Advanced Manufacturing of 4,5-Disubstituted-2-Aminothiazole Intermediates via Mild Cyclization
Advanced Manufacturing of 4,5-Disubstituted-2-Aminothiazole Intermediates via Mild Cyclization
The pharmaceutical and agrochemical industries rely heavily on heterocyclic scaffolds, with the thiazole ring system standing out as a cornerstone for bioactive molecule design. Specifically, 4,5-disubstituted-2-aminothiazole compounds exhibit a broad spectrum of biological activities, ranging from antibacterial and antifungal properties to anticancer and antiviral applications. Recognizing the critical demand for efficient access to these valuable intermediates, recent advancements detailed in patent CN108341789B have introduced a transformative synthetic methodology. This innovation shifts the paradigm from traditional, resource-intensive protocols to a streamlined, one-pot cyclization strategy that leverages nitroepoxy compounds as key synthons. By utilizing ammonium thiocyanate and a mild inorganic base, this process achieves exceptional conversion rates under remarkably gentle thermal conditions, addressing long-standing challenges in impurity control and operational safety that have plagued previous generations of thiazole synthesis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the 2-aminothiazole core has been dominated by the classic Hantzsch condensation, a reaction that typically involves the coupling of α-halo ketones or aldehydes with thioamides or thiourea. While foundational, this legacy approach suffers from significant drawbacks that hinder modern manufacturing efficiency. The reliance on α-halo carbonyl precursors often introduces issues related to原料 availability and stability, while the reaction conditions frequently necessitate prolonged heating or the use of specialized catalysts to drive the cyclization to completion. Furthermore, alternative improvements reported in literature, such as microwave-assisted protocols or solid-supported catalysis, often introduce complexity in terms of equipment requirements or catalyst recovery. These conventional pathways can lead to inconsistent yields, difficult purification profiles due to side reactions, and elevated operational costs associated with energy consumption and waste management, creating a bottleneck for the reliable supply of high-purity pharmaceutical intermediates.
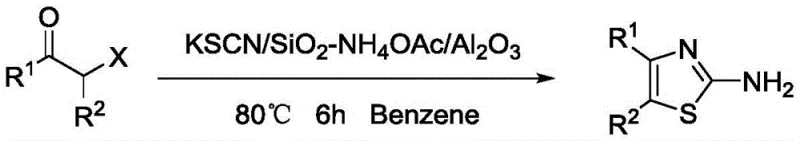
The Novel Approach
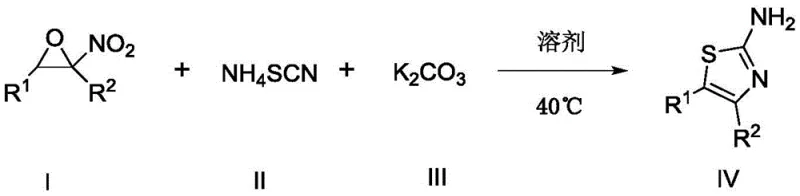
In stark contrast to these legacy constraints, the methodology disclosed in CN108341789B presents a robust and economically superior alternative. This novel route employs nitroepoxy compounds, which serve as versatile bifunctional electrophiles, reacting seamlessly with ammonium thiocyanate in the presence of a basic catalyst such as potassium carbonate. The reaction proceeds efficiently in a polar protic solvent like n-propanol at a modest temperature of 40±5°C, eliminating the need for high-energy inputs or hazardous reagents. This one-pot transformation not only simplifies the operational workflow by reducing unit operations but also dramatically enhances the overall atom economy. The mildness of the reaction environment preserves sensitive functional groups on the aromatic rings, allowing for a broader substrate scope including various halogenated and alkyl-substituted phenyl groups, thereby offering a versatile platform for the rapid generation of diverse thiazole libraries essential for drug discovery and process development.
Mechanistic Insights into Nitroepoxy-Mediated Cyclization
The success of this synthetic strategy lies in the unique reactivity profile of the nitroepoxy starting material. Structurally, the nitroepoxy compound possesses two adjacent electrophilic centers that are primed for nucleophilic attack, facilitating a cascade cyclization event. Upon exposure to the thiocyanate anion generated in situ, the epoxide ring undergoes regioselective opening, followed by an intramolecular cyclization that constructs the five-membered thiazole ring. The presence of the nitro group plays a crucial electronic role, activating the adjacent carbon centers and stabilizing the transition states involved in the ring closure. This mechanistic pathway is distinct from the SN2-type displacement seen in Hantzsch synthesis, offering a cleaner reaction profile with fewer byproducts. The use of potassium carbonate as a base is particularly strategic; it is strong enough to deprotonate the intermediate species and drive the elimination of water or other leaving groups necessary for aromatization, yet mild enough to prevent the degradation of the sensitive thiazole product or the hydrolysis of the starting epoxide under the reaction conditions.
From an impurity control perspective, this mechanism offers substantial advantages for GMP manufacturing. The low operating temperature of 40°C significantly suppresses thermal decomposition pathways that often lead to polymeric tars or complex degradation products in high-temperature reactions. Additionally, the stoichiometry is carefully optimized, typically employing a molar ratio of 1:3:2 for the nitroepoxy compound, ammonium thiocyanate, and base respectively, ensuring complete consumption of the limiting reagent while minimizing excess reagent carryover. The resulting crude reaction mixture is relatively clean, allowing for straightforward isolation via aqueous workup and extraction with ethyl acetate. Final purification using standard silica gel chromatography with a petroleum ether and ethyl acetate gradient yields the target 4,5-disubstituted-2-aminothiazole compounds with high purity, as evidenced by sharp melting points and consistent NMR spectral data across multiple examples, ensuring the material meets the stringent quality specifications required for downstream API synthesis.
