Advanced Enzymatic Resolution Technology for High-Purity Loxoprofen Intermediates and Commercial Scale-Up
The pharmaceutical industry continuously seeks robust methodologies for the production of chiral intermediates, particularly for non-steroidal anti-inflammatory drugs (NSAIDs) where enantiomeric purity dictates therapeutic efficacy and safety profiles. Patent CN108642119B introduces a groundbreaking stereoselective enzyme-catalyzed esterification method specifically designed for the resolution of 2-(4-methylphenyl)propionic acid enantiomers, a critical precursor in the synthesis of Loxoprofen Sodium. This technology leverages the exceptional catalytic efficiency and stereoselectivity of immobilized lipases within an optimized organic solvent system, addressing the longstanding challenges of low conversion rates and poor optical purity found in traditional aqueous systems. By shifting the reaction medium to organic solvents, the process significantly enhances the thermal stability of the biocatalyst, thereby driving substrate conversion to unprecedented levels while maintaining a green chemical profile. For R&D directors and procurement specialists, this patent represents a viable pathway to securing high-purity raw materials that meet stringent regulatory standards without incurring the prohibitive costs associated with classical resolution techniques. The ability to produce the pharmacologically active (S)-enantiomer with minimal side reactions positions this method as a cornerstone for modern, sustainable pharmaceutical manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the isolation of single enantiomers from racemic mixtures has relied heavily on techniques such as chiral chromatography, chemical resolution using chiral acids or bases, and membrane separation, each carrying distinct operational burdens that hinder large-scale commercial viability. Chiral chromatography, while capable of delivering high purity, is often restricted to laboratory-scale preparations due to the exorbitant cost of chiral stationary phases and the low throughput inherent in column-based separations. Chemical resolution methods frequently suffer from a theoretical maximum yield of 50% unless dynamic kinetic resolution is employed, which adds complexity and requires additional reagents that complicate downstream purification and waste management. Furthermore, traditional aqueous enzymatic processes often struggle with the poor solubility of hydrophobic substrates and the instability of enzymes in water, leading to inconsistent reaction rates and difficulties in product recovery. These conventional approaches collectively contribute to inflated manufacturing costs, extended lead times, and a larger environmental footprint, creating significant bottlenecks for supply chain managers aiming to optimize production efficiency. The reliance on these outdated methodologies often results in supply chain fragility, where minor fluctuations in reagent availability or equipment maintenance can disrupt the entire production schedule for critical API intermediates.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes immobilized Candida antarctica lipase B within a carefully selected organic solvent system to achieve superior stereoselectivity and operational simplicity. This method capitalizes on the unique ability of lipases to function effectively in non-aqueous media, where the enzyme's conformation is stabilized, and the solubility of hydrophobic substrates like 2-(4-methylphenyl)propionic acid is markedly improved. The organic medium not only facilitates higher substrate concentrations but also simplifies the downstream processing by allowing for easy separation of the product from the low-boiling solvent through distillation or evaporation. Unlike aqueous systems where enzyme recovery is challenging, the immobilized nature of the lipase in this organic setup allows for repeated reuse of the biocatalyst, drastically reducing the cost per batch and minimizing biological waste. This shift towards a non-aqueous enzymatic process represents a paradigm change in chiral synthesis, offering a scalable, cost-effective, and environmentally benign alternative that aligns perfectly with the principles of green chemistry. For procurement teams, this translates to a more reliable supply of high-quality intermediates with reduced dependency on complex purification infrastructure.
