Scalable and Safe Manufacturing of Regadenoson Intermediates via Novel Cesium Catalysis
Introduction to Patent CN114249785A
The pharmaceutical industry is constantly seeking robust synthetic routes that balance high purity with stringent safety standards, particularly for potent cardiovascular agents like Regadenoson. Patent CN114249785A introduces a groundbreaking preparation method for 2-adenosine N-pyrazole derivatives, specifically targeting the synthesis of Regadenoson, a selective A2A adenosine receptor agonist used in radionuclide myocardial perfusion imaging. This technology represents a significant paradigm shift by completely eliminating the use of hydrazine hydrate, a known genotoxic reagent, from the synthetic pathway. Instead, it utilizes 2-chloroadenosine as a stable starting material, employing a cesium carbonate-catalyzed coupling strategy in an environmentally benign polyethylene glycol medium. For R&D directors and procurement specialists, this patent offers a compelling solution to the challenges of impurity control and regulatory compliance associated with mutagenic reagents.
The core innovation lies in the strategic redesign of the bond formation between the purine base and the pyrazole ring. Traditional methods often rely on harsh conditions or toxic precursors that complicate downstream purification and pose occupational health risks. By contrast, this novel approach operates under mild thermal conditions, typically between 10°C and 35°C for protection steps and around 100°C for the key coupling reaction. The process demonstrates exceptional control over stereochemistry and regioselectivity, ensuring that the final API intermediate meets the rigorous purity specifications required for injectable formulations. This technical advancement not only enhances the safety profile of the manufacturing process but also streamlines the supply chain by reducing the dependency on controlled substances.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Regadenoson has been plagued by the use of hazardous reagents and severe reaction conditions that compromise both operational safety and product quality. Prior art, such as the processes disclosed in earlier patents, frequently employs hydrazine hydrate to construct the pyrazole ring or to modify the purine scaffold. Hydrazine hydrate is classified as a Class 1 mutagen with confirmed carcinogenic potential, necessitating extremely strict control limits under ICH M7 guidelines. The presence of such genotoxic impurities requires extensive analytical validation, specialized equipment cleaning protocols, and often results in lower overall yields due to the aggressive nature of the chemistry involved. Furthermore, conventional routes often utilize strong bases or copper-containing catalysts at temperatures exceeding 150°C, which can lead to the degradation of the sensitive ribose sugar moiety and the formation of difficult-to-remove side products.
The Novel Approach
The methodology described in CN114249785A circumvents these critical bottlenecks by adopting a convergent synthesis strategy that prioritizes atom economy and operator safety. The process initiates with the protection of 2-chloroadenosine using p-toluenesulfonic acid, a stable and non-toxic reagent, to form a key intermediate that preserves the integrity of the nucleoside structure. 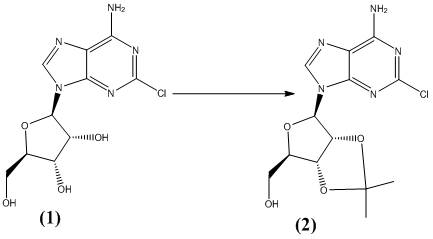 This protected species is then coupled with a pre-formed pyrazole amide building block, which is synthesized separately from ethyl 4-pyrazolecarboxylate and methylamine. The elimination of hydrazine from the entire workflow removes the burden of genotoxicity testing and allows for a more straightforward regulatory filing path. Additionally, the use of polyethylene glycol as a solvent replaces volatile organic compounds, aligning the process with green chemistry principles and reducing environmental disposal costs.
This protected species is then coupled with a pre-formed pyrazole amide building block, which is synthesized separately from ethyl 4-pyrazolecarboxylate and methylamine. The elimination of hydrazine from the entire workflow removes the burden of genotoxicity testing and allows for a more straightforward regulatory filing path. Additionally, the use of polyethylene glycol as a solvent replaces volatile organic compounds, aligning the process with green chemistry principles and reducing environmental disposal costs.
