Optimizing Setastine Hydrochloride Production for Commercial Scale-up and High-Purity API Manufacturing
The pharmaceutical industry continuously seeks robust synthetic routes that balance high purity with environmental safety and economic efficiency. Patent CN101486687B introduces a significant advancement in the preparation technology of Setastine Hydrochloride, a potent antianaphylaxis medicament. This innovation addresses critical bottlenecks found in traditional overseas production methods, specifically targeting the high toxicity of solvents and excessive reaction durations that plague legacy processes. By shifting from benzene to xylene and implementing a dual-catalyst system involving potassium iodide and polyethyleneglycol 400, this technology achieves a total yield of 50 percent to 60 percent, drastically outperforming the historical 30 percent benchmark. For R&D Directors and Procurement Managers, this represents a tangible opportunity to enhance the cost reduction in API manufacturing while ensuring the final product quality fully meets rigorous national and international standards. The strategic implementation of this patent data allows for a more sustainable supply chain, reducing the environmental footprint associated with volatile organic compounds and improving overall production throughput.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Setastine Hydrochloride has been hindered by reliance on hazardous solvents and inefficient reaction kinetics that compromise both safety and profitability. Conventional overseas production techniques typically utilize benzene as the primary solvent, a substance known for its severe toxicity and carcinogenic properties, posing significant health risks to workers and complicating waste disposal protocols. Furthermore, the condensation reaction in these legacy methods requires prolonged heating periods exceeding 20 hours, which not only consumes excessive energy but also increases the likelihood of side reactions and impurity formation. The low total yield of approximately 30 percent in these traditional routes results in substantial raw material wastage, driving up the cost of goods sold and limiting the scalability of the process for high-purity pharmaceutical intermediate demands. These inefficiencies create a fragile supply chain where lead times are extended, and the environmental compliance costs associated with benzene handling become a significant burden for manufacturing facilities aiming for green chemistry certification.
The Novel Approach
The innovative methodology outlined in the patent data offers a transformative solution by replacing benzene with xylene, a solvent with a significantly lower toxicity profile that simplifies environmental management and enhances worker safety. This novel approach integrates potassium iodide and polyethyleneglycol 400 as catalysts, which act synergistically to accelerate the condensation reaction, reducing the required reaction time from over 20 hours to a mere 6 hours. This drastic reduction in processing time allows for higher batch turnover rates, directly contributing to cost reduction in API manufacturing by maximizing equipment utilization and minimizing energy consumption. The optimized conditions facilitate a total yield improvement to the 50 percent to 60 percent range, effectively doubling the efficiency compared to conventional methods and ensuring a more reliable pharmaceutical intermediate supplier capability. By maintaining stringent control over reaction parameters such as temperature and catalyst loading, this route ensures that the product quality consistently meets high standards, making it an ideal candidate for commercial scale-up of complex pharmaceutical additives.
Mechanistic Insights into PEG400-KI Catalyzed Condensation
The core of this technological breakthrough lies in the sophisticated interplay between the phase transfer catalyst polyethyleneglycol 400 and potassium iodide within the xylene medium. The mechanism involves the activation of the nucleophile through the formation of a more reactive species facilitated by the polyether chain of PEG400, which solvates cations and enhances the nucleophilicity of the anionic species in the organic phase. Potassium iodide further assists by participating in halogen exchange or acting as a Lewis acid catalyst to lower the activation energy of the condensation step between the N-(2-chloroethyl)-azepane derivative and p-chlorodiphenyl ethanol. This catalytic system ensures that the reaction proceeds smoothly at 120-125°C, preventing the degradation of sensitive functional groups that might occur under harsher conditions required in non-catalyzed systems. The result is a cleaner reaction profile with fewer by-products, which simplifies downstream purification and ensures the high-purity Setastine Hydrochloride required for sensitive antianaphylaxis applications.
Impurity control is meticulously managed through the specific sequence of neutralization and extraction steps prior to the main condensation. The initial neutralization of N-(2-chloroethyl)-nitrogen-heterocycle heptane hydrochloride with sodium hydroxide generates the free base, which is then extracted into xylene, effectively removing inorganic salts and water-soluble impurities before they can interfere with the catalytic cycle. The use of anhydrous magnesium sulfate for drying the organic layer prior to the addition of catalysts prevents hydrolysis of the reactive intermediates, which is a common source of yield loss in similar etherification reactions. Furthermore, the decolorization step using activated carbon at temperatures below 20°C ensures that any colored impurities or polymeric by-products formed during the high-temperature reaction are removed before the final salt formation. This multi-stage purification strategy guarantees that the final product exhibits the white crystalline powder appearance and purity levels exceeding 99 percent, satisfying the rigorous demands of R&D Directors regarding impurity profiles.
