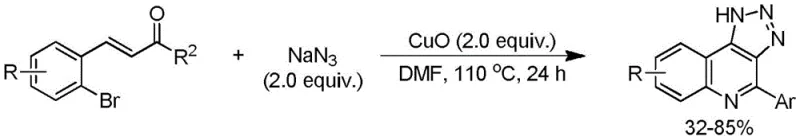 These conditions create a volatile environment where the risk of explosion is markedly elevated, posing severe threats to plant safety and personnel security. Furthermore, the reliance on transition metal catalysts introduces complex downstream processing challenges, as removing trace metal residues to meet ppm-level specifications requires additional purification steps that erode profit margins. The generation of substantial quantities of copper-containing waste also complicates environmental compliance, forcing facilities to invest heavily in waste treatment infrastructure. Additionally, these older methods often suffer from limited substrate scope, failing to efficiently produce 4-alkyl substituted derivatives which are increasingly valuable in modern medicinal chemistry pipelines. The cumulative effect of long reaction times, low yields for specific substrates, and high energy consumption renders these legacy processes economically unsustainable for a reliable agrochemical intermediate supplier or pharma partner seeking long-term viability.
These conditions create a volatile environment where the risk of explosion is markedly elevated, posing severe threats to plant safety and personnel security. Furthermore, the reliance on transition metal catalysts introduces complex downstream processing challenges, as removing trace metal residues to meet ppm-level specifications requires additional purification steps that erode profit margins. The generation of substantial quantities of copper-containing waste also complicates environmental compliance, forcing facilities to invest heavily in waste treatment infrastructure. Additionally, these older methods often suffer from limited substrate scope, failing to efficiently produce 4-alkyl substituted derivatives which are increasingly valuable in modern medicinal chemistry pipelines. The cumulative effect of long reaction times, low yields for specific substrates, and high energy consumption renders these legacy processes economically unsustainable for a reliable agrochemical intermediate supplier or pharma partner seeking long-term viability.
The Novel Approach
In stark contrast to these cumbersome legacy techniques, the methodology described in CN111057058A introduces a paradigm shift by employing azidotrimethylsilane (TMS-N3) as a benign nitrogen source coupled with a catalytic amount of a fluorine-containing reagent. This strategic substitution eliminates the explosive risks associated with inorganic azides while enabling the reaction to proceed smoothly at room temperature, typically between 25°C and 30°C. The absence of transition metals means that the post-reaction workup is significantly simplified, often requiring only standard extraction and chromatography without the need for specialized metal scavengers. This metal-free characteristic is particularly advantageous for cost reduction in electronic chemical manufacturing, where metal contamination can ruin the performance of organic semiconductors or OLED materials. Moreover, the reaction demonstrates exceptional versatility, accommodating a wide range of substituents on the aromatic ring, including electron-withdrawing and electron-donating groups, without compromising yield. By operating under such mild conditions, the process not only enhances safety but also reduces energy costs, making it an ideal candidate for the commercial scale-up of complex polymer additives or specialty chemicals where thermal sensitivity is a concern.
Mechanistic Insights into Fluorine-Catalyzed Cyclization
To fully appreciate the technical superiority of this route, one must delve into the mechanistic intricacies of the fluorine-catalyzed tandem reaction. The process initiates with the activation of the azidotrimethylsilane by the fluoride ion, which acts as a potent nucleophile to generate the reactive azide species in situ. This activated azide then engages in a [3+2] cycloaddition with the alkyne moiety of the β-(2-aminoaryl)-α,β-alkynone substrate, forming a triazole intermediate through a classic Click chemistry mechanism. 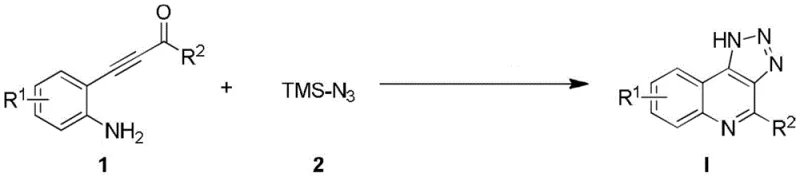 Following the cycloaddition, the proximal amino group facilitates an intramolecular dehydration cyclization, effectively fusing the triazole ring with the quinoline scaffold to yield the target 1H-[1,2,3]-triazolo[4,5-c]quinoline structure. The fluorine catalyst plays a dual role, not only promoting the initial azide formation but also assisting in the proton transfer steps necessary for the final aromatization. This elegant cascade avoids the formation of stable metal-azide complexes that often stall conventional reactions, thereby ensuring high turnover frequencies and excellent atom economy. For the R&D Director, this mechanism offers a clear pathway to optimizing reaction parameters, as the lack of metal coordination spheres simplifies the kinetic modeling and allows for precise control over reaction rates. The result is a highly reproducible process that consistently delivers high-purity OLED material or API precursors with minimal batch-to-batch variation, a critical factor for maintaining supply chain integrity.
