Advanced Synthesis of Piperazinyl Ionic Liquid Surfactants for Commercial Scale-Up
Introduction to Next-Generation Ionic Liquid Surfactants
The chemical industry is currently witnessing a significant paradigm shift towards more sustainable and efficient surfactant technologies, driven by the urgent need for greener processing methods and higher performance materials. Patent CN112707875A introduces a groundbreaking class of piperazinyl ionic liquid surfactants that address the longstanding limitations of conventional ionic liquid synthesis. Historically, the preparation of ionic liquid surfactants has been plagued by complex multi-step procedures, often involving difficult purification stages and the use of expensive transition metal catalysts or harsh quaternization agents. This new technology disrupts that status quo by leveraging a straightforward acid-base neutralization strategy between piperazine derivatives and long-chain carboxylic acids. The result is a robust, scalable, and highly pure product family that serves as a critical enabler for advanced applications in pharmaceutical formulations and specialty chemical systems. For R&D directors and procurement specialists alike, this represents a tangible opportunity to streamline supply chains while accessing superior material performance characteristics that were previously cost-prohibitive.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional methodologies for synthesizing ionic liquid surfactants, particularly those based on imidazolium or pyridinium cores, often suffer from inherent inefficiencies that hinder large-scale industrial adoption. As highlighted in the background art of the patent, existing routes frequently necessitate complex grafting of long-chain alkyl groups onto cationic heads through tedious alkylation reactions that require precise temperature control and extended reaction times. Furthermore, anionic variants typically demand ion exchange processes to swap simple halide anions for functional long-chain carboxylates or sulfonates, a step that generates substantial salt waste and complicates downstream purification. These legacy processes not only inflate the cost of goods sold due to high energy consumption and reagent usage but also introduce variability in product quality, often requiring multiple recrystallization steps to achieve acceptable purity levels. Consequently, the commercial viability of many promising ionic liquid surfactants has been severely restricted, limiting their deployment in cost-sensitive sectors such as agrochemicals and personal care.
The Novel Approach
In stark contrast to these cumbersome legacy protocols, the novel approach disclosed in CN112707875A utilizes a remarkably elegant one-step synthesis that proceeds under mild conditions. By directly reacting readily available piperazine derivatives with long-chain carboxylic acids in common organic solvents, the process eliminates the need for complex catalysts or hazardous alkylating agents. The reaction occurs efficiently at room temperature, typically completing within a window of 3 to 24 hours, which drastically reduces the energy footprint of the manufacturing process. Perhaps most significantly for commercial production, the resulting products precipitate or can be isolated simply by solvent removal and washing, achieving high purity levels of over 97% without the need for resource-intensive recrystallization. This streamlined workflow not only accelerates the time-to-market for new formulations but also fundamentally alters the cost structure, making high-performance ionic liquid surfactants accessible for mass-market applications in microemulsions and gel systems.
Mechanistic Insights into Piperazine-Based Acid-Base Neutralization
The core chemical transformation driving this innovation is a classic yet highly effective Brønsted-Lowry acid-base neutralization occurring within the piperazine heterocyclic framework. The piperazine ring, possessing two nitrogen atoms, offers unique structural versatility; in this specific synthesis, one nitrogen is substituted with a variable group (R1), while the secondary amine nitrogen acts as the basic center. When a long-chain carboxylic acid (R2COOH, where R2 is a C9-C17 alkyl chain) is introduced, a proton transfer occurs from the carboxylic acid to the secondary amine nitrogen of the piperazine ring. This protonation generates a positively charged ammonium cation on the ring, which is electrostatically stabilized by the corresponding long-chain carboxylate anion. This ionic pairing creates the amphiphilic structure essential for surfactant activity, where the hydrophilic ionic head group interacts with polar phases and the hydrophobic alkyl tail aligns with non-polar phases. The simplicity of this mechanism ensures high atom economy and minimizes the formation of side products, which is critical for maintaining the stringent purity profiles required in pharmaceutical intermediates.
