Advanced Solid-State Synthesis of N-methyl-N'-tetrahydrofuran Formyl Propane Diamine Oxalate for Commercial API Production
Advanced Solid-State Synthesis of N-methyl-N'-tetrahydrofuran Formyl Propane Diamine Oxalate for Commercial API Production
The pharmaceutical industry continuously seeks robust synthetic routes that ensure the highest purity standards for Active Pharmaceutical Ingredients (APIs), particularly for widely prescribed medications like Alfuzosin used in treating benign prostatic hyperplasia. A pivotal advancement in this domain is detailed in patent CN114591273A, which discloses a novel synthesis method for N-methyl-N'-tetrahydrofuran formyl propane diamine oxalate, a critical intermediate in the production of Alfuzosin and various isoquinoline medicaments. This technology addresses a longstanding bottleneck in fine chemical manufacturing where traditional methods yielded oily intermediates that were notoriously difficult to purify and stabilize. By converting this key amine intermediate into a stable oxalate salt, the process facilitates a dramatic improvement in purity, reaching levels exceeding 99 percent, while simultaneously simplifying the logistical challenges associated with transporting and storing reactive amine species. This report analyzes the technical merits and commercial implications of adopting this solid-state intermediate strategy for large-scale pharmaceutical production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the preparation of N-methyl-N'-tetrahydrofuranoylpropanediamine, often referred to as Compound 3 in technical literature, resulted in an oily substance that presented severe processing disadvantages for industrial scale-up. The primary defect of this oily state is its propensity to entrap significant quantities of organic solvents within its viscous matrix, making the complete removal of these volatiles extremely difficult even under rigorous vacuum conditions. Furthermore, because the material remains an oil at room temperature, it cannot be subjected to conventional crystallization techniques which are the gold standard for purification in the fine chemical industry. Manufacturers were forced to rely on distillation, a method that is inherently inefficient for removing impurities with boiling points similar to the target compound, leading to a final purity that typically hovers around only 90 percent. This lack of purity is critical because residual solvents and structural impurities, specifically identified as Compound 4 and Compound 5 in the patent data, can carry over into subsequent reaction steps. These carryover impurities participate in side reactions during the synthesis of the final drug, generating new contaminants that are exceptionally difficult to remove from the final API, thereby compromising patient safety and regulatory compliance.
The Novel Approach
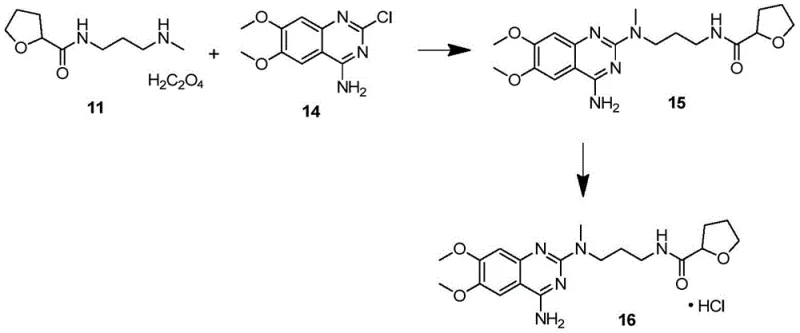
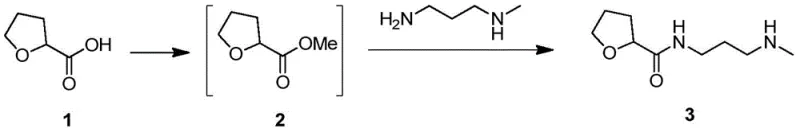
The innovative methodology described in the patent fundamentally alters the physical state of the intermediate by converting the free base amine into its corresponding oxalate salt, designated as Compound 11. This transformation is achieved through a streamlined three-step sequence beginning with the esterification of 2-tetrahydrofuran carboxylic acid, followed by amidation with N-methyl-1,3-propyl diamine, and concluding with a salt formation step using oxalic acid. The introduction of the oxalic acid moiety induces precipitation of the product as a crystalline solid from the reaction mixture, effectively separating it from the liquid mother liquor where the majority of impurities remain dissolved. This phase change from liquid to solid enables the use of filtration and washing techniques that are far more efficient than distillation, allowing for the removal of solvent residues and non-salt-forming impurities with high precision. The result is a high-purity solid intermediate with a GC purity of 99.43 percent, which serves as a superior starting material for downstream coupling reactions. This approach not only resolves the solvent entrapment issue but also enhances the thermal stability of the molecule, making it significantly more robust for storage and transportation compared to its unstable oily predecessor.