Mechanistic Insights into Lipase-Catalyzed Esterification Resolution
The core of this technological advancement lies in the precise mechanistic interaction between the immobilized lipase and the racemic substrate within the organic phase, where the enzyme exhibits a profound preference for one enantiomer over the other. The lipase active site is structured to accommodate the (R)-enantiomer of the acid for esterification, leaving the desired (S)-2-(4-methylphenyl)propionic acid unreacted in the solution, which can then be isolated with high optical purity. This kinetic resolution is driven by the specific spatial arrangement of the enzyme's catalytic triad, which selectively binds to the steric configuration of the (R)-isomer, facilitating the nucleophilic attack by the alcohol substrate to form the corresponding ester. The use of an organic solvent is critical here, as it modulates the enzyme's flexibility and hydration shell, preventing the denaturation that often occurs in water and enhancing the rigidity required for high stereoselectivity. By fine-tuning parameters such as solvent polarity, alcohol chain length, and temperature, the reaction can be pushed towards near-perfect enantiomeric excess, as evidenced by the patent data showing optical activity values reaching 97.84%. This level of control over the reaction mechanism ensures that impurity profiles are kept to an absolute minimum, satisfying the rigorous quality control standards demanded by global regulatory bodies for pharmaceutical ingredients.
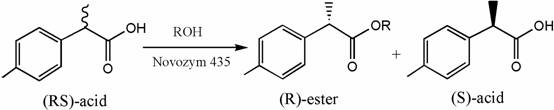
Furthermore, the impurity control mechanism is inherently built into the specificity of the biocatalyst, which avoids the formation of side products commonly associated with harsh chemical catalysts. In traditional chemical synthesis, strong acids or bases can lead to racemization, polymerization, or degradation of sensitive functional groups, necessitating extensive purification steps that lower overall yield. The enzymatic process operates under mild conditions, typically at moderate temperatures and neutral pH levels, preserving the integrity of the molecular structure throughout the transformation. The immobilization of the enzyme further prevents leaching of protein residues into the product stream, ensuring that the final API intermediate is free from biological contaminants. This inherent purity reduces the burden on quality assurance teams and minimizes the risk of batch rejection due to out-of-specification impurities. For R&D directors, understanding this mechanism provides confidence in the robustness of the process, allowing for smoother technology transfer from the laboratory to commercial manufacturing scales without the need for extensive re-optimization of purification protocols.
How to Synthesize 2-(4-methylphenyl)propionic Acid Efficiently
Implementing this synthesis route requires a systematic approach to reaction setup and parameter control to maximize the benefits of the enzymatic resolution process described in the patent. The process begins with the preparation of the reaction mixture, where racemic 2-(4-methylphenyl)propionic acid and a selected alcohol are dissolved in an organic solvent such as n-hexane to achieve optimal molar concentrations for kinetic resolution. Following the dissolution, a specific amount of immobilized Candida antarctica lipase B is introduced to the system, initiating the stereoselective esterification under controlled stirring and heating conditions. The reaction progress is monitored via high-performance liquid chromatography (HPLC) to determine the precise endpoint where substrate conversion and optical purity are balanced for maximum yield. Detailed standardized synthesis steps see the guide below for specific operational parameters and safety considerations.
- Prepare the reaction system by dissolving racemic 2-(4-methylphenyl)propionic acid and an alcohol substrate in an organic solvent such as n-hexane to achieve specific molar concentrations.
- Introduce immobilized Candida antarctica lipase B as the biocatalyst into the mixture, ensuring the enzyme concentration is optimized for maximum stereoselectivity and thermal stability.
- Maintain the reaction at a controlled temperature with stirring for a defined period, followed by HPLC analysis to verify substrate conversion and optical purity exceeding 97%.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this enzymatic resolution technology offers substantial advantages that directly address the core concerns of procurement managers and supply chain heads regarding cost, reliability, and scalability. The elimination of expensive chiral chromatography columns and the reduction in solvent usage due to the efficiency of the organic system lead to a significant reduction in overall manufacturing costs, making the final intermediate more price-competitive in the global market. Additionally, the ability to recover and reuse the immobilized enzyme multiple times without significant loss of activity further drives down the variable costs associated with raw materials, providing a sustainable economic model for long-term production. For supply chain planners, the simplicity of the operation and the robustness of the enzyme in organic solvents mean that production schedules are less prone to disruptions caused by equipment failure or complex process deviations. This reliability ensures a consistent flow of high-purity intermediates, reducing the need for safety stock and minimizing the risk of production stoppages in downstream API synthesis. The green nature of the process also aligns with increasingly strict environmental regulations, mitigating the risk of compliance-related delays and enhancing the corporate sustainability profile of the manufacturing entity.