Mechanistic Insights into Cesium Carbonate-Catalyzed Coupling
The heart of this synthetic innovation is the nucleophilic aromatic substitution reaction facilitated by cesium carbonate in a polyethylene glycol matrix. In this step, the protected 2-chloroadenosine acts as the electrophile, while the pyrazole amide serves as the nucleophile. Cesium carbonate is selected specifically for its unique solubility profile and basicity; it is strong enough to deprotonate the pyrazole nitrogen to generate the reactive nucleophile but mild enough to prevent the hydrolysis of the ester or amide functionalities present in the molecule. The polyethylene glycol solvent plays a dual role: it solvates the cesium cation effectively, enhancing the nucleophilicity of the pyrazole anion through cation coordination, and it provides a high-boiling, non-volatile medium that supports the reaction at 100°C without the need for pressurized vessels. 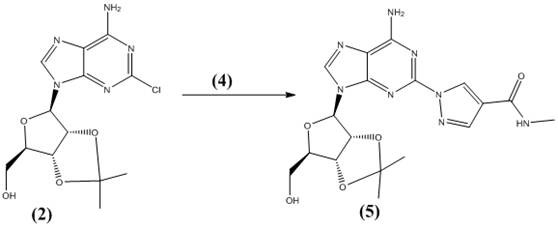 This synergistic effect drives the reaction to completion with high regioselectivity, favoring the formation of the N-1 linked product over potential N-2 isomers.
This synergistic effect drives the reaction to completion with high regioselectivity, favoring the formation of the N-1 linked product over potential N-2 isomers.
From an impurity control perspective, this mechanism offers distinct advantages over transition metal-catalyzed alternatives. The absence of copper or palladium catalysts eliminates the risk of heavy metal contamination, a common concern in pharmaceutical manufacturing that requires costly scavenging steps. The reaction proceeds cleanly to yield the coupled intermediate with a purity exceeding 98%, as evidenced by HPLC analysis. The subsequent deprotection step utilizes perchloric acid in an ethanol-water mixture, which efficiently removes the protecting groups without epimerizing the chiral centers on the ribose ring. 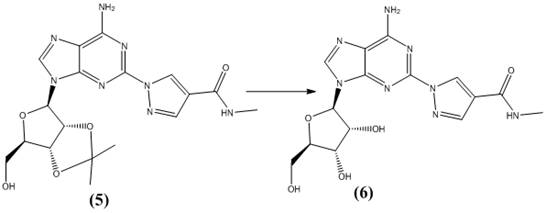 The final pH adjustment to neutrality ensures that the product precipitates in a highly crystalline form, facilitating easy filtration and drying. This mechanistic clarity provides R&D teams with a robust framework for troubleshooting and optimization, ensuring consistent batch-to-batch reproducibility.
The final pH adjustment to neutrality ensures that the product precipitates in a highly crystalline form, facilitating easy filtration and drying. This mechanistic clarity provides R&D teams with a robust framework for troubleshooting and optimization, ensuring consistent batch-to-batch reproducibility.
How to Synthesize Regadenoson Efficiently
The synthesis of Regadenoson via this patented route involves a logical sequence of protection, building block preparation, coupling, and deprotection. The process is designed to be scalable, utilizing readily available starting materials and avoiding exotic reagents. The initial protection of 2-chloroadenosine sets the stage for the subsequent coupling by masking the hydroxyl groups, preventing unwanted side reactions. The parallel synthesis of the pyrazole amide ensures that both fragments are ready for the convergent step, maximizing throughput. The detailed standardized synthesis steps, including specific molar ratios, temperature controls, and workup procedures, are outlined below to guide process chemists in replicating this high-efficiency route.
- Protect 2-chloroadenosine using p-toluenesulfonic acid in acetone to form the protected intermediate.
- React ethyl 4-pyrazolecarboxylate with aqueous methylamine to generate the pyrazole amide building block.
- Couple the protected adenosine and pyrazole amide using cesium carbonate in polyethylene glycol at elevated temperatures.
