Scalable Copper-Catalyzed Synthesis of Sulfonamide-Substituted Pyrrolidones for Commercial API Manufacturing
Introduction to Advanced Pyrrolidone Synthesis Technology
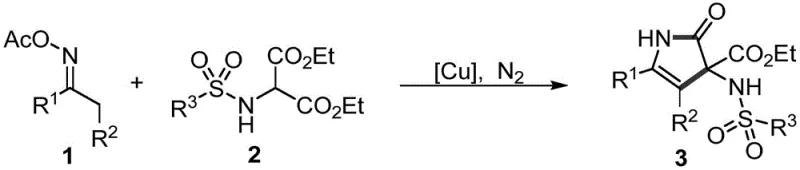
The pharmaceutical industry continuously seeks robust and scalable synthetic routes for bioactive heterocyclic scaffolds, particularly pyrrolidone derivatives which serve as critical cores in antimalarial, antiviral, and anticancer agents. A significant breakthrough in this domain is detailed in patent CN111087338B, which discloses a novel preparation method for sulfonamide-substituted pyrrolidones derivatives. This technology represents a paradigm shift from traditional multi-step syntheses involving harsh reagents to a streamlined, copper-catalyzed cyclization process. By leveraging oxime esters and sulfonamide malonates as primary building blocks, this method achieves high efficiency through an elegant internal oxidation mechanism. For R&D directors and procurement specialists, understanding this innovation is crucial as it promises to enhance the supply chain reliability of high-purity pharmaceutical intermediates while drastically simplifying the manufacturing workflow.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
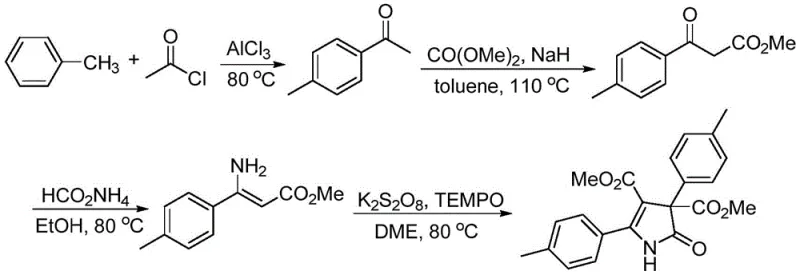
Historically, the construction of the pyrrolidone ring has been fraught with synthetic challenges that impede large-scale commercialization. Traditional pathways often rely on aggressive Lewis acids such as anhydrous aluminum chloride or require stoichiometric amounts of strong oxidants like potassium persulfate, which complicate post-reaction workup and generate substantial hazardous waste. Furthermore, existing literature describes methods utilizing precious metal catalysts like platinum or rhodium, which are not only prohibitively expensive but also pose significant supply chain risks due to their scarcity. Some protocols necessitate cryogenic conditions or complex nano-catalyst preparations, rendering them impractical for ton-scale production. These legacy processes frequently suffer from low atom economy, difficult purification steps, and safety concerns associated with handling powerful oxidizing agents, creating a bottleneck for the cost-effective manufacturing of complex API intermediates.
The Novel Approach
In stark contrast, the methodology outlined in the patent introduces a highly efficient copper-catalyzed system that circumvents these historical hurdles. The core innovation lies in the utilization of oxime esters, which function dually as substrates and internal oxidants, thereby eliminating the need for external oxidizing agents. This approach employs readily available and inexpensive copper salts, such as copper(I) chloride, operating under mild thermal conditions typically around 80 °C in common solvents like toluene. The reaction demonstrates exceptional functional group tolerance, accommodating a wide array of electronic and steric variations on both the ketoxime and sulfonamide components. This simplicity translates directly into operational excellence, allowing for shorter synthetic routes, easier product isolation, and a significantly reduced environmental footprint compared to prior art methods.
Mechanistic Insights into Copper-Catalyzed Cyclization
The mechanistic elegance of this transformation is rooted in the unique reactivity of the oxime ester functionality under copper catalysis. The copper species likely coordinates with the oxime nitrogen, facilitating the homolytic or heterolytic cleavage of the weak N-O bond. This cleavage generates a reactive nitrogen-centered radical or imino species in situ, which subsequently undergoes intramolecular cyclization with the activated methylene group of the sulfonamide malonate. Crucially, the oxygen atom from the N-O bond acts as the terminal oxidant, driving the aromatization or saturation of the ring system without requiring external additives. This internal redox neutral or oxidative process ensures that the reaction proceeds with high atom economy. For process chemists, this mechanism implies a cleaner reaction profile with fewer side products derived from oxidant decomposition, thereby simplifying the impurity profile and easing the burden on downstream purification units.
Furthermore, the choice of copper catalyst provides a tunable platform for optimizing reaction kinetics and selectivity. The patent data indicates that various copper salts, including CuCl, CuBr, and Cu(OAc)2, are effective, suggesting a robust catalytic cycle that is not overly sensitive to the specific counterion. The reaction tolerates diverse substituents on the aromatic rings, including electron-withdrawing groups like nitro and halogens, as well as electron-donating groups like methoxy and methyl. This broad substrate scope is indicative of a mechanism that can accommodate significant steric and electronic variation without compromising yield. Such versatility is paramount for medicinal chemistry campaigns where rapid analog synthesis is required to establish structure-activity relationships, ensuring that this method serves as a reliable tool for generating diverse libraries of bioactive pyrrolidone scaffolds.