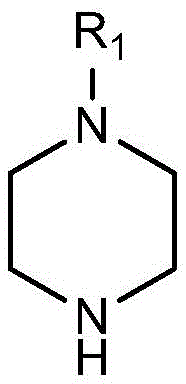
Beyond the primary reaction mechanism, the structural tunability of this system offers profound advantages for impurity control and property optimization. The R1 group on the piperazine ring can be varied extensively—ranging from simple alkyl groups like methyl and ethyl to functional groups like hydroxyethyl or phenyl—allowing chemists to fine-tune the hydrophilicity and steric bulk of the surfactant head. This modularity enables the precise engineering of critical micelle concentrations (CMC) and interfacial tension properties without altering the fundamental synthetic route. Furthermore, because the reaction relies on stoichiometric acid-base chemistry rather than kinetic-controlled catalysis, the risk of generating complex byproduct mixtures is inherently low. The patent data confirms that elemental analysis and NMR spectroscopy of the products match theoretical values closely, indicating that the ionic lattice forms cleanly. This mechanistic clarity provides R&D teams with a predictable platform for developing custom surfactants tailored to specific solubilization challenges, such as the encapsulation of poorly water-soluble drugs in microemulsion systems.
How to Synthesize Piperazinyl Ionic Liquid Surfactant Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for laboratory and pilot-scale production, emphasizing operational simplicity and safety. The process begins with the dissolution of the chosen piperazine derivative in a volatile organic solvent such as dichloromethane, methanol, or ethyl acetate, ensuring a homogeneous reaction medium. Subsequently, the long-chain carboxylic acid is added gradually to manage the exothermic nature of the neutralization, often with the aid of an ice-water bath to maintain thermal stability. Following the addition, the mixture is stirred at ambient temperature for a defined period, allowing the ionic salt to form completely before the solvent is removed under reduced pressure.
- Dissolve the specific piperazine derivative (where R1 is methyl, ethyl, hydroxyethyl, etc.) in a suitable organic solvent such as dichloromethane, methanol, or chloroform within a reaction vessel.
- Gradually add the long-chain carboxylic acid (C9 to C17 chain length) to the piperazine solution while maintaining the reaction mixture in an ice-water bath to control exothermic heat.
- Stir the reaction mixture at room temperature for a period ranging from 3 to 24 hours, then remove the solvent via vacuum distillation, wash the resulting solid, and dry to obtain the final surfactant product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this piperazinyl ionic liquid technology translates into immediate and tangible strategic benefits regarding cost efficiency and supply reliability. The elimination of complex catalytic systems and multi-step purification sequences means that the raw material bill of materials is significantly reduced, relying instead on commodity chemicals like fatty acids and simple piperazines which are abundantly available in the global market. This simplification of the supply chain reduces exposure to price volatility associated with specialized reagents and minimizes the logistical complexity of sourcing hard-to-find catalysts. Moreover, the ability to achieve high purity without recrystallization implies a drastic reduction in solvent usage and waste generation, which directly lowers the environmental compliance costs and waste disposal fees associated with manufacturing. These factors combine to create a highly competitive cost structure that allows for aggressive pricing strategies while maintaining healthy margins.
- Cost Reduction in Manufacturing: The economic impact of this synthesis route is driven primarily by the reduction in unit operations and energy consumption. By conducting the reaction at room temperature, the need for expensive heating or cooling infrastructure is minimized, leading to substantial savings in utility costs over the lifecycle of the product. Additionally, the one-step nature of the process reduces labor hours and equipment occupancy time, effectively increasing the throughput capacity of existing manufacturing facilities without the need for capital expenditure on new reactors. The avoidance of transition metal catalysts also removes the costly and time-consuming step of heavy metal scavenging, which is often a regulatory requirement for pharmaceutical intermediates. Collectively, these efficiencies result in a significantly lower cost of goods sold, enabling more competitive positioning in the global marketplace for specialty surfactants.