Following the cycloaddition, the proximal amino group facilitates an intramolecular dehydration cyclization, effectively fusing the triazole ring with the quinoline scaffold to yield the target 1H-[1,2,3]-triazolo[4,5-c]quinoline structure. The fluorine catalyst plays a dual role, not only promoting the initial azide formation but also assisting in the proton transfer steps necessary for the final aromatization. This elegant cascade avoids the formation of stable metal-azide complexes that often stall conventional reactions, thereby ensuring high turnover frequencies and excellent atom economy. For the R&D Director, this mechanism offers a clear pathway to optimizing reaction parameters, as the lack of metal coordination spheres simplifies the kinetic modeling and allows for precise control over reaction rates. The result is a highly reproducible process that consistently delivers high-purity OLED material or API precursors with minimal batch-to-batch variation, a critical factor for maintaining supply chain integrity.
Controlling the impurity profile is another cornerstone of this mechanistic advantage, particularly regarding the suppression of side reactions common in metal-catalyzed systems. In traditional copper-mediated processes, oxidative homocoupling of alkynes or Glaser coupling often competes with the desired cycloaddition, leading to difficult-to-remove dimeric impurities. The metal-free nature of the fluorine-catalyzed route inherently precludes these oxidative side pathways, resulting in a cleaner crude reaction mixture. Furthermore, the use of TMS-N3 minimizes the formation of hydrazoic acid, a volatile and toxic byproduct often generated when using acidic conditions with sodium azide. This inherent safety feature translates directly into reduced containment costs and simpler ventilation requirements for the manufacturing facility. The ability to run the reaction in common organic solvents like DMF or acetonitrile further enhances its adaptability, allowing for easy solvent recovery and recycling. Consequently, the final product exhibits a superior impurity spectrum, reducing the burden on quality control labs and accelerating the release of high-purity pharmaceutical intermediates to the market.
How to Synthesize 4-Phenyl-1H-[1,2,3]-triazolo[4,5-c]quinoline Efficiently
Implementing this synthesis on a pilot or production scale requires strict adherence to the optimized parameters outlined in the patent to ensure maximum efficiency and safety. The general procedure involves dissolving the β-(2-aminoaryl)-α,β-alkynone starting material in a polar aprotic solvent, followed by the sequential addition of the fluorine catalyst and the azidotrimethylsilane reagent. It is imperative to maintain the reaction temperature within the specified 0°C to 60°C window, although ambient conditions are generally sufficient to drive the transformation to completion within 3 to 8 hours. Detailed standardized synthesis steps see the guide below.
- Mix β-(2-aminoaryl)-α,β-alkynone with azidotrimethylsilane and a fluorine-containing catalyst such as tetrabutylammonium fluoride in a suitable organic solvent like DMF.
- Maintain the reaction mixture at a temperature between 0°C and 60°C, preferably at room temperature, for 3 to 8 hours while monitoring conversion via TLC.
- Upon completion, dilute with water, extract with ethyl acetate, dry over anhydrous sodium sulfate, and purify the crude product via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this metal-free methodology offers tangible benefits that extend far beyond the laboratory bench, impacting the bottom line through operational excellence. The elimination of expensive transition metal catalysts such as palladium or copper directly correlates to a significant reduction in raw material costs, as fluorine salts are commoditized and widely available at a fraction of the price. Additionally, the simplified workup procedure reduces the consumption of auxiliary materials like metal scavengers and specialized filtration media, further driving down the cost of goods sold. The mild reaction conditions also imply lower energy expenditures for heating and cooling, contributing to a more sustainable and economically viable manufacturing process. By adopting this route, companies can mitigate the risks associated with volatile metal prices and supply disruptions, ensuring a more stable cost structure for their high-purity pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The removal of transition metals from the catalytic cycle fundamentally alters the economic model of production by eliminating the need for costly metal recovery and purification steps. Without the requirement for ppm-level metal removal, manufacturers can bypass expensive chromatography resins or scavenger treatments, leading to substantial cost savings in downstream processing. The high atom economy of the Click/dehydration tandem reaction ensures that a greater proportion of raw materials are converted into the final product, minimizing waste disposal fees. Furthermore, the use of inexpensive fluorine catalysts allows for potential catalyst recycling or reduced loading, enhancing the overall process efficiency. These factors combine to create a leaner manufacturing operation that is less susceptible to fluctuations in the global metals market, securing better margins for the reliable pharmaceutical intermediates supplier.