How to Synthesize Sulfonamide-Substituted Pyrrolidones Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires adherence to specific operational parameters to maximize yield and purity. The general protocol involves dissolving the oxime ester and sulfonamide malonate precursors in an anhydrous organic solvent, followed by the addition of the copper catalyst under an inert atmosphere. The mixture is then heated to the optimal temperature range, monitored via TLC or HPLC, and worked up using standard extraction techniques. While the patent provides specific molar ratios and solvent choices, scaling this reaction requires careful attention to heat transfer and mixing efficiency to maintain the delicate balance of the catalytic cycle. The following guide outlines the standardized steps derived from the patent examples to ensure reproducible results for high-purity pharmaceutical intermediates.
- Combine oxime ester and sulfonamide malonate substrates with a copper salt catalyst (e.g., CuCl) in an organic solvent like toluene.
- Heat the reaction mixture to 60-140 °C under a nitrogen atmosphere to facilitate the cyclization utilizing the internal N-O oxidant.
- Upon completion, extract the product with ethyl acetate and water, dry the organic layer, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, this copper-catalyzed methodology offers compelling advantages that directly impact the bottom line and supply chain resilience. The substitution of precious metal catalysts with abundant copper salts results in a drastic reduction in raw material costs, removing the volatility associated with rhodium or platinum pricing. Moreover, the elimination of hazardous external oxidants simplifies regulatory compliance and waste disposal procedures, leading to substantial cost savings in environmental health and safety management. The mild reaction conditions and short synthetic route enhance the overall throughput of the manufacturing facility, allowing for faster turnaround times and improved responsiveness to market demand fluctuations. These factors collectively position this technology as a superior choice for the commercial scale-up of complex pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The transition from expensive noble metal catalysts to inexpensive copper salts fundamentally alters the cost structure of the synthesis. By avoiding the use of ligands and specialized nano-catalysts that require complex preparation, the direct material costs are significantly lowered. Additionally, the internal oxidant strategy removes the expense of purchasing and handling strong chemical oxidants, further contributing to economic efficiency. This cost optimization makes the final API intermediate more competitive in the global market, providing a distinct advantage for procurement managers negotiating long-term supply contracts.
- Enhanced Supply Chain Reliability: Reliance on scarce precious metals introduces significant supply chain vulnerability, whereas copper is globally abundant and stable in price. The use of common solvents like toluene and simple starting materials ensures that raw material availability is not a bottleneck. This stability guarantees consistent production schedules and reduces the risk of delays caused by raw material shortages. For supply chain heads, this means a more predictable and secure source of critical intermediates, essential for maintaining uninterrupted API production lines and meeting strict delivery commitments to downstream pharmaceutical partners.
- Scalability and Environmental Compliance: The operational simplicity of this method, characterized by mild temperatures and atmospheric pressure, facilitates easy scale-up from gram to ton quantities without requiring specialized high-pressure equipment. The absence of toxic heavy metals and strong oxidants simplifies the effluent treatment process, aligning with increasingly stringent environmental regulations. This green chemistry profile not only reduces the environmental footprint but also minimizes the administrative burden of waste management. Consequently, manufacturers can achieve higher production volumes with greater sustainability, appealing to eco-conscious stakeholders and regulatory bodies alike.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding this synthesis technology. They are derived from the detailed experimental data and comparative analysis provided in the patent documentation. Understanding these aspects helps stakeholders evaluate the feasibility of adopting this method for their specific production needs. The answers reflect the robustness and versatility of the copper-catalyzed system as demonstrated across numerous substrate examples.
Q: What are the key advantages of this copper-catalyzed method over traditional platinum or rhodium routes?
A: This method utilizes inexpensive and abundant copper salts instead of precious metals like platinum or rhodium, significantly lowering raw material costs. Additionally, it operates under milder conditions without requiring cryogenic temperatures or complex ligand systems.
Q: Does this synthesis require hazardous external oxidants?
A: No, the process ingeniously uses the N-O bond within the oxime ester substrate as an internal oxidant. This eliminates the need for strong, hazardous external oxidants like potassium persulfate, enhancing operational safety and simplifying waste treatment.
Q: What is the substrate scope for this pyrrolidone synthesis?
A: The method demonstrates excellent functional group tolerance, accommodating various substituted phenyl groups, heterocycles such as pyridine and furan, and different sulfonamide moieties, making it highly versatile for diverse drug discovery programs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Sulfonamide-Substituted Pyrrolidones Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this copper-catalyzed synthesis for the pharmaceutical industry. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods are seamlessly translated into robust industrial processes. Our state-of-the-art facilities are equipped to handle the specific requirements of this chemistry, maintaining stringent purity specifications through our rigorous QC labs. We are committed to delivering high-purity pharmaceutical intermediates that meet the exacting standards of global drug developers, leveraging our technical expertise to optimize yield and quality at every stage of production.
We invite you to collaborate with us to explore the full capabilities of this advanced synthesis route. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Please contact us to request specific COA data and route feasibility assessments for your target molecules. By partnering with NINGBO INNO PHARMCHEM, you gain access to a reliable supply chain partner dedicated to driving efficiency and innovation in your drug development pipeline.