- Deprotect the coupled intermediate using perchloric acid followed by pH adjustment to isolate pure regadenoson.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route translates into tangible operational efficiencies and risk mitigation. The primary advantage is the complete removal of hydrazine hydrate from the supply chain. Sourcing and handling Class 1 mutagens require specialized storage facilities, strict inventory tracking, and extensive safety training, all of which add significant overhead to the cost of goods sold. By switching to a hydrazine-free process, manufacturers can drastically simplify their EHS (Environment, Health, and Safety) compliance protocols and reduce insurance liabilities. Furthermore, the use of 2-chloroadenosine as a starting material leverages a well-established supply chain for nucleoside derivatives, ensuring reliable availability and price stability compared to more exotic heterocyclic precursors.
- Cost Reduction in Manufacturing: The elimination of heavy metal catalysts and genotoxic reagents leads to substantial cost savings in downstream processing. Traditional routes often require expensive metal scavengers and multiple purification steps to meet residual solvent and metal limits. This new method produces a crude product of high purity, reducing the load on chromatography columns and crystallization steps. The use of polyethylene glycol, a low-cost and recyclable solvent, further decreases raw material expenses compared to high-purity anhydrous DMF or NMP. Additionally, the mild reaction conditions reduce energy consumption associated with heating and cooling cycles, contributing to a lower carbon footprint and reduced utility costs.
- Enhanced Supply Chain Reliability: The robustness of this chemistry enhances supply continuity by minimizing the risk of batch failures due to reagent variability. Hydrazine hydrate is subject to strict regulatory controls in many jurisdictions, which can lead to shipping delays and import restrictions. By removing this bottleneck, the supply chain becomes more resilient and agile. The starting materials, such as 2-chloroadenosine and ethyl 4-pyrazolecarboxylate, are commodity chemicals produced by multiple vendors globally, reducing the risk of single-source dependency. This diversification allows procurement teams to negotiate better terms and secure long-term contracts with confidence.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated to work effectively in standard glass-lined reactors without the need for specialized high-pressure equipment. The aqueous workup and simple filtration steps are easily adapted to large-scale production, facilitating a smooth transition from pilot plant to commercial manufacturing. From an environmental standpoint, the reduction in hazardous waste generation aligns with corporate sustainability goals. The avoidance of chlorinated solvents in the final steps and the use of water for precipitation minimize the volume of organic waste requiring incineration. This eco-friendly profile not only reduces disposal fees but also enhances the company's reputation as a responsible manufacturer in the global pharmaceutical market.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Regadenoson synthesis technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation. Understanding these nuances is crucial for technical teams evaluating the feasibility of technology transfer and for commercial teams assessing the value proposition of this manufacturing route.
Q: Why is the avoidance of hydrazine hydrate critical in Regadenoson synthesis?
A: Hydrazine hydrate is a Class 1 mutagenic agent with positive carcinogenic data, strictly regulated under ICH M7 guidelines. Eliminating it removes the need for complex residual control and genotoxicity testing, significantly simplifying quality assurance and regulatory filing.
Q: What are the advantages of using Polyethylene Glycol (PEG) as a solvent?
A: PEG serves as an environmentally friendly reaction medium that facilitates the solubility of polar intermediates and stabilizes the transition state during cesium carbonate catalysis. It allows for milder reaction conditions compared to traditional polar aprotic solvents like DMF, reducing waste treatment costs.
Q: How does the cesium carbonate catalysis improve the coupling efficiency?
A: Cesium carbonate acts as a mild yet effective base that promotes the nucleophilic aromatic substitution without degrading the sensitive ribose sugar moiety. This results in higher selectivity for the N-1 position of the pyrazole ring, minimizing regioisomer impurities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Regadenoson Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting safe and efficient synthetic routes for high-value cardiovascular intermediates like Regadenoson. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and reliability. Our state-of-the-art facilities are equipped to handle the specific requirements of nucleoside chemistry, including moisture-sensitive reactions and cryogenic conditions. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of Regadenoson intermediate meets the highest international standards for potency and impurity profiles.
We invite you to collaborate with us to leverage this advanced hydrazine-free technology for your next project. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. Please contact our technical procurement team today to request specific COA data, route feasibility assessments, and a comprehensive quotation. Let us help you optimize your supply chain and bring safer, high-quality medications to patients faster.
