Revolutionizing Orobactit Production: Safe Suzuki Coupling for High-Purity Intermediates
Revolutionizing Orobactit Production: Safe Suzuki Coupling for High-Purity Intermediates
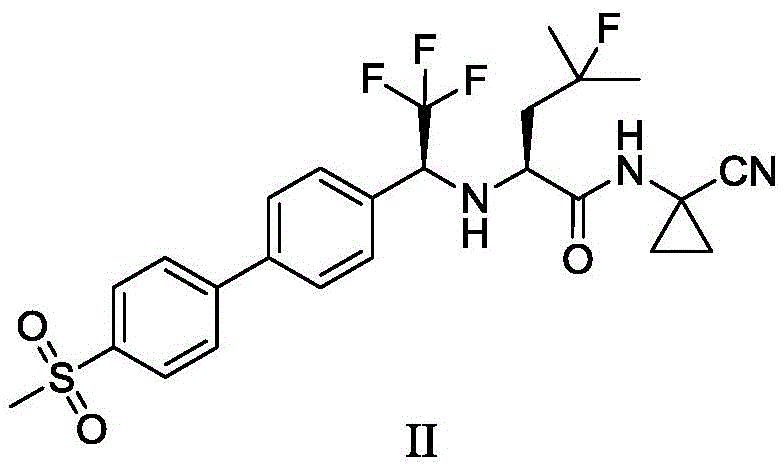
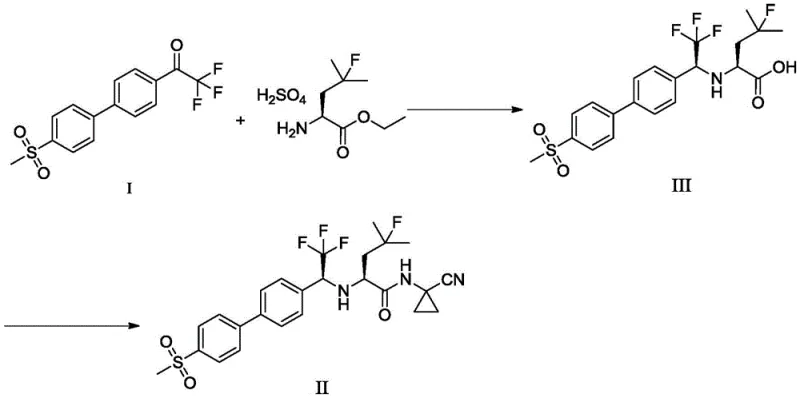
The pharmaceutical landscape for osteoporosis and bone metastasis treatments has been significantly advanced by the development of Orobactit (Odanacatib), a potent cathepsin K inhibitor. However, the commercial viability of such complex molecules often hinges on the efficiency and safety of their synthetic routes. Patent CN108912020B introduces a transformative preparation method for the critical trifluoroacetone biphenyl methylsulfone intermediate, designated as Intermediate I. This innovation addresses long-standing challenges in the synthesis of fluorinated biaryl scaffolds, shifting away from hazardous organometallic protocols toward a robust, palladium-catalyzed Suzuki-Miyaura coupling strategy. By leveraging this novel approach, manufacturers can achieve superior purity profiles while mitigating the severe safety risks associated with traditional lithiation chemistries.
This technical insight report analyzes the strategic advantages of adopting the methodology disclosed in CN108912020B. For R&D directors, the focus is on the mechanistic elegance and impurity control; for procurement and supply chain leaders, the emphasis lies on the drastic simplification of operations and the elimination of cryogenic dependencies. As a reliable pharmaceutical intermediate supplier, understanding these nuances is paramount for securing a competitive edge in the global API market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this invention, the synthesis of the trifluoroacetone biphenyl methylsulfone core relied heavily on a multi-step sequence originating from 4,4'-dibromobiphenyl, as illustrated in the literature (Tetrahedron; 2006, 62(21), 5092-5098). This legacy route is fraught with operational hazards and inefficiencies that render it suboptimal for large-scale industrial production. The process necessitates the use of highly flammable and pyrophoric reagents, specifically lithium tributylmagnesium and n-butyllithium, which must be handled under strictly anhydrous conditions at cryogenic temperatures ranging from -78°C to -10°C. Such extreme conditions not only escalate energy consumption but also introduce significant safety liabilities regarding thermal runaway and fire hazards.