- Enhanced Supply Chain Reliability: Safety is a critical component of supply chain continuity, and the substitution of hazardous sodium azide with the safer azidotrimethylsilane significantly reduces the risk of unplanned shutdowns due to safety incidents. The mild operating conditions reduce the stress on reactor vessels and ancillary equipment, extending asset life and decreasing maintenance downtime. Because the reaction is robust and tolerant of various functional groups, sourcing of diverse starting materials becomes more flexible, reducing dependency on single-source suppliers for specialized reagents. This flexibility ensures that production schedules can be maintained even when specific raw material streams face temporary constraints. Ultimately, a safer and more flexible process translates to more predictable lead times for reducing lead time for high-purity pharmaceutical intermediates, fostering stronger relationships with downstream clients.
- Scalability and Environmental Compliance: Scaling chemical processes often amplifies environmental challenges, but this metal-free route inherently aligns with green chemistry principles by minimizing heavy metal effluent. The absence of copper or palladium waste simplifies wastewater treatment protocols, making it easier to comply with increasingly stringent environmental regulations across different jurisdictions. The ability to run the reaction at room temperature reduces the carbon footprint associated with energy generation, supporting corporate sustainability goals. Moreover, the high yields and broad substrate scope mean that fewer batches are required to meet production targets, reducing the overall volume of solvent and reagent consumption. This scalability ensures that the commercial scale-up of complex pharmaceutical intermediates can be achieved without compromising environmental stewardship or regulatory compliance.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route, providing clarity for stakeholders evaluating its adoption. These answers are derived directly from the experimental data and beneficial effects reported in the patent documentation, ensuring accuracy and relevance for decision-makers. Understanding these details is essential for assessing the feasibility of integrating this technology into existing production lines or new product development pipelines. The insights provided here bridge the gap between theoretical chemistry and practical industrial application.
Q: Why is azidotrimethylsilane preferred over sodium azide in this synthesis?
A: Azidotrimethylsilane is utilized as a safer alternative to sodium azide because it significantly reduces toxicity and explosion risks associated with handling inorganic azides, while maintaining high reactivity under mild conditions.
Q: What are the advantages of using a fluorine-containing catalyst instead of transition metals?
A: Using a fluorine-containing catalyst eliminates the need for expensive transition metals like copper or palladium, thereby simplifying post-treatment processes, removing heavy metal residues, and ensuring the final product meets stringent pharmaceutical purity standards.
Q: Can this method be scaled for industrial production of electronic materials?
A: Yes, the method operates at room temperature with wide substrate applicability and high yields, making it highly suitable for commercial scale-up of complex pharmaceutical intermediates and electronic chemicals without requiring extreme thermal conditions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1H-[1,2,3]-triazolo[4,5-c]quinoline Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the metal-free catalytic route described in CN111057058A and possess the technical expertise to bring this innovation to commercial reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale discovery to industrial manufacturing is seamless and efficient. We understand that achieving stringent purity specifications is non-negotiable for pharmaceutical applications, which is why our rigorous QC labs are equipped to monitor every step of the synthesis, guaranteeing that the final 1H-[1,2,3]-triazolo[4,5-c]quinoline products meet the highest global standards. Our commitment to quality and safety makes us the ideal partner for companies looking to leverage this advanced chemistry for their drug development programs.
We invite you to collaborate with us to optimize your supply chain and reduce manufacturing costs through the adoption of this superior synthetic methodology. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. We encourage you to contact us to request specific COA data and route feasibility assessments, allowing you to make informed decisions about integrating this metal-free technology into your portfolio. By partnering with us, you gain access to a reliable supply of high-quality intermediates backed by decades of chemical engineering excellence.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