- Cost Reduction in Manufacturing: The transition to this enzymatic method eliminates the need for costly chiral resolving agents and reduces solvent consumption through efficient recovery processes, resulting in substantial cost savings per kilogram of product. By avoiding the theoretical 50% yield limit of traditional resolution through optimized kinetic control, the process maximizes raw material utilization, effectively lowering the cost of goods sold. The reuse of the immobilized biocatalyst over multiple batches further amortizes the initial catalyst cost, contributing to a leaner and more efficient production budget. These economic benefits allow for more competitive pricing strategies without compromising on the quality or purity of the final pharmaceutical intermediate.
- Enhanced Supply Chain Reliability: The robustness of the immobilized lipase in organic solvents ensures consistent reaction performance across different batches, minimizing the variability that often plagues biological processes. This consistency translates to predictable lead times and reliable delivery schedules, which are critical for maintaining uninterrupted API production lines for major pharmaceutical clients. The simplified downstream processing, facilitated by the volatility of the organic solvents, reduces the time required for product isolation and drying, accelerating the overall turnaround time from raw material to finished intermediate. Supply chain heads can therefore plan inventory levels with greater confidence, knowing that the production process is stable and less susceptible to external fluctuations in reagent quality or environmental conditions.
- Scalability and Environmental Compliance: The process is designed for easy scale-up from laboratory to industrial reactors, as the mass transfer and heat exchange requirements are manageable within standard chemical engineering frameworks. The use of green solvents and the absence of heavy metal catalysts ensure that waste streams are easier to treat and dispose of, complying with stringent environmental protection laws. This environmental compliance reduces the administrative burden and potential fines associated with hazardous waste management, making the facility more attractive for long-term investment. The scalability ensures that as demand for Loxoprofen Sodium grows, the production capacity can be expanded seamlessly without the need for fundamentally new technology or infrastructure.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this enzymatic resolution technology, based on the specific data and claims found within the patent documentation. These answers are designed to provide clarity on the operational feasibility and quality outcomes expected from adopting this method for the production of chiral pharmaceutical intermediates. Understanding these details is crucial for stakeholders evaluating the potential integration of this technology into their existing manufacturing portfolios. The responses reflect the objective technical advantages verified through the experimental examples provided in the patent.
Q: Why is enzymatic resolution preferred over chromatography for this intermediate?
A: Enzymatic resolution eliminates the need for expensive chiral separation media and complex equipment associated with chromatography, significantly reducing production costs while maintaining high optical purity suitable for industrial scale-up.
Q: How does the organic solvent system improve the process efficiency?
A: The use of organic solvents like n-hexane enhances the thermal stability and catalytic efficiency of the lipase, prevents enzyme dissolution for easier recovery, and facilitates product separation due to lower boiling points.
Q: What is the achievable optical purity using this method?
A: According to the patent data, this method can achieve an optical activity of the substrate greater than or equal to 97.84%, ensuring the high quality required for downstream pharmaceutical synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-(4-methylphenyl)propionic acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-purity chiral intermediates in the development of next-generation anti-inflammatory therapies, and we are fully equipped to support your supply needs with this advanced enzymatic technology. Our CDMO expertise allows us to scale diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can grow from clinical trials to full market launch without supply bottlenecks. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 2-(4-methylphenyl)propionic acid meets the highest international standards for optical activity and chemical purity. Our team of experienced chemists is dedicated to optimizing this lipase-catalyzed process to maximize yield and minimize costs, providing you with a competitive edge in the pharmaceutical market. By partnering with us, you gain access to a reliable supply chain that prioritizes quality, consistency, and regulatory compliance above all else.
We invite you to engage with our technical procurement team to discuss how this innovative resolution method can be tailored to your specific project requirements and volume needs. Request a Customized Cost-Saving Analysis to understand the potential economic benefits of switching to this enzymatic route for your Loxoprofen intermediate supply. Our team is ready to provide specific COA data and route feasibility assessments to demonstrate our capability to deliver high-quality materials on time and within budget. Let us help you optimize your supply chain and secure a stable source of critical chiral building blocks for your pharmaceutical formulations.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
