Scalable Synthesis of 3-Succinic Acid-30-Stearyl Glycyrrhetinate for Liver Targeting Drug Delivery
The pharmaceutical industry's relentless pursuit of targeted drug delivery systems has placed significant emphasis on the development of high-performance ligands capable of directing therapeutic agents to specific organs. Patent CN103833820A discloses a groundbreaking synthetic methodology for 3-succinic acid-30-stearyl glycyrrhetinate, a critical liver-targeting ligand that enhances the affinity of liposomes for hepatic parenchymal cells. This technical disclosure represents a paradigm shift from traditional, low-efficiency condensation reactions to a streamlined, high-yield nucleophilic substitution strategy. By fundamentally re-engineering the synthetic pathway, the inventors have addressed long-standing bottlenecks regarding yield, purity, and operational complexity that have historically plagued the manufacturing of this complex triterpenoid derivative. For R&D directors and procurement specialists alike, understanding the nuances of this patented process is essential for securing a reliable supply chain of high-purity pharmaceutical intermediates.
Furthermore, the transition from microwave-assisted batch processes to a robust thermal reaction system underscores the scalability of this approach. The elimination of hazardous or expensive condensing agents in the initial alkylation step not only aligns with green chemistry principles but also drastically simplifies the downstream processing requirements. As we delve deeper into the mechanistic and commercial implications of this technology, it becomes evident that this synthesis route offers a viable solution for the commercial scale-up of complex polymer additives and lipid-based delivery vectors. The ability to produce this key intermediate with minimal waste generation positions it as a cornerstone for next-generation hepatotropic drug formulations.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to the innovations detailed in the patent data, the synthesis of 3-succinic acid-30-stearyl glycyrrhetinate was fraught with inefficiencies that rendered large-scale production economically unviable. Traditional routes predominantly relied on dicyclohexylcarbodiimide (DCC) as a condensing agent to couple glycyrrhetinic acid with stearyl alcohol, often requiring cuprous chloride catalysis in N,N-dimethylformamide solvents. This approach suffered from severe drawbacks, including the generation of substantial amounts of dicyclohexylurea byproducts which were difficult to separate from the desired product. Consequently, extensive purification via column chromatography was mandatory, leading to significant material loss and solvent consumption. Reported total yields for these legacy methods hovered around a dismal 42%, with some variations dropping as low as 7.6%, making them entirely unsuitable for industrial manufacturing where cost efficiency is paramount.
The Novel Approach
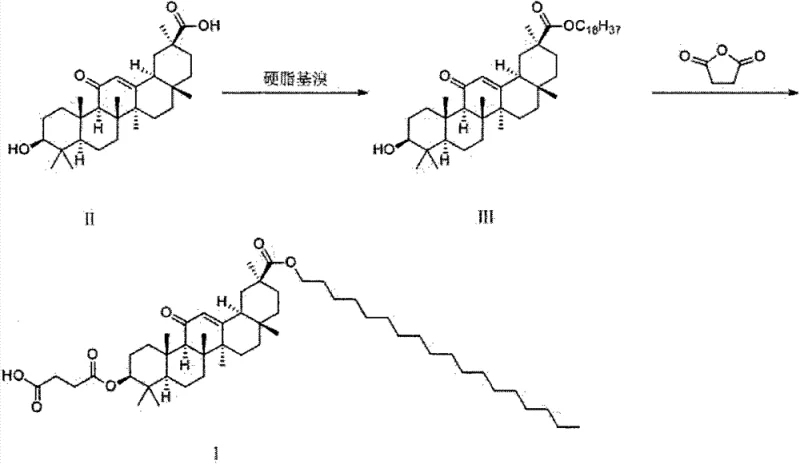
In stark contrast, the novel methodology introduced in the patent utilizes a direct nucleophilic substitution reaction between glycyrrhetinic acid and octadecyl bromide, bypassing the need for problematic condensing agents entirely. This strategic modification allows for the formation of the 30-stearyl glycyrrhetinate intermediate with exceptional efficiency, achieving isolated yields exceeding 96% in optimized examples. The subsequent esterification with succinic anhydride is conducted under mild conditions using 4-dimethylaminopyridine (DMAP) as a catalyst, further streamlining the workflow. Crucially, the workup procedure involves simple water precipitation and pH adjustment, completely obviating the need for column chromatography. This radical simplification of the process flow not only enhances the overall throughput but also ensures a higher degree of product consistency, which is vital for regulatory compliance in pharmaceutical applications.
Mechanistic Insights into Nucleophilic Substitution and Esterification
The core of this synthetic breakthrough lies in the exploitation of the carboxylic acid group at the C-30 position of the glycyrrhetinic acid skeleton. In the first step, the carboxylate anion, generated in situ by the action of an acid-binding agent such as sodium carbonate or potassium carbonate, acts as a potent nucleophile. It attacks the electrophilic carbon of the octadecyl bromide in an SN2-type displacement reaction. The choice of N,N-dimethylformamide (DMF) as the solvent is critical here, as its high polarity stabilizes the transition state and ensures the solubility of the bulky triterpenoid substrate. Maintaining the reaction temperature between 65°C and 80°C provides sufficient activation energy to drive the reaction to completion within a short timeframe of 1 to 4 hours, minimizing the risk of thermal degradation of the sensitive oleanane skeleton.