Mechanistic Insights into Oxalate Salt Formation and Purification
The core mechanism driving the success of this synthesis lies in the acid-base chemistry utilized to form the oxalate salt, which leverages differences in solubility and lattice energy to achieve purification. When oxalic acid or its hydrate is introduced to the solution containing the free base amine, a proton transfer occurs, generating a cationic ammonium species that pairs with the oxalate anion to form a stable ionic lattice. This lattice structure is highly ordered and energetically favorable, causing the product to precipitate out of the organic solvent system, typically ethyl acetate or isopropanol, while neutral or weakly basic impurities remain in the solution. Specifically, the patent highlights that impurities such as Compound 4 and Compound 5, which arise from side reactions during the amidation step, do not form stable salts under these conditions or possess solubility profiles that keep them in the mother liquor. Consequently, a simple filtration step effectively isolates the pure salt, breaking the cycle of impurity propagation that plagues the conventional oily route. This mechanistic advantage ensures that the intermediate fed into the subsequent palladium-catalyzed coupling or nucleophilic substitution reactions is of exceptional quality, minimizing the formation of complex byproduct mixtures that would otherwise require costly chromatographic purification.
Furthermore, the stability of the oxalate salt provides a kinetic barrier against degradation that is absent in the free base form. The ionic nature of the salt reduces the nucleophilicity of the nitrogen atoms, rendering the molecule less susceptible to oxidation or unwanted reactions with atmospheric moisture during storage. This chemical inertness is crucial for maintaining the integrity of the supply chain, as it eliminates the need for specialized cold-chain logistics or inert atmosphere packaging that might be required for the sensitive oily amine. In the context of the downstream synthesis of Alfuzosin, the high purity of the oxalate salt translates directly into cleaner reaction profiles during the critical C-N bond formation steps. Whether utilizing a palladium-catalyzed Buchwald-Hartwig coupling or a direct nucleophilic aromatic substitution, the absence of competing nucleophiles (from impurities) ensures that the catalyst turnover is directed solely toward the desired product, thereby maximizing atom economy and reducing the burden on downstream purification units.
How to Synthesize N-methyl-N'-tetrahydrofuran Formyl Propane Diamine Oxalate Efficiently
The synthesis protocol outlined in the patent offers a reproducible and scalable pathway for producing this high-value intermediate, beginning with the careful control of reaction temperatures and stoichiometry to maximize yield. The process initiates with an esterification step where 2-tetrahydrofuran carboxylic acid is reacted with methanol in the presence of concentrated sulfuric acid, followed by the addition of the diamine to form the amide backbone. The critical final step involves the addition of oxalic acid dihydrate to the crude amine solution, inducing precipitation of the target oxalate salt which is then isolated via centrifugation or filtration. Detailed operational parameters, including specific temperature ranges for esterification and salt formation, are essential to replicate the high purity results reported in the examples. For a comprehensive guide on the exact reagent quantities, reaction times, and workup procedures required to implement this technology in a GMP environment, please refer to the standardized synthesis instructions provided below.
- Perform esterification of 2-tetrahydrofuran carboxylic acid with methanol and concentrated sulfuric acid at low temperature, followed by heating.
- React the resulting ester with N-methyl-1,3-propyl diamine under heated conditions to form the free base amine.
- Treat the amine solution with oxalic acid or its hydrate in a solvent system to precipitate the pure oxalate salt.
Commercial Advantages for Procurement and Supply Chain Teams
Adopting this oxalate salt synthesis route offers profound economic and logistical benefits for procurement managers and supply chain directors overseeing the production of urological medications. The transition from an oily, difficult-to-handle intermediate to a free-flowing crystalline solid drastically simplifies material handling operations, reducing the risk of spillage, contamination, and measurement errors during batch compounding. From a cost perspective, the elimination of energy-intensive distillation steps in favor of simple filtration and drying represents a significant reduction in utility consumption and processing time. Moreover, the substantial increase in purity minimizes the loss of expensive downstream reagents, such as palladium catalysts and protected heterocycles, which would otherwise be wasted reacting with impurities present in lower-grade starting materials. This efficiency gain cascades through the entire production line, lowering the overall cost of goods sold for the final API while simultaneously enhancing the reliability of the manufacturing schedule by reducing the frequency of failed batches or reprocessing events.