How to Synthesize 4,5-Disubstituted-2-Aminothiazole Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires adherence to specific procedural parameters to maximize yield and reproducibility. The process begins with the precise weighing of the nitroepoxy precursor, ammonium thiocyanate, and potassium carbonate, which are then suspended in n-propanol. The reaction mixture is heated gently to maintain the temperature window of 40±5°C, with continuous stirring to ensure homogeneous mass transfer. Monitoring the reaction progress via TLC is critical, looking for the disappearance of the starting nitroepoxy spot, which typically occurs within 10 to 14 hours depending on the specific substituents. Once the reaction is deemed complete, the workup involves a simple aqueous quench followed by liquid-liquid extraction, avoiding complex filtration steps often associated with heterogeneous catalysis. The detailed standardized operating procedures for scaling this reaction from gram to kilogram quantities are outlined below to ensure consistent batch-to-batch quality.
- React nitroepoxy compounds with ammonium thiocyanate and potassium carbonate in n-propanol at 40±5°C for 10-14 hours.
- Quench the reaction with water, extract the organic layer with ethyl acetate, wash with saturated brine, dry, and concentrate.
- Purify the crude concentrate via silica gel column chromatography using a petroleum ether and ethyl acetate mixture to obtain the pure product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route translates directly into tangible operational efficiencies and risk mitigation. The shift away from specialized alpha-halo ketones to more commodity-grade nitroepoxy compounds and ammonium salts reduces dependency on niche suppliers, thereby enhancing supply chain resilience. The elimination of expensive transition metal catalysts or proprietary solid supports removes a significant cost driver from the bill of materials, while the mild reaction conditions lower energy consumption and reduce the load on HVAC and cooling systems in the production facility. Furthermore, the simplified workup procedure minimizes solvent usage and waste generation, aligning with increasingly strict environmental regulations and sustainability goals without compromising on output volume. These factors collectively contribute to a more stable and predictable cost structure for the manufacturing of complex pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the use of inexpensive, commercially available inorganic reagents like potassium carbonate and ammonium thiocyanate, which replace costly catalysts and specialized precursors. By operating at near-ambient temperatures, the process eliminates the need for high-energy heating or cryogenic cooling, leading to substantial utility savings. Additionally, the high conversion rates minimize the loss of valuable starting materials, ensuring that the theoretical yield is closely approached in practice, which effectively lowers the cost per kilogram of the final active intermediate.
- Enhanced Supply Chain Reliability: Sourcing reliability is significantly improved as the key raw materials are bulk chemicals with established global supply chains, reducing the risk of shortages that often plague specialized fine chemical reagents. The robustness of the reaction conditions means that the process is less sensitive to minor variations in raw material quality or environmental fluctuations, ensuring consistent production schedules. This stability allows for better inventory planning and reduces the need for safety stock, optimizing working capital and ensuring timely delivery to downstream API manufacturers.
- Scalability and Environmental Compliance: The one-pot nature of the reaction facilitates seamless scale-up from laboratory benchtop to multi-ton commercial production without the need for complex reactor modifications. The absence of heavy metals and the use of benign solvents simplify waste treatment protocols, reducing the environmental footprint and compliance costs associated with hazardous waste disposal. This green chemistry profile not only meets current regulatory standards but also future-proofs the manufacturing process against tightening environmental legislation, securing long-term operational continuity.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating this technology for their specific applications, we have compiled answers to common inquiries regarding the process parameters and product specifications. These insights are derived directly from the experimental data and optimization studies presented in the patent documentation, providing a clear picture of the method's capabilities and limitations. Understanding these details is crucial for integrating this synthesis into existing production lines or for designing new campaigns for thiazole-based drug candidates.
Q: What are the key advantages of this new synthesis method over the traditional Hantzsch method?
A: Unlike the traditional Hantzsch method which often requires harsh conditions and alpha-halo ketones, this novel approach utilizes readily available nitroepoxy compounds and operates at a mild temperature of 40°C, resulting in significantly higher yields and easier post-processing.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the process is highly scalable due to its one-pot nature, use of inexpensive inorganic bases like potassium carbonate, and avoidance of extreme temperatures or pressures, making it ideal for industrial manufacturing.
Q: What is the typical purity and yield achievable with this method?
A: The method consistently delivers high yields, often exceeding 80% and reaching up to 91% for specific derivatives like 4-methyl-5-phenyl-2-aminothiazole, with high purity achievable through standard silica gel chromatography.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4,5-Disubstituted-2-Aminothiazole Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent literature to commercial reality requires deep technical expertise and robust infrastructure. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising yields observed in the lab are faithfully reproduced on an industrial scale. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of 4,5-disubstituted-2-aminothiazole intermediate meets the exacting standards required for global pharmaceutical registration. We are committed to delivering not just a chemical product, but a reliable supply solution that supports your drug development timelines.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis can optimize your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain a clearer understanding of the potential economic impact of switching to this efficient route. We encourage you to contact us today to obtain specific COA data for our available thiazole derivatives and to schedule a consultation for route feasibility assessments tailored to your unique molecular targets.