Following the isolation of the 30-stearyl intermediate, the second step involves the acylation of the hydroxyl group at the C-3 position. This is achieved through a condensation reaction with succinic anhydride, facilitated by the nucleophilic catalyst DMAP. The mechanism involves the formation of a highly reactive acylpyridinium intermediate, which is subsequently attacked by the C-3 hydroxyl group of the triterpenoid. The reaction is carefully controlled at temperatures between 45°C and 60°C to prevent side reactions such as anhydride hydrolysis or migration of the double bond. The final workup involves adjusting the pH to 4-5 using dilute acid after a basic wash, which protonates the succinate moiety and induces precipitation of the final product from the aqueous-organic mixture, effectively removing unreacted starting materials and catalyst residues.
How to Synthesize 3-Succinic Acid-30-Stearyl Glycyrrhetinate Efficiently
The implementation of this synthesis route requires precise control over stoichiometry and reaction parameters to maximize yield and purity. The process is divided into two distinct stages: the alkylation of the C-30 carboxyl group and the succinylation of the C-3 hydroxyl group. Each stage demands specific attention to molar ratios, particularly the slight excess of octadecyl bromide and succinic anhydride used to drive the equilibrium forward. The following guide outlines the standardized operational procedure derived from the patent examples, serving as a foundational reference for process chemists aiming to replicate this high-efficiency pathway in a pilot or production setting.
- Perform nucleophilic substitution of glycyrrhetinic acid with octadecyl bromide using an acid-binding agent in DMF at 65-80°C.
- Isolate the 30-stearyl glycyrrhetinate intermediate via water precipitation and filtration.
- React the intermediate with succinic anhydride and DMAP catalyst, followed by pH adjustment and precipitation to obtain the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthetic route translates directly into tangible operational benefits and risk mitigation. The most significant advantage is the drastic reduction in raw material costs associated with the elimination of expensive condensing agents like DCC and the removal of silica gel required for column chromatography. By simplifying the purification process to mere filtration and washing, the consumption of organic solvents is significantly lowered, which in turn reduces waste disposal costs and environmental compliance burdens. This streamlined workflow ensures a more predictable production schedule, as the bottlenecks associated with complex purification steps are entirely removed from the critical path.
- Cost Reduction in Manufacturing: The economic impact of switching to this alkyl bromide-based substitution method is profound. By avoiding the use of stoichiometric amounts of carbodiimides, which are costly and generate non-recyclable urea waste, the direct material cost per kilogram of product is substantially decreased. Furthermore, the high yield of over 89% compared to the historical average of 42% means that less starting material is required to produce the same amount of final API intermediate. This efficiency gain compounds across large batches, resulting in significant cost savings that can be passed down the supply chain or reinvested into R&D initiatives.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as octadecyl bromide, succinic anhydride, and common inorganic bases enhances the resilience of the supply chain. Unlike specialized catalysts or proprietary reagents that may face availability issues, these raw materials are widely sourced from multiple global suppliers. Additionally, the robustness of the reaction conditions, which tolerate moderate temperature fluctuations without compromising yield, reduces the risk of batch failures. This reliability ensures consistent delivery timelines for downstream pharmaceutical manufacturers who depend on a steady flow of high-quality liver-targeting ligands for their formulation pipelines.
- Scalability and Environmental Compliance: From an environmental and scaling perspective, this process is inherently superior. The absence of heavy metal catalysts like cuprous chloride eliminates the need for rigorous metal scavenging steps, which are often a regulatory hurdle in pharmaceutical production. The simplified workup involving water precipitation generates an aqueous waste stream that is easier to treat compared to the complex organic mixtures resulting from column chromatography. This alignment with green chemistry principles facilitates easier permitting for larger production volumes, allowing manufacturers to scale from laboratory grams to multi-ton annual capacities with minimal infrastructure modifications.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating the feasibility of integrating this intermediate into their supply chain, we have compiled answers to common inquiries regarding the process specifications and quality attributes. These responses are grounded in the experimental data provided in the patent documentation, offering a transparent view of the method's capabilities and limitations. Understanding these technical details is crucial for establishing clear quality agreements and ensuring that the material meets the stringent requirements of modern drug delivery applications.
Q: How does this synthesis method improve yield compared to traditional DCC coupling?
A: By replacing carbodiimide condensation with direct nucleophilic substitution using octadecyl bromide, the process avoids urea byproduct formation and complex purification, boosting total yield from roughly 42% to nearly 89%.
Q: Is column chromatography required for purification in this novel route?
A: No, the optimized protocol utilizes simple water precipitation and pH adjustment for isolation, completely eliminating the need for costly and time-consuming column chromatography steps.
Q: What are the critical reaction conditions for the succinylation step?
A: The succinylation requires mild temperatures between 45°C and 60°C with DMAP as a catalyst, ensuring selective esterification at the C-3 position without degrading the sensitive triterpenoid skeleton.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Succinic Acid-30-Stearyl Glycyrrhetinate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-purity intermediates play in the success of advanced drug delivery systems. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with precision and consistency. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to guarantee that every batch of 3-succinic acid-30-stearyl glycyrrhetinate meets the highest industry standards. Our facility is equipped to handle the specific solvent and temperature requirements of this nucleophilic substitution process, delivering a product that is ready for immediate formulation into liposomal carriers.
We invite you to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific application. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic advantages of sourcing this intermediate through our streamlined manufacturing channel. We encourage potential partners to contact us for specific COA data and route feasibility assessments, allowing us to demonstrate our capability to support your long-term supply goals with reliability and technical excellence.