- Cost Reduction in Manufacturing: The implementation of this solid-state purification strategy eliminates the need for complex fractional distillation equipment and the associated high energy costs required to separate close-boiling impurities from the oily free base. By relying on crystallization and filtration, manufacturers can utilize standard, lower-cost reactor setups that are easier to clean and maintain, leading to reduced capital expenditure and operational overhead. Additionally, the higher yield and purity of the intermediate mean that less raw material is required to produce a kilogram of the final drug, optimizing the utilization of expensive precursors like tetrahydrofuran carboxylic acid and specialized diamines. This material efficiency directly contributes to a leaner cost structure, allowing companies to maintain competitive pricing in the generic pharmaceutical market without sacrificing margin.
- Enhanced Supply Chain Reliability: The physical stability of the oxalate salt intermediate significantly mitigates risks associated with storage and transportation, as the solid form is not prone to the solvent retention issues that plague the oily counterpart. This stability ensures that the material retains its specified quality attributes over extended periods, reducing the likelihood of stock expiration or the need for frequent quality re-testing upon arrival at the manufacturing site. Suppliers can ship the material in standard packaging without the need for specialized containment solutions to prevent leakage or degradation, streamlining the logistics network and ensuring consistent availability for production planning. This reliability is critical for maintaining continuous API manufacturing schedules, preventing costly downtime caused by the rejection of sub-standard raw materials or the delay of shipments due to hazardous material classifications often associated with unstable liquids.
- Scalability and Environmental Compliance: Scaling up a crystallization-based purification process is inherently more straightforward and predictable than scaling a distillation process, particularly when dealing with thermally sensitive compounds that may degrade at high temperatures. The ability to produce high-purity material at ambient or mild temperatures reduces the generation of thermal degradation byproducts, simplifying waste stream management and lowering the environmental footprint of the manufacturing process. Furthermore, the efficient removal of solvents through filtration and washing reduces the volume of volatile organic compounds (VOCs) released into the atmosphere, aiding facilities in meeting stringent environmental regulations. This green chemistry advantage not only ensures compliance with global sustainability standards but also enhances the corporate reputation of the manufacturer as a responsible producer of high-quality pharmaceutical ingredients.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis method, derived directly from the experimental data and comparative examples provided in the patent documentation. Understanding these nuances is essential for R&D teams evaluating the feasibility of switching from conventional oily intermediates to the stabilized oxalate salt form. The answers reflect the specific improvements in purity, yield, and process robustness observed when utilizing the patented technology compared to historical methods.
Q: Why is the oxalate salt form preferred over the free base oil for Alfuzosin synthesis?
A: The free base exists as an oil that traps solvents and impurities, making purification difficult. The oxalate salt is a stable solid that can be purified via crystallization, achieving purities over 99% and removing critical impurities that affect downstream drug quality.
Q: What are the storage advantages of the oxalate intermediate?
A: Unlike the oily free base which requires special handling to prevent solvent retention and degradation, the oxalate salt exhibits high thermal stability and does not degrade easily at elevated temperatures, simplifying logistics and warehousing.
Q: How does this method impact the purity of the final Alfuzosin product?
A: By eliminating solvent residues and structural impurities (such as compounds 4 and 5) at the intermediate stage, the final coupling reaction proceeds with higher selectivity, resulting in Alfuzosin with significantly improved HPLC purity profiles.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-methyl-N'-tetrahydrofuran Formyl Propane Diamine Oxalate Supplier
As the global demand for high-quality urological treatments continues to rise, securing a dependable source of critical intermediates like N-methyl-N'-tetrahydrofuran formyl propane diamine oxalate is paramount for pharmaceutical success. NINGBO INNO PHARMCHEM stands at the forefront of this sector, leveraging extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production to deliver this advanced intermediate with unmatched consistency. Our state-of-the-art facilities are equipped to handle the precise temperature controls and filtration requirements necessary to reproduce the high-purity specifications outlined in patent CN114591273A, ensuring that every batch meets stringent purity specifications and rigorous QC labs standards. We understand that the transition to a new intermediate grade requires validation, and our technical team is prepared to support your process development efforts with samples and technical data packages that demonstrate the superiority of our solid-state material.
We invite procurement leaders and technical directors to engage with us for a Customized Cost-Saving Analysis that quantifies the potential efficiencies of integrating this oxalate salt into your existing Alfuzosin supply chain. By partnering with NINGBO INNO PHARMCHEM, you gain access to a supply partner committed to innovation and quality, capable of providing specific COA data and route feasibility assessments tailored to your production needs. Contact our technical procurement team today to discuss how we can optimize your raw material strategy and ensure a uninterrupted supply of this vital pharmaceutical building block.