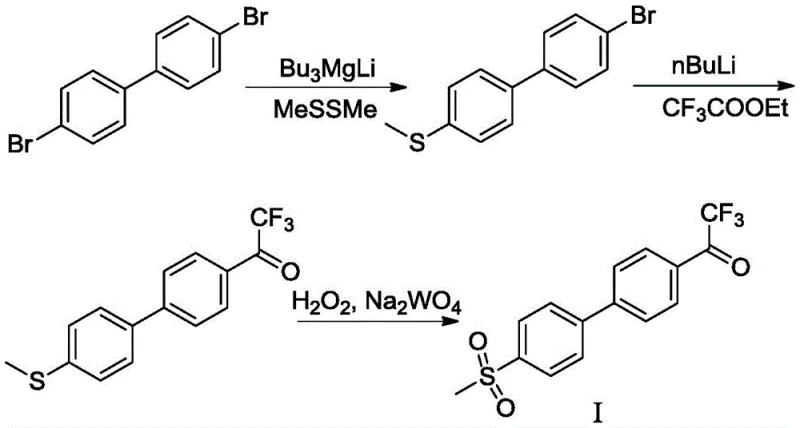
Furthermore, the conventional pathway involves the generation of malodorous thioether intermediates, creating unpleasant working environments and complicating waste management protocols. From a yield perspective, the cumulative efficiency of this linear sequence is disappointing, with the patent data indicating a total yield of merely 49.5% based on the starting dibromobiphenyl. The combination of low throughput, high safety risks, and environmental concerns makes this traditional method economically unsustainable for modern cost reduction in API manufacturing.
The Novel Approach
In stark contrast, the methodology disclosed in CN108912020B offers a streamlined, two-step convergent synthesis that bypasses the need for dangerous organolithium reagents entirely. The core innovation lies in the construction of the biaryl bond via a Suzuki-Miyaura cross-coupling reaction between 2,2,2-trifluoroacetophenone-4-boronic acid pinacol ester and 4-bromobenzenesulfone. This transformation is catalyzed by palladium complexes, such as tetrakis(triphenylphosphine)palladium, and proceeds under mild thermal conditions, typically between 70°C and 100°C. The use of a biphasic solvent system comprising aromatic hydrocarbons like toluene and water further enhances the reaction's practicality and safety profile.
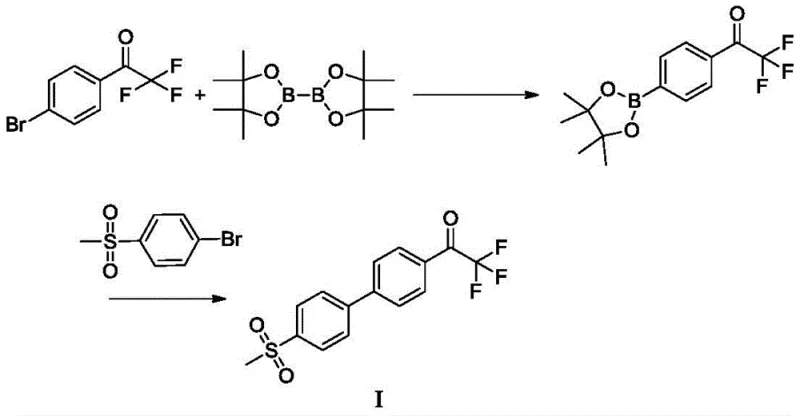
This novel approach delivers exceptional results, achieving isolated yields of Intermediate I exceeding 73%, with HPLC purity consistently above 99.00%. By eliminating the cryogenic steps and hazardous reagents, the new route drastically simplifies the operational complexity, making it highly suitable for commercial scale-up of complex fluorinated intermediates. The transition from a linear, hazard-heavy process to a convergent, catalytic one represents a paradigm shift in process chemistry, offering substantial cost savings and improved supply chain reliability.
Mechanistic Insights into Palladium-Catalyzed Suzuki Coupling
The success of this new synthetic route hinges on the efficiency of the palladium-catalyzed cross-coupling mechanism. The reaction initiates with the oxidative addition of the aryl bromide (4-bromobenzenesulfone) to the active Pd(0) species, forming an aryl-palladium(II) complex. This is followed by transmetallation, where the boronate species—activated by the inorganic base such as potassium phosphate or cesium carbonate—transfers its aryl group to the palladium center. The presence of water in the solvent mixture plays a crucial role in facilitating the formation of the reactive boronate 'ate' complex, thereby accelerating the transmetallation step. Finally, reductive elimination releases the desired biaryl product and regenerates the Pd(0) catalyst, closing the catalytic cycle.