- Enhanced Supply Chain Reliability: From a supply chain resilience perspective, the reliance on widely available feedstocks such as long-chain fatty acids and piperazine derivatives mitigates the risk of supply disruptions. Unlike proprietary catalysts or exotic reagents that may have single-source suppliers, the key inputs for this process are produced by multiple vendors globally, ensuring a robust and redundant supply network. The short reaction time and simple workup procedure further enhance agility, allowing manufacturers to respond quickly to fluctuations in demand without maintaining excessive inventory buffers. This responsiveness is crucial for just-in-time manufacturing models prevalent in the pharmaceutical and personal care industries, where delays in intermediate supply can halt downstream production lines. Consequently, partnering with a supplier utilizing this technology ensures a steady and predictable flow of materials.
- Scalability and Environmental Compliance: The inherent safety and simplicity of the acid-base neutralization mechanism make this process exceptionally well-suited for commercial scale-up. The absence of hazardous reagents and the mild operating conditions reduce the safety risks associated with large-scale batch processing, facilitating easier regulatory approval for new manufacturing sites. Furthermore, the reduced solvent load and lack of heavy metal waste align perfectly with modern green chemistry principles and increasingly stringent environmental regulations. This compliance advantage not only future-proofs the supply chain against tightening regulatory frameworks but also enhances the brand value of the end products by supporting sustainability claims. The ability to scale from kilogram to multi-ton quantities with consistent quality makes this technology a viable long-term solution for industrial applications.
Frequently Asked Questions (FAQ)
To assist technical decision-makers in evaluating the feasibility of integrating these materials into their existing workflows, we have compiled answers to common inquiries based on the specific technical disclosures within the patent literature. These questions address critical aspects regarding the stability, application scope, and physical properties of the piperazinyl ionic liquid surfactants. Understanding these nuances is essential for formulators who need to ensure compatibility with their active ingredients and processing equipment. The following insights are derived directly from the experimental data and beneficial effects described in the intellectual property documentation.
Q: What is the primary advantage of this piperazinyl ionic liquid synthesis over traditional methods?
A: The primary advantage is the drastic simplification of the process. Unlike traditional ionic liquid surfactants that require complex quaternization or multi-step ion exchange procedures, this method utilizes a direct one-step acid-base neutralization at room temperature, eliminating the need for harsh conditions or expensive catalysts.
Q: Can this surfactant be used for drug delivery systems?
A: Yes, the patent explicitly demonstrates the application of these surfactants in forming microemulsions and microemulsion gels. They have been successfully tested for encapsulating active ingredients like curcumin and berberine hydrochloride, showing high drug loading capacities and thermal reversibility.
Q: What represents the purity level achievable with this synthesis route?
A: The synthesis route is capable of achieving high purity levels, specifically noted as reaching more than 97% without the need for complex recrystallization steps. Experimental examples in the patent data show purities exceeding 98% and even 99% for specific derivatives like 1-(2-hydroxyethyl) piperazine dodecanoate.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Piperazinyl Ionic Liquid Surfactant Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this piperazinyl ionic liquid technology and are fully equipped to support its transition from laboratory curiosity to commercial reality. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with unwavering consistency. Our state-of-the-art facilities are designed to handle the specific solvent systems and isolation techniques required for these surfactants, while our rigorous QC labs enforce stringent purity specifications to guarantee that every batch meets the highest industry standards. We understand that in the fast-paced world of fine chemicals, reliability is just as important as innovation, and our dedicated team is committed to delivering both.
We invite you to explore how this advanced synthesis route can optimize your formulation costs and enhance your product performance. Our technical sales team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and application targets. We encourage you to contact our technical procurement team today to request specific COA data for our pilot batches and to discuss detailed route feasibility assessments for your projects. Let us collaborate to bring these high-performance surfactant solutions to your market faster and more efficiently than ever before.