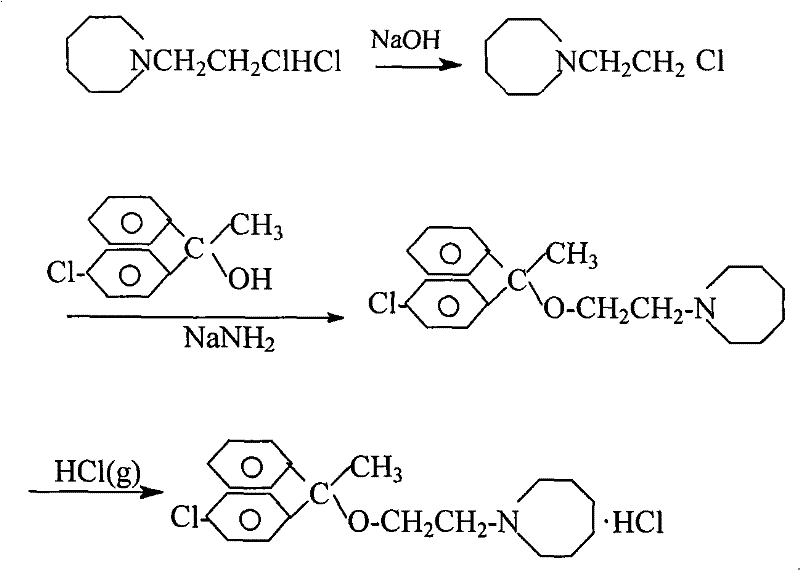
How to Synthesize Setastine Hydrochloride Efficiently
The synthesis of Setastine Hydrochloride via this optimized route requires precise adherence to the stoichiometric ratios and temperature profiles defined in the patent to achieve the reported 50-60 percent yield. The process begins with the careful neutralization of the amine salt followed by a controlled condensation in xylene, leveraging the catalytic effects of PEG400 and KI to drive the reaction to completion within 6 hours. Detailed standard operating procedures regarding the addition rates of sodium amide and the specific cooling curves for crystallization are critical for reproducibility on a manufacturing scale. For a comprehensive understanding of the exact operational parameters, the detailed standardized synthesis steps are provided in the guide below.
- Neutralize N-(2-chloroethyl)-azepane hydrochloride with NaOH and extract with Xylene to prepare the free base solution.
- React p-chlorodiphenyl ethanol with Sodium Amide in Xylene, then add the free base solution with KI and PEG400 catalysts at 120-125°C for 6 hours.
- Quench the reaction with water, separate layers, dry, decolorize, and perform salt formation with HCl gas followed by recrystallization in acetone.
Commercial Advantages for Procurement and Supply Chain Teams
For Procurement Managers and Supply Chain Heads, the adoption of this synthesis technology translates into substantial cost savings and enhanced operational resilience without compromising on quality. The shift away from benzene eliminates the need for expensive safety measures and specialized waste treatment protocols associated with carcinogenic solvents, thereby reducing the overall overhead costs of production. The significant reduction in reaction time from 20 hours to 6 hours allows for a drastic increase in production capacity using existing infrastructure, effectively reducing lead time for high-purity pharmaceutical intermediates and ensuring a more consistent supply flow to downstream formulation partners. This efficiency gain means that manufacturers can respond more agilely to market demand fluctuations, securing a competitive edge in the global supply of antianaphylaxis medications.
- Cost Reduction in Manufacturing: The elimination of toxic benzene and the reduction in energy consumption due to shorter reaction times lead to significantly reduced operational expenditures. By avoiding the need for extensive solvent recovery systems required for benzene, the capital expenditure for plant setup is also optimized, while the higher yield ensures that raw material costs are amortized over a larger output of saleable product. This qualitative improvement in process efficiency directly impacts the bottom line, making the final API more cost-competitive in the global market without the need for compromising on raw material quality.
- Enhanced Supply Chain Reliability: The use of readily available solvents like xylene and acetone, combined with robust catalysts, minimizes the risk of supply disruptions caused by the scarcity of specialized reagents. The simplified process flow reduces the number of unit operations, thereby decreasing the probability of mechanical failures or batch failures that could interrupt the supply chain. This reliability is crucial for maintaining continuous production schedules, ensuring that partners receive their orders on time and can maintain their own inventory levels without the need for excessive safety stock.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing standard chemical engineering principles that allow for seamless transition from pilot scale to commercial production of 100 MT or more. The reduced toxicity of the solvent system aligns with increasingly stringent environmental regulations, reducing the risk of regulatory fines and facilitating easier permitting for plant expansions. This environmental compliance not only protects the company's reputation but also future-proofs the manufacturing asset against evolving global standards for green chemistry and sustainable manufacturing practices.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route, based on the specific advantages and data points outlined in the patent documentation. Understanding these details is essential for stakeholders evaluating the feasibility of integrating this technology into their existing manufacturing portfolios. The answers provided reflect the technical realities of the process, ensuring transparency and alignment with the documented performance metrics.
Q: How does the new synthesis method improve upon conventional overseas production techniques for Setastine Hydrochloride?
A: The new method replaces toxic benzene with xylene and utilizes a PEG400/KI catalyst system, reducing reaction time from over 20 hours to 6 hours and increasing total yield from 30% to 50-60%.
Q: What are the critical quality control parameters for the raw materials in this synthesis route?
A: Critical parameters include p-chlorodiphenyl ethanol content >90%, Sodium Amide >98%, and N-(2-chloroethyl)-azepane hydrochloride >90% to ensure high-purity Setastine Hydrochloride output.
Q: Is this synthesis process suitable for large-scale commercial manufacturing?
A: Yes, the process uses standard solvents like xylene and acetone and operates at manageable temperatures (120-125°C), making it highly scalable for industrial production with reduced environmental toxicity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Setastine Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic routes like the one described in CN101486687B to maintain competitiveness in the fine chemical sector. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and efficient. We are committed to delivering products with stringent purity specifications and utilize our rigorous QC labs to verify that every batch of Setastine Hydrochloride meets the highest international standards for antianaphylaxis medicaments. Our infrastructure is designed to handle the specific requirements of this xylene-based process, guaranteeing consistency and reliability for our global partners.
We invite you to collaborate with us to leverage these technological advancements for your supply chain optimization. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how this specific synthesis route can benefit your specific production needs. We encourage you to contact our technical procurement team to索取 specific COA data and route feasibility assessments, allowing you to make informed decisions based on hard data and expert evaluation. Let us partner to drive efficiency and quality in your pharmaceutical manufacturing operations.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