Impurity control is another critical aspect addressed by this mechanism. The high chemoselectivity of the Suzuki coupling ensures that the sensitive trifluoromethyl ketone moiety remains intact, avoiding side reactions common with nucleophilic organolithium reagents. The patent specifies rigorous purification protocols, including quenching with ammonium chloride, washing with sodium bicarbonate and brine, and filtration through kieselguhr. A final recrystallization from cyclohexane at controlled temperatures (cooling to 0-5°C) effectively removes trace palladium residues and unreacted starting materials. This meticulous downstream processing guarantees that the intermediate meets stringent purity specifications (>99.42%), which is essential for preventing the carryover of genotoxic impurities into the final drug substance.
How to Synthesize Trifluoroacetone Biphenyl Methylsulfone Efficiently
Implementing this synthesis requires precise adherence to the optimized conditions outlined in the patent to maximize yield and safety. The process begins with the preparation of the boronic ester building block, followed by the key coupling reaction. Operators must maintain an inert atmosphere using nitrogen or argon to protect the catalyst from oxidation. The reaction progress should be monitored via HPLC or TLC, ensuring complete consumption of the boronic ester before proceeding to workup. The following overview illustrates the complete synthetic sequence from simple precursors to the final API, highlighting the pivotal role of Intermediate I.
- Prepare 2,2,2-trifluoroacetophenone-4-boronic acid pinacol ester via palladium-catalyzed borylation of 4'-bromo-2,2,2-trifluoroacetophenone.
- Perform Suzuki coupling between the boronic ester and 4-bromobenzenesulfone using a palladium catalyst and inorganic base in a toluene/water system.
- Purify the crude product through filtration, washing, and recrystallization from cyclohexane to achieve >99% purity.
Detailed standardized operating procedures for each step, including specific reagent grades, addition rates, and temperature ramping profiles, are critical for reproducibility.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of the CN108912020B process translates into tangible strategic benefits beyond mere chemical elegance. The elimination of cryogenic cooling requirements (-78°C) significantly reduces utility costs and capital expenditure on specialized low-temperature reactors. Furthermore, the replacement of short-shelf-life, hazardous organolithium reagents with stable, commercially available boronic esters and palladium catalysts enhances supply chain resilience. This stability allows for bulk purchasing and longer inventory holding periods without the risk of reagent degradation, thereby smoothing out production schedules and reducing lead time for high-purity pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The new route achieves a significant increase in overall yield compared to the prior art (73% vs 49.5%), which directly lowers the cost of goods sold (COGS). By removing the need for expensive, hazardous reagents like n-BuLi and the associated safety infrastructure, the process inherently drives down operational expenses. The simplified workup and purification steps also reduce solvent consumption and waste disposal costs, contributing to substantial cost savings in the overall manufacturing budget.
- Enhanced Supply Chain Reliability: Reliance on stable reagents such as pinacol boronic esters and aryl bromides mitigates the risk of supply disruptions often associated with hazardous chemicals that have strict transport regulations. The robustness of the Suzuki coupling allows for flexible batch sizing and easier technology transfer between manufacturing sites. This reliability ensures consistent delivery of high-quality intermediates, safeguarding the continuity of the downstream API production schedule against raw material volatility.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing common solvents like toluene and water that are easily recovered and recycled. The absence of malodorous thioethers and pyrophoric wastes simplifies environmental permitting and reduces the burden on effluent treatment plants. This alignment with green chemistry principles not only ensures regulatory compliance but also enhances the corporate sustainability profile, a key factor for partnerships with major multinational pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis route. These insights are derived directly from the experimental data and comparative examples provided in patent CN108912020B, offering clarity on the practical aspects of scaling this technology.
Q: Why is the new Suzuki coupling route safer than the traditional lithiation method?
A: The traditional method requires highly flammable lithium reagents like n-BuLi at cryogenic temperatures (-78°C), posing significant fire and explosion risks. The new Suzuki route operates at mild temperatures (70-100°C) using stable boronic esters and standard palladium catalysts, eliminating pyrophoric hazards.
Q: What purity levels can be achieved with this new intermediate synthesis?
A: The patent demonstrates that the trifluoroacetone biphenyl methylsulfone intermediate can be produced with HPLC purity exceeding 99.00%, often reaching 99.42%. This high purity facilitates the production of the final API, Orobactit, with purity greater than 99.5%.
Q: How does this process impact environmental compliance?
A: By avoiding malodorous thioether intermediates and hazardous organolithium waste streams, the new process significantly reduces environmental burden. The use of recyclable solvents like toluene and water, along with efficient workup procedures, aligns with modern green chemistry principles.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Orobactit Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust process chemistry in the successful commercialization of complex therapeutics like Orobactit. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to pilot plant is seamless and efficient. We are committed to delivering intermediates with stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify every batch against the highest industry standards.
We invite you to collaborate with us to leverage this advanced synthetic technology for your supply chain. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized processes can drive value and efficiency for your organization.
